Abstract
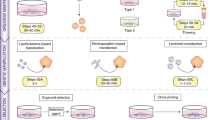
Methods to identify and enumerate primitive, and typically rare, undifferentiated cells in normal tissue using functional endpoints are powerful tools for acquiring insights into the mechanisms that regulate normal tissue stem cell turnover and differentiation. In this paper, we describe a xenotransplantation-based protocol that allows mammary stem cells with in vivo tissue regenerative properties to be specifically detected and quantified among the heterogeneous cell populations obtained from dissociated normal human mammary tissue. This methodology involves implanting a collagen gel containing the test cells in combination with supportive fibroblasts under the kidney capsule of highly immune-deficient, hormone-supplemented mice and then, 4 weeks later, searching for regenerated human cells with in vitro clonogenic activity. Quantification of the input human mammary stem cells is achieved using standard limiting dilution transplant approaches. This approach circumvents the need to modify the mouse mammary fat pad, and is objective, rapid (∼5 weeks) and economical to perform.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others

References
Stingl, J. et al. Purification and unique properties of mammary epithelial stem cells. Nature 439, 993–997 (2006).
Shackleton, M. et al. Generation of a functional mammary gland from a single stem cell. Nature 439, 84–88 (2006).
Stingl, J., Eaves, C.J., Kuusk, U. & Emerman, J.T. Phenotypic and functional characterization in vitro of a multipotent epithelial cell present in the normal adult human breast. Differentiation 63, 201–213 (1998).
Stingl, J., Eaves, C.J., Zandieh, I. & Emerman, J.T. Characterization of bipotent mammary epithelial progenitor cells in normal adult human breast tissue. Breast Cancer Res. Treat. 67, 93–109 (2001).
Al Hajj, M., Wicha, M.S., Benito-Hernandez, A., Morrison, S.J. & Clarke, M.F. Prospective identification of tumorigenic breast cancer cells. Proc. Natl. Acad. Sci. USA 100, 3983–3988 (2003).
Sheffield, L.G. & Welsch, C.W. Transplantation of human breast epithelia to mammary-gland-free fat-pads of athymic nude mice: influence of mammotrophic hormones on growth of breast epithelia. Int. J. Cancer 41, 713–719 (1988).
Kuperwasser, C. et al. Reconstruction of functionally normal and malignant human breast tissues in mice. Proc. Natl. Acad. Sci. USA 101, 4966–4971 (2004).
Proia, D.A. & Kuperwasser, C. Reconstruction of human mammary tissues in a mouse model. Nat. Protoc. 1, 206–214 (2006).
Lim, E. et al. Aberrant luminal progenitors as the candidate target population for basal tumor development in BRCA1. Nat. Med. 15, 907–913 (2009).
Stingl, J., Raouf, A., Emerman, J.T. & Eaves, C.J. Epithelial progenitors in the normal human mammary gland. J. Mammary Gland Biol. Neoplasia 10, 49–59 (2005).
Eirew, P. et al. A method for quantifying normal human mammary epithelial stem cells with in vivo regenerative ability. Nat. Med. 14, 1384–1389 (2008).
Buck, A.C. Differentiation of first- and second-set grafts of neonatal testis, ovary, intestine and spleen implanted beneath the kidney capsule of adult albino rat hosts. Am. J. Anat. 113, 189–213 (1963).
Parmar, H. et al. A novel method for growing human breast epithelium in vivo using mouse and human mammary fibroblasts. Endocrinology 143, 4886–4896 (2002).
Hu, Y. & Smyth, G.K. ELDA: extreme limiting dulution analysis for comparing depleted and enriched population in stem cell and other assays. J. Immunol. Methods 347, 70–78 (2009).
Richards, J. et al. Method for culturing mammary epithelial cells in a rat tail collagen gel matrix. Methods Cell Sci. 8, 31–36 (1983).
Laidlaw, I.J. et al. The proliferation of normal human breast tissue implanted into athymic nude mice is stimulated by estrogen but not progesterone. Endocrinology 136, 164–171 (1995).
Acknowledgements
We acknowledge the excellent technical contributions of D. Wilkinson, G. Edin, the staff of the Flow Cytometry Facility of the Terry Fox Laboratory and the Centre for Translational and Applied Genomics. J. Emerman helped organize the accrual of the mammoplasty material, which was obtained with informed patient consent, with the assistance of J. Sproul, P. Lennox, N. Van Laeken and R. Warren. The project was funded by grants from Genome BC/Genome Canada, the Canadian Stem Cell Network, the Canadian Breast Cancer Foundation BC and Yukon Division and the Canadian Cancer Society. P.E. was a recipient of a US Department of Defense Breast Cancer Research Program Studentship, a Terry Fox Foundation Research Studentship from the National Cancer Institute of Canada, a Canadian Imperial Bank of Commerce interdisciplinary award and a Canadian Stem Cell Network Studentship. J.S. held a Canadian Breast Cancer Foundation BC and Yukon Division Fellowship and a National Science and Engineering Research Council Industrial Fellowship.
Author information
Authors and Affiliations
Contributions
A.K. and J.E.G. designed the research; all authors conducted experiments and contributed to the writing of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Methods
Comparison of the statistical resolution of MRU frequency determinations by LDA versus “bulk CFC” endpoints. (DOC 30 kb)
Supplementary Figure 1
Simulation of the comparative ability of LDA and bulk CFC measurements to resolve differences in MRU frequencies that exist between 2 populations. The bars indicate the range of a measured MRU frequency for hypothetical sample #2 for which the null hypothesis of no statistical difference from measured MRU frequency = 100 for hypothetical sample #1 cannot be rejected with 95% confidence. These are obtained from Monte-Carlo simulations of experiments comprising 20 transplants (10 for each of the 2 samples). For the simulations using the LDA endpoint, transplants were assumed to be made at limiting dilution (i.e., an average of 1 MRU per transplant). Likelihood ratio tests for inequality in frequency between samples were carried out using the ELDA web tool2, which uses a generalized linear model assuming a single-hit Poisson process. For the simulations using the bulk CFC endpoint, the regenerated CFC output per transplant was assumed to follow a normal distribution with an average of 70 CFCs and a standard deviation of 30 CFCs, which is consistent with technical replicates series carried out by us previously (data not shown). Student t-tests were carried out to test for inequality. (PDF 21 kb)
Rights and permissions
About this article
Cite this article
Eirew, P., Stingl, J. & Eaves, C. Quantitation of human mammary epithelial stem cells with in vivo regenerative properties using a subrenal capsule xenotransplantation assay. Nat Protoc 5, 1945–1956 (2010). https://doi.org/10.1038/nprot.2010.148
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2010.148
This article is cited by
-
Single-cell analysis of autophagy activity in normal and de novo transformed human mammary cells
Scientific Reports (2020)
-
Isolation and characterization of a new basal-like luminal progenitor in human breast tissue
Stem Cell Research & Therapy (2019)
-
A robust cell culture system for large scale feeder cell-free expansion of human breast epithelial progenitors
Stem Cell Research & Therapy (2018)
-
Characterization of mammary epithelial stem/progenitor cells and their changes with aging in common marmosets
Scientific Reports (2016)
-
Barcoding reveals complex clonal dynamics of de novo transformed human mammary cells
Nature (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.