Abstract
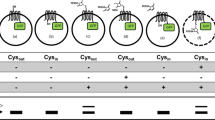
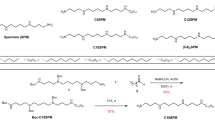
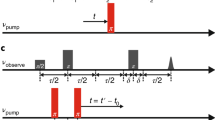
This protocol describes a detailed method to study the static and dynamic features of membrane proteins, as well as solvent accessibility, by utilizing the lactose permease of Escherichia coli (LacY) as a model. The method relies on the use of functional single-Cys mutants, an affinity tag and a PhosphoImager. The membrane-permeant, radioactive thiol reagent N-[ethyl-1-14C]ethylmaleimide ([14C]NEM) is used to detect site-directed alkylation of engineered single-Cys mutants in situ. The solvent accessibility of the Cys residues is also determined by blockage of [14C]NEM labeling with membrane-impermeant thiol reagents such as methanethiosulfonate ethylsulfonate (MTSES). The labeled proteins are purified by mini-scale affinity chromatography and analyzed by gel electrophoresis. Gels are dried and exposed to a PhosphoImager screen for 1–5 d, and incorporation of radioactivity is visualized. Initial results can be obtained in 24 h.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Frillingos, S., Sahin-Tóth, M., Persson, B. & Kaback, H.R. Cysteine-scanning mutagenesis of putative helix VII in the lactose permease of Escherichia coli. Biochemistry 33, 8074–8081 (1994).
Frillingos, S., Sahin-Toth, M., Wu, J. & Kaback, H.R. Cys-scanning mutagenesis: a novel approach to structure function relationships in polytopic membrane proteins. FASEB J. 12, 1281–1299 (1998).
Kaback, H.R. et al. Site-directed alkylation and the alternating access model for LacY. Proc. Natl. Acad. Sci. USA 104, 491–494 (2007).
Kaback, H.R., Sahin-Toth, M. & Weinglass, A.B. The kamikaze approach to membrane transport. Nat. Rev. Mol. Cell Biol. 2, 610–620 (2001).
Guan, L. & Kaback, H.R. Lessons from lactose permease. Annu. Rev. Biophys. Biomol. Struct. 35, 67–91 (2006).
Akabas, M.H., Stauffer, D.A., Xu, M. & Karlin, A. Acetylcholine receptor channel structure probed in cysteine-substitution mutants. Science 258, 307–310 (1992).
Karlin, A. & Akabas, M.H. Substituted-cysteine accessibility method. Methods Enzymol. 293, 123–145 (1998).
Guan, L., Sahin-Toth, M., Kalai, T., Hideg, K. & Kaback, H.R. Probing the mechanism of a membrane transport protein with affinity inactivators. J. Biol. Chem. 278, 10641–10648 (2003).
Kwaw, I., Zen, K.C., Hu, Y. & Kaback, H.R. Site-directed sulfhydryl labeling of the lactose permease of Escherichia coli: helices IV and V that contain the major determinants for substrate binding. Biochemistry 40, 10491–10499 (2001).
Frillingos, S. & Kaback, H.R. Probing the conformation of the lactose permease of Escherichia coli by in situ site-directed sulfhydryl modification. Biochemistry 35, 3950–3956 (1996).
Sahin-Tóth, M., Akhoon, K.M., Runner, J. & Kaback, H.R. Ligand recognition by the lactose permease of Escherichia coli: specificity and affinity are defined by distinct structural elements of galactopyranosides. Biochemistry 39, 5097–5103 (2000).
Consler, T.G. et al. Properties and purification of an active biotinylated lactose permease from Escherichia coli. Proc. Natl. Acad. Sci. USA 90, 6934–6938 (1993).
Pouny, Y., Weitzman, C. & Kaback, H.R. In vitro biotinylation provides quantitative recovery of highly purified active lactose permease in a single step. Biochemistry 37, 15713–15719 (1998).
Falke, J.J., Bass, R.B., Butler, S.L., Chervitz, S.A. & Danielson, M.A. The two-component signaling pathway of bacterial chemotaxis: a molecular view of signal transduction by receptors, kinases, and adaptation enzymes. Annu. Rev. Cell Dev. Biol. 13, 457–512 (1997).
Yang, Q. et al. Experimental tests of a homology model for OxlT, the oxalate transporter of Oxalobacter formigenes. Proc. Natl. Acad. Sci. USA 102, 8513–8518 (2005).
Koide, K., Maegawa, S., Ito, K. & Akiyama, Y. Environment of the active site region of RseP, an Escherichia coli regulated intramembrane proteolysis protease, assessed by site-directed cysteine alkylation. J. Biol. Chem. 282, 4553–4560 (2007).
Kaback, H.R. Transport in isolated bacterial membrane vesicles. Methods Enzymol. 31, 698–709 (1974).
Short, S.A., Kaback, H.R. & Kohn, L.D. Localization of d-lactate dehydrogenase in native and reconstituted Escherichia coli membrane vesicles. J. Biol. Chem. 250, 4291–4296 (1975).
Guan, L. & Kaback, H.R. Binding affinity of lactose permease is not altered by the H+ electrochemical gradient. Proc. Natl. Acad. Sci. USA 101, 12148–12152 (2004).
Venkatesan, P., Hu, Y. & Kaback, H.R. Site-directed sulfhydryl labeling of the lactose permease of Escherichia coli: helix X. Biochemistry 39, 10656–10661 (2000).
Venkatesan, P., Liu, Z., Hu, Y. & Kaback, H.R. Site-directed sulfhydryl labeling of the lactose permease of Escherichia coli: helix II. Biochemistry 39, 10649–10655 (2000).
Acknowledgements
The authors acknowledge support from National Institutes of Health (NIH) grants DK51131 and DK06946, GM074929 and National Science Foundation (NSF) grant 0450970 (to H.R.K.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Guan, L., Ronald Kaback, H. Site-directed alkylation of cysteine to test solvent accessibility of membrane proteins. Nat Protoc 2, 2012–2017 (2007). https://doi.org/10.1038/nprot.2007.275
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2007.275
This article is cited by
-
Plant glucose transporter structure and function
Pflügers Archiv - European Journal of Physiology (2020)
-
Site-Directed Alkylation Detected by In-Gel Fluorescence (SDAF) to Determine the Topology Map and Probe the Solvent Accessibility of Membrane Proteins
Scientific Reports (2019)
-
Functional Consequences of Sulfhydryl Modification of the γ-Aminobutyric Acid Transporter 1 at a Single Solvent-Exposed Cysteine Residue
The Journal of Membrane Biology (2012)
-
New Protein Footprinting: Fast Photochemical Iodination Combined with Top-Down and Bottom-Up Mass Spectrometry
Journal of the American Society for Mass Spectrometry (2012)
-
The Alternating Access Transport Mechanism in LacY
The Journal of Membrane Biology (2011)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.