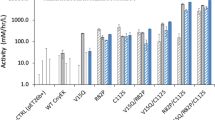
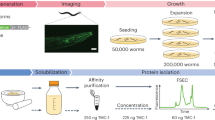
Abstract
Cell-free expression is emerging as a prime method for the rapid production of preparative quantities of high-quality membrane protein samples. The technology facilitates easy access to large numbers of proteins that have been extremely difficult to obtain. Most frequently used are cell-free systems based on extracts of Escherichia coli cells, and the reaction procedures are reliable and efficient. This protocol describes the preparation of all essential reaction components such as the E. coli cell extract, T7 RNA polymerase, DNA templates as well as the individual stock solutions. The setups of expression reactions in analytical and preparative scales, including a variety of reaction designs, are illustrated. We provide detailed reaction schemes that allow the preparation of milligram amounts of functionally folded membrane proteins of prokaryotic and eukaryotic origin in less than 24 h.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Yokoyama, S. Protein expression systems for structural genomics and proteomics. Curr. Opin. Chem. Biol. 7, 39–43 (2003).
Klammt, C. et al. High level cell-free expression and specific labeling of integral membrane proteins. Eur. J. Biochem. 271, 568–580 (2004).
Klammt, C. et al. Cell-free production of G protein-coupled receptors for functional and structural studies. J. Struct. Biol. 158, 482–493 (2007).
Schwarz, D. et al. Preparative scale cell-free expression systems: new tools for the large scale preparation of integral membrane proteins for functional and structural studies. Methods 41, 355–369 (2007).
Berrier, C. et al. Cell-free synthesis of a functional ion channel in the absence of a membrane and in the presence of detergent. Biochemistry 43, 12585–12591 (2004).
Ishihara, G. et al. Expression of G protein coupled receptors in a cell-free translational system using detergents and thioredoxin-fusion vectors. Protein Expr. Purif. 41, 27–37 (2005).
Elbaz, Y., Steiner-Mordoch, S., Danieli, T. & Schuldiner, S. In vitro synthesis of fully functional EmrE, a multidrug transporter, and study of its oligomeric state. Proc. Natl. Acad. Sci. USA 101, 1519–15124 (2004).
Savage, D.F., Anderson, C.L., Robles-Colmenares, Y., Newby, Z.E. & Stroud, R.M. Cell-free complements in vivo expression of the E. coli membrane proteome. Protein Sci. 16, 966–976 (2007).
Shimada, Y., Wang, Z.Y., Mochizuki, Y., Kobayashi, M. & Nozawa, T. Functional expression and characterization of a bacterial light-harvesting membrane protein in Escherichia coli and cell-free synthesis systems. Biosci. Biotechnol. Biochem. 68, 1942–1948 (2004).
Spirin, A.S., Baranov, V.I., Ryabova, L.A., Ovodov, S.Y. & Alakhov, Y.B. A continuous cell-free translation system capable of producing polypeptides in high yield. Science 242, 1162–1164 (1988).
Kigawa, T. & Yokoyama, S. A continuous cell-free protein synthesis system for coupled transcription–translation. J. Biochem. (Tokyo) 110, 166–168 (1991).
Shirokov, V.A., Kommer, A., Kolb, V.A. & Spirin, A.S. Continuous-exchange protein-synthesizing systems. in Methods of Molecular Biology (ed., Grandi, G.) 19–57 (Humana Press, Totowa, NJ, 2007).
Kim, T.W. et al. An economical and highly productive cell-free protein synthesis system utilizing fructose-1,6-bisphosphate as an energy source. J. Biotechnol. 130, 389–393 (2007).
Zubay, G. In vitro synthesis of protein in microbial systems. Annu. Rev. Genet. 7, 267–287 (1973).
Klammt, C., Schwarz, D., Dötsch, V. & Bernhard, F. Cell-free production of integral membrane proteins on a preparative scale. in Methods of Molecular Biology (ed., Grandi, G.) 57–79 (Humana Press, Totowa, NJ, 2007).
Li, Y., Wang, E. & Wang, Y. A modified procedure for fast purification of T7 RNA polymerase. Protein Expr. Purif. 16, 355–358 (1999).
Trbovic, N. et al. Efficient strategy for the rapid backbone assignment of membrane proteins. J. Am. Chem. Soc. 127, 13504–13505 (2005).
Ozawa, K., Dixon, N.E. & Otting, G. Cell-free synthesis of 15N-labeled proteins for NMR studies. IUBMB Life 57, 615–622 (2005).
Kainosho, M. et al. Optimal isotope labelling for NMR protein structure determinations. Nature 440, 52–57 (2006).
Kim, D.M. & Swartz, J.R. Prolonging cell-free protein synthesis by selective reagent additions. Biotechnol. Prog. 16, 385–390 (2000).
Kim, R.G. & Choi, C.Y. Expression-independent consumption of substrates in cell-free expression system from Escherichia coli. J. Biotechnol. 84, 27–32 (2001).
Ryabova, L.A., Vinokurov, L.M., Shekhovtsova, E.A., Alakhov, Y.B. & Spirin, A.S. Acetyl phosphate as an energy source for bacterial cell-free translation systems. Anal. Biochem. 226, 184–186 (1995).
Anderson, C.W., Straus, J.W. & Dudock, B.S. Preparation of a cell-free protein-synthesizing system from wheat germ. Methods Enzymol. 101, 635–644 (1983).
Spirin, A.S. High-throughput cell-free systems for synthesis of functionally active proteins. Trends Biotechnol. 22, 538–545 (2004).
Kim, D.M. & Swartz, J.R. Prolonging cell-free protein synthesis with a novel ATP regeneration system. Biotechnol. Bioeng. 66, 180–188 (1999).
Jewett, M.C. & Swartz, J.R. Mimicking the Escherichia coli cytoplasmic environment activates long-lived and efficient cell-free protein synthesis. Biotechnol. Bioeng. 86, 19–26 (2004).
Kim, T.W., Kim, D.M. & Choi, C.Y. Rapid production of milligram quantities of proteins in a batch cell-free protein synthesis system. J. Biotechnol. 124, 373–380 (2006).
Gonzalez de Valdivia, E.I. & Isaksson, L.A. A codon window in mRNA downstream of the initiation codon where NGG codons give strongly reduced gene expression in Escherichia coli. Nucleic Acids Res. 32, 5198–5205 (2004).
Hahn, G.H. & Kim, D.M. Production of milligram quantities of recombinant proteins from PCR-amplified DNAs in a continuous-exchange cell-free protein synthesis system. Anal. Biochem. 355, 151–153 (2006).
Ahn, J.H. et al. Cell-free synthesis of recombinant proteins from PCR-amplified genes at a comparable productivity to that of plasmid-based reactions. Biochem. Biophys. Res. Commun. 338, 1346–1352 (2005).
Kalmbach, R. et al. Functional cell-free synthesis of a seven helix membrane protein: in situ insertion of bacteriorhodopsin into liposomes. J. Mol. Biol. 371, 639–648 (2007).
Klammt, C. et al. Evaluation of detergents for the soluble expression of alpha-helical and beta-barrel-type integral membrane proteins by a preparative scale individual cell-free expression system. FEBS J. 272, 6024–6038 (2005).
Klammt, C. et al. Cell-free expression as an emerging technique for the large scale production of integral membrane protein. FEBS J. 273, 4141–4153 (2006).
Klammt, C. et al. Functional analysis of cell-free-produced human endothelin B receptor reveals transmembrane segment 1 as an essential area for ET-1 binding and homodimer formation. FEBS J. 274, 3257–3269 (2007).
Katzen, F. & Kudlicki, W. Efficient generation of insect-based cell-free translation extracts active in glycosylation and signal sequence processing. J. Biotechnol. 125, 194–197 (2006).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schwarz, D., Junge, F., Durst, F. et al. Preparative scale expression of membrane proteins in Escherichia coli-based continuous exchange cell-free systems. Nat Protoc 2, 2945–2957 (2007). https://doi.org/10.1038/nprot.2007.426
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2007.426
This article is cited by
-
Cryo-EM structure of cell-free synthesized human histamine 2 receptor/Gs complex in nanodisc environment
Nature Communications (2024)
-
E. coli “Stablelabel” S30 lysate for optimized cell-free NMR sample preparation
Journal of Biomolecular NMR (2023)
-
Cell-free synthesis of amyloid fibrils with infectious properties and amenable to sub-milligram magic-angle spinning NMR analysis
Communications Biology (2022)
-
Towards a generic prototyping approach for therapeutically-relevant peptides and proteins in a cell-free translation system
Nature Communications (2022)
-
Switching at the ribosome: riboswitches need rProteins as modulators to regulate translation
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.