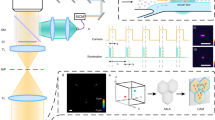
Abstract
The wealth of information available from advanced fluorescence imaging techniques used to analyze biological processes with high spatial and temporal resolution calls for high-throughput image analysis methods. Here, we describe a fully automated approach to analyzing cellular interaction behavior in 3D fluorescence microscopy images. As example application, we present the analysis of drug-induced and S1P1-knockout-related changes in bone–osteoclast interactions. Moreover, we apply our approach to images showing the spatial association of dendritic cells with the fibroblastic reticular cell network within lymph nodes and to microscopy data regarding T–B lymphocyte synapse formation. Such analyses that yield important information about the molecular mechanisms determining cellular interaction behavior would be very difficult to perform with approaches that rely on manual/semi-automated analyses. This protocol integrates adaptive threshold segmentation, object detection, adaptive color channel merging, and neighborhood analysis and permits rapid, standardized, quantitative analysis and comparison of the relevant features in large data sets.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Germain, R.N. et al. An extended vision for dynamic high-resolution intravital immune imaging. Semin. Immunol. 17, 431–441 (2005).
Stoll, S., Delon, J., Brotz, T.M. & Germain, R.N. Dynamic imaging of T cell–dendritic cell interactions in lymph nodes. Science 296, 1873–1876 (2002).
Xu, X., Meier-Schellersheim, M., Yan, J. & Jin, T. Locally controlled inhibitory mechanisms are involved in eukaryotic GPCR-mediated chemosensing. J. Cell Biol. 178, 141–153 (2007).
Fisher, D.T. et al. Hurdles to lymphocyte trafficking in the tumor microenvironment: implications for effective immunotherapy. Immunol. Invest. 35, 251–277 (2006).
Nishida, H. & Okabe, S. Visualization of synapse–glia dynamics. Brain Nerve 59, 755–761 (2007).
Wollman, R. & Stuurman, N. High throughput microscopy: from raw images to discoveries. J. Cell Sci. 120, 3715–3722 (2007).
Yamada, C. et al. The murine glucagon-like peptide-1 receptor is essential for control of bone resorption. Endocrinology 149, 574–579 (2008).
Nakamura, T. et al. Estrogen prevents bone loss via estrogen receptor alpha and induction of Fas ligand in osteoclasts. Cell 130, 811–823 (2007).
Lee, S.H. et al. v-ATPase V0 subunit d2-deficient mice exhibit impaired osteoclast fusion and increased bone formation. Nat. Med. 12, 1403–1409 (2006).
Castellino, F. et al. Chemokines enhance immunity by guiding naive CD8+ T cells to sites of CD4+ T cell–dendritic cell interaction. Nature 440, 890–895 (2006).
Henrickson, S.E. et al. T cell sensing of antigen dose governs interactive behavior with dendritic cells and sets a threshold for T cell activation. Nat. Immunol. 9, 282–291 (2008).
Ishii, M. et al. Sphingosine-1-phosphate mobilizes osteoclast precursors and regulates bone homeostasis. Nature 458, 524–528 (2009).
Qi, H., Cannons, J.L., Klauschen, F., Schwartzberg, P.L. & Germain, R.N. SAP-controlled T–B cell interactions underlie germinal centre formation. Nature 455, 764–769 (2008).
Sezgin, H. & Sankur, B. Survey over image thresholding techniques and quantitative performance evaluation. J. Electron. Imaging 13, 146–168 (2004).
Suzuki, K., Horiba, I. & Sugie, N. Linear-time connected-component labeling based on sequential local operations. Comput. Vis. Image Underst. 89, 1–23 (2003).
Kervrann, C. & Trubuli, A. An adaptive window approach for Poisson noise reduction and structure preserving in confocal microscopy. IEEE Int. Symp. Biomed. Imaging 1, 788–791 (2004).
Bar-Shavit, Z. The osteoclast: a multinucleated, hematopoietic-origin, bone-resorbing osteoimmune cell. J. Cell. Biochem. 102, 1130–1139 (2007).
Wada, T. et al. The molecular scaffold Gab2 is a crucial component of RANK signaling and osteoclastogenesis. Nat. Med. 11, 394–399 (2005).
Parfitt, A.M. et al. Bone histomorphometry: standardization of nomenclature, symbols, and units. Report of the ASBMR Histomorphometry Nomenclature Committee. J. Bone Miner Res. 2, 595–610 (1987).
Acknowledgements
This research was supported by the Intramural Research Program of NIAID, NIH. M.I. was supported by a fellowship grant from the International Human Frontier Science Program.
Author information
Authors and Affiliations
Contributions
F.K. designed, implemented and tested the method. F.K., M.M.-S. and R.N.G. prepared the paper. M.M.-S. and R.N.G. supervised the project. M.I., H.Q., M.B., J.G.E. and F.K. generated and provided experimental data.
Corresponding authors
Supplementary information
Rights and permissions
About this article
Cite this article
Klauschen, F., Ishii, M., Qi, H. et al. Quantifying cellular interaction dynamics in 3D fluorescence microscopy data. Nat Protoc 4, 1305–1311 (2009). https://doi.org/10.1038/nprot.2009.129
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2009.129
This article is cited by
-
In Vivo 3D Histomorphometry Quantifies Bone Apposition and Skeletal Progenitor Cell Differentiation
Scientific Reports (2018)
-
CognitionMaster: an object-based image analysis framework
Diagnostic Pathology (2013)
-
New TACTICS for finding Numb
Immunology & Cell Biology (2013)
-
Dynamic live imaging of bone: opening a new era with ‘bone histodynametry’
Journal of Bone and Mineral Metabolism (2013)
-
In vivo imaging of therapy-induced anti-cancer immune responses in humans
Cellular and Molecular Life Sciences (2013)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.