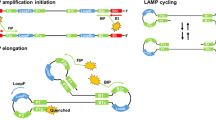
Abstract
As the human genome is decoded and its involvement in diseases is being revealed through postgenome research, increased adoption of genetic testing is expected. Critical to such testing methods is the ease of implementation and comprehensible presentation of amplification results. Loop-mediated isothermal amplification (LAMP) is a simple, rapid, specific and cost-effective nucleic acid amplification method when compared to PCR, nucleic acid sequence-based amplification, self-sustained sequence replication and strand displacement amplification. This protocol details an improved simple visual detection system for the results of the LAMP reaction. In LAMP, a large amount of DNA is synthesized, yielding a large pyrophosphate ion by-product. Pyrophosphate ion combines with divalent metallic ion to form an insoluble salt. Adding manganous ion and calcein, a fluorescent metal indicator, to the reaction solution allows a visualization of substantial alteration of the fluorescence during the one-step amplification reaction, which takes 30–60 min. As the signal recognition is highly sensitive, this system enables visual discrimination of results without costly specialized equipment. This detection method should be helpful in basic research on medicine and pharmacy, environmental hygiene, point-of-care testing and more.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
12 January 2012
In the version of this article initially published, the legend for Figure 1 contained errors. In the description of panel b, the sentence "The released single strand forms a loop structure at its 3′ end (structure 3)" should have ended with "a loop structure at its 5′ end (structure 4)." In the description of panel c, "Using self-structure as the template..." should have read "Using structure 5 as the template..." The last sentence of the legend ("Structures 9 and 10 are produced from structures 6 and 8, respectively, and more elongated structures (11, 12) are also produced.") has been replaced with the following: "Specifically, intermediate structures 7a and 9a and structures 5a and 10a (in the yellow boxes) are produced from structures 6 and 8, respectively. Structures 9a and 10a then form structures 9 and 10, respectively, whereas the displaced strands 7a and 5a form the dumbbell-like structures 7 and 5, respectively. More elongated structures (11, 12) are also produced." Finally, in panel c of the figure, structure 9 (with downstream structure 11) and structure 10 (with downstream structure 12) were shown in incorrect locations, and additional structures (5a, 7a, 9a, and 10a) were omitted. These errors have been corrected in the HTML and PDF versions of the article. The authors acknowledge S. Subramanian and R.D. Gomez of the University of Maryland, who brought the error to their attention and supplied the corrected version of Figure 1c and legend.
References
Saiki, R.K. et al. Enzymatic amplification of β-globin genomic sequences and restriction site analysis for diagnosis of sickle cell anemia. Science 230, 1350–1354 (1985).
Higuchi, R., Dollinger, G., Walsh, P.S. & Griffith, R. Simultaneous amplification and detection of specific DNA sequences. Biotechnology (NY) 10, 413–417 (1992).
Higuchi, R., Fockler, C., Dollinger, G. & Watson, R. Kinetic PCR analysis: real-time monitoring of DNA amplification reactions. Biotechnology (NY) 11, 1026–1030 (1993).
Holland, P.M., Abramson, R.D., Watson, R. & Gelfand, D.H. Detection of specific polymerase chain reaction product by utilizing the 5′→3′ exonuclease activity of Thermus aquaticus DNA polymerase. Proc. Natl. Acad. Sci. USA 88, 7276–7280 (1991).
Tyagi, S. & Kramer, F.R. Molecular beacons: probes that fluoresce upon hybridization. Nat. Biotechnol. 14, 303–308 (1996).
Whitcombe, D., Theaker, J., Guy, S.P., Brown, T. & Little, S. Detection of PCR products using self-probing amplicons and fluorescence. Nat. Biotechnol. 17, 804–807 (1999).
Zhang, D.Y., Brandwein, M., Hsuih, T.C. & Li, H. Amplification of target-specific, ligation-dependent circular probe. Gene 211, 277–285 (1998).
Compton, J. Nucleic acid sequence-based amplification. Nature 350, 91–92 (1991).
Guatelli, J.C. et al. Isothermal, in vitro amplification of nucleic acids by a multienzyme reaction modeled after retroviral replication. Proc. Natl. Acad. Sci. USA 87, 1874–1878 (1990).
Walker, G.T. et al. Strand displacement amplification—an isothermal, in vitro DNA amplification technique. Nucleic Acids Res. 20, 1691–1696 (1992).
Notomi, T. et al. Loop-mediated isothermal amplification of DNA. Nucleic Acids Res. 28, e63 (2000).
Fukuta, S. et al. Detection of Japanese yam mosaic virus by RT-LAMP. Archives of Virology 148, 1713–1720 (2003).
Mori, Y., Nagamine, K., Tomita, N. & Notomi, T. Detection of loop-mediated isothermal amplification reaction by turbidity derived from magnesium pyrophosphate formation. Biochem. Biophys. Res. Commun. 289, 150–154 (2001).
Diehl, H. & Ellingboe, J.L. Indicator for titration of calcium in presence of magnesium using disodium dihydrogen ethylenediamine tetraacetate. Anal. Chem. 28, 882–884 (1956).
Kepner, B.L. & Hercules, D.M. Fluorometric determination of calcium in blood serum. Anal. Chem. 35, 1238–1240 (1963).
Hoelzl Wallach, D.F. & Steck, T.L. Fluorescence techniques in the microdetermination of metals in biological materials. Utility of 2, 4-bis-(N,N′-di-(carboxymethyl)aminomethyl) fluorescein in the fluorometric estimation of Al+3, alkaline earths, Co+2, Cu+2, Ni+2, and Zn+2 in micromolar concentrations. Anal. Chem. 35, 1035–1044 (1963).
Körbl, J. & Vydra, F. Metallochromic indicators. IV. A note on the preparation and properties of “Calcein”. Collect. Czech. Chem. Commun. 23, 622–627 (1958).
Demertzis, M.A. Fluorimetric determination of calcium in serum with calcein: complexation of calcein with calcium and alkali metals. Anal. Chim. Acta. 209, 303–308 (1988).
Nagamine, K., Watanabe, K., Ohtsuka, K., Hase, T. & Notomi, T. Loop-mediated isothermal amplification reaction using a nondenatured template. Clin. Chem. 47, 1742–1743 (2001).
Acknowledgements
We thank S. Kojiya for help in preparing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tomita, N., Mori, Y., Kanda, H. et al. Loop-mediated isothermal amplification (LAMP) of gene sequences and simple visual detection of products. Nat Protoc 3, 877–882 (2008). https://doi.org/10.1038/nprot.2008.57
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2008.57
This article is cited by
-
Development and clinical application of loop-mediated isothermal amplification combined with lateral flow assay for rapid diagnosis of SARS-CoV-2
BMC Infectious Diseases (2024)
-
Cross-pathogenicity of Phytophthora palmivora associated with bud rot disease of oil palm and development of biomarkers for detection
World Journal of Microbiology and Biotechnology (2024)
-
Ultrasensitive and fast detection of SARS-CoV-2 using RT-LAMP without pH-dependent dye
Functional & Integrative Genomics (2024)
-
Loop-Mediated Isothermal Amplification for On-Site Visual Identification of Leech Species
Food Analytical Methods (2024)
-
Optimization and comparative analysis of LAMP and PCR techniques for the detection of leptospiral DNA in Golden Syrian hamsters
Veterinary Research Communications (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.