Abstract
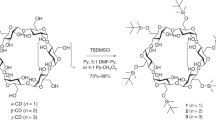
We describe a protocol for the synthesis of mono-6-amino-6-deoxy-cyclodextrin hydrochloride (CD-NH3Cl), applicable to α-, β- and γ-cyclodextrin. These structurally simplest, highly water-soluble cationic cyclodextrins can be widely used in molecular recognition, chiral separation and drug delivery studies. Starting from commercially available chemicals, CD-NH3Cl is synthesized in four steps: (i) selective tosylation of cyclodextrin by the use of p-toluenesulfonyl chloride to afford mono-6-(p-toluenesulfonyl)-6-deoxy-cyclodextrin (Ts-CD); (ii) azide substitution of Ts-CD with sodium azide to afford mono-6-azido-6-deoxy-cyclodextrin (CD-N3); (iii) reduction of CD-N3 with triphenylphospine followed by hydrolysis to prepare mono-6-amino-6-deoxy-cyclodextrin (CD-NH2); and (iv) treatment of CD-NH2 with hydrochloric acid to afford the titled CD-NH3Cl with good yield. The overall protocol requires ∼2 weeks.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Easton, C.J. & Lincoln, S.F. Chiral discrimination by modified cyclodextrins. Chem. Soc. Rev. 25, 163–170 (1996).
Liu, Y. & Chen, Y. Cooperative binding and multiple recognition by bridged bis(β- cyclodextrin)s with functional linkers. Acc. Chem. Res. 39, 681–691 (2006).
Armstrong, D.W., Ward, T.J., Armstrong, R.D. & Beesley, T.E. Separation of drug stereoisomers by the formation of β-cyclodextrin inclusion complexes. Science 232, 1132–1135 (1986).
Uekama, K., Hirayama, F. & Irie, T. Cyclodextrin drug carrier systems. Chem. Rev. 98, 2045–2076 (1998).
Harata, K. Structural aspects of stereodifferentiation in the solid state. Chem. Rev. 98, 1803–1827 (1998).
Fujimura, K., Suzuki, S., Hayashi, K. & Masuda, S. Retention behavior and chiral recognition mechanism of several cyclodextrin-based stationary phases for dansyl amino acids. Anal. Chem. 62, 2198–2205 (1990).
Rekharsky, M.V. & Inoue, Y. Complexation thermodynamics of cyclodextrins. Chem. Rev. 98, 1875–1917 (1998).
Hapiot, F. & Monflier, E. cyclodextrins as supramolecular hosts for organometallic complexes. Chem. Rev. 106, 767–781 (1998).
Tang, W.H., Ong, T.T., Muderawan, I.W., Ng, S.C. & Chan, H.S.O. Synthesis and applications of single-isomer 6-mono(alkylimidazolium)-β-cyclodextrins as chiral selectors in chiral capillary electrophoresis. Electrophoresis 26, 3839–3848 (2005).
Khan, A.R., Forgo, P., Stine, K.J. & D'Souza, V.T. Methods for selective modifications of cyclodextrins. Chem. Rev. 98, 1977–1996 (1998).
de Boer, T., de Zeeuw, R.A., de Jong, G.J. & Ensing, K. Recent innovations in the use of charged cyclodextrins in capillary electrophoresis for chiral separations in pharmaceutical analysis. Electrophoresis 21, 3220–3239 (2000).
Brown, S.E., Coates, J.H., Coghlan, D.R. & Easton, C.J. Synthesis and properties of 6A- amino-6A-deoxy-α -and β-cyclodextrin. Aust. J. Chem. 46, 953–958 (1992).
Petter, R.C., Salek, J.S., Sikorski, C.T., Kumaravel, G. & Lin, F.T. Cooperative binding by aggregated mono-6-(alkylamino)-β-cyclodextrins. J. Am. Chem. Soc. 112, 3860–3868 (1990).
Tang, W.H., Muderawan, I.W., Ong, T.T., Ng, S.C. & Chan, H.S.O. Synthesis and application of mono-6-ammonium-6-deoxy-β-cyclodextrin chloride as chiral selector for capillary electrophoresis. J. Chromatogr. A 1094, 187–191 (2005).
Tang, W.H., Muderawan, I.W., Ong, T.T. & Ng, S.C. Facile synthesis and enantioseparation performance of positively charged single-isomers of α- and γ- cyclodextrin. Tetrahedron Asymmetry 18, 1548–1553 (2007).
Muderawan, I.W., Ong, T.T., Lee, T.C., Young, D.J., Ching, C.B. & Ng, S.C. A reliable synthesis of 2- and 6-amino-β-cyclodextrin and permethylated- β- cyclodextrin. Tetrahedron Lett. 46, 7905–7907 (2005).
Tang, W.H. & Ng, S.C. Synthesis of cationic single-isomer cyclodextrins for the chiral separation of amino acids and anionic pharmaceuticals. Nat. Protoc. 2, 3195–3200 (2007).
Arita, S., Yabuuchi, T. & Kusumi, T. Resolution of 1- and 2-naphthylmethoxyacetic acids, NMR reagents for absolute configuration determination, by use of l- phenylalaninol. Chirality 15, 609–614 (2003).
Acknowledgements
We are grateful to the National University of Singapore (NUS) and Institute of Chemical and Engineering Sciences, Singapore for financial support. W.T. expresses deepest gratitude to S.-C.N., Professor Chan Sze On, Hardy, Dr. Ong Teng Teng and Dr. I. Wayan Muderawan for their invaluable advice and great help in the completion of his research works on cationic cyclodextrins.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Tang, W., Ng, SC. Facile synthesis of mono-6-amino-6-deoxy-α-, β-, γ-cyclodextrin hydrochlorides for molecular recognition, chiral separation and drug delivery. Nat Protoc 3, 691–697 (2008). https://doi.org/10.1038/nprot.2008.37
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2008.37
This article is cited by
-
Mirror-image cyclodextrins
Nature Synthesis (2024)
-
Polyamide 6/MXene-grafted graphene oxide hybrid nanocomposites
Iranian Polymer Journal (2023)
-
New ternary water-soluble support from self-assembly of β-cyclodextrin-ionic liquid and an anionic polymer for a dialysis device
Environmental Science and Pollution Research (2022)
-
Sensitive determination of ascorbic acid, dopamine and uric acid by glassy carbon electrodes modified with β-cyclodextrin and graphene oxide
Journal of Materials Science: Materials in Electronics (2022)
-
Synthesis of cyclodextrin–pyrrole conjugates possessing tuneable carbon linkers
Journal of Inclusion Phenomena and Macrocyclic Chemistry (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.