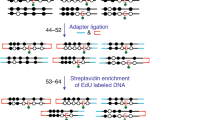
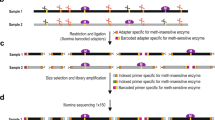
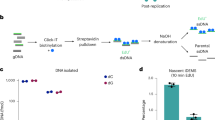
Abstract
DNA methylation is an important epigenetic modification of DNA in mammalian genomes. DNA methylation patterns are established early in development, modulated during tissue-specific differentiation and disrupted in many disease states, including cancer. To understand further the biological functions of these changes, accurate and reproducible methods are required to fully analyze the DNA methylation sequence. Here, we describe the 'gold-standard' bisulphite conversion protocol that can be used to re-sequence DNA from mammalian cells in order to determine and quantify the methylation state of a gene or genomic region at single-nucleotide resolution. The process of bisulphite treatment exploits the different sensitivities of cytosine and 5-methylcytosine (5-MeC) to deamination by bisulphite under acidic conditions—in which cytosine undergoes conversion to uracil, whereas 5-MeC remains unreactive. Bisulphite conversion of DNA, in either single tubes or in a 96-well format, can be performed in a minimum of 8 h and a maximum of 18 h, depending on the amount and quality of starting DNA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Warnecke, P.M. & Clark, S.J. DNA methylation profile of the mouse skeletal alpha-actin promoter during development and differentiation. Mol. Cell. Biol. 19, 164–172 (1999).
Jones, P.A. Overview of cancer epigenetics. Semin. Hematol. 42, S3–S8 (2005).
Grigg, G. & Clark, S. Sequencing 5-methylcytosine residues in genomic DNA. Bioessays 16, 431–436 (1994).
Clark, S.J. & Frommer, M. in DNA and Nucleoprotein Structure In Vivo Springer-Verlag (eds. Saluz, H.P. & Wiebauer, K.) 123–132 (R.G. Landes Company, Austin, Texas, 1995).
Clark, S.J. & Frommer, M. in Laboratory Methods for the Detection of Mutations and Polymorphisms in DNA (ed. Taylor, G.) 151–162 (CRC Press, New York, 1997).
Frommer, M. et al. A genomic sequencing protocol that yields a positive display of 5-methylcytosine residues in individual DNA strands. Proc. Natl. Acad. Sci. USA 89, 1827–1831 (1992).
Clark, S.J., Harrison, J., Paul, C.L. & Frommer, M. High sensitivity mapping of methylated cytosines. Nucleic Acids Res. 22, 2990–2997 (1994).
Rakyan, V.K. et al. DNA methylation profiling of the human major histocompatibility complex: a pilot study for the human epigenome project. PLoS Biol. 2, e405 (2004).
Rauscher, F.J. It is time for a Human Epigenome Project. Cancer Res. 65, 11229 (2005).
Murrell, A., Rakyan, V.K. & Beck, S. From genome to epigenome. Hum. Mol. Genet. 14 (Spec No. 1): R3–R10 (2005).
Jones, P.A. & Martienssen, R. A blueprint for a Human Epigenome Project: the AACR Human Epigenome Workshop. Cancer Res. 65, 11241–11246 (2005).
Callinan, P.A. & Feinberg, A.P. The emerging science of epigenomics. Hum. Mol. Genet. 15 (Spec No. 1): R95–R101 (2006).
Garber, K. Momentum building for human epigenome project. J. Natl. Cancer Inst. 98, 84–86 (2006).
Esteller, M. The necessity of a human epigenome project. Carcinogenesis 27, 1121–1125 (2006).
Singal, R. & Grimes, S.R. Microsoft Word macro for analysis of cytosine methylation by the bisulfite deamination reaction. Biotechniques 30, 116–120 (2001).
Li, L.C. & Dahiya, R. MethPrimer: designing primers for methylation PCRs. Bioinformatics 18, 1427–1431 (2002).
Tusnady, G.E., Simon, I., Varadi, A. & Aranyi, T. BiSearch: primer-design and search tool for PCR on bisulfite-treated genomes. Nucleic Acids Res. 33, e9 (2005).
Warnecke, P.M. et al. Identification and resolution of artifacts in bisulfite sequencing. Methods 27, 101–107 (2002).
Grunau, C., Clark, S.J. & Rosenthal, A. Bisulfite genomic sequencing: systematic investigation of critical experimental parameters. Nucleic Acids Res. 29, E65–5 (2001).
Paul, C.L. & Clark, S.J. Cytosine methylation: quantitation by automated genomic sequencing and GENESCAN analysis. Biotechniques 21, 126–133 (1996).
Boyd, V.L. & Zon, G. Bisulfite conversion of genomic DNA for methylation analysis: protocol simplification with higher recovery applicable to limited samples and increased throughput. Anal. Biochem. 326, 278–280 (2004).
Lewin, J., Schmitt, A.O., Adorjan, P., Hildmann, T. & Piepenbrock, C. Quantitative DNA methylation analysis based on four-dye trace data from direct sequencing of PCR amplificates. Bioinformatics 20, 3005–3012 (2004).
Han, W., Cauchi, S., Herman, J.G. & Spivack, S.D. DNA methylation mapping by tag-modified bisulfite genomic sequencing. Anal. Biochem. 355, 50–61 (2006).
Tost, J., Dunker, J. & Gut, I.G. Analysis and quantification of multiple methylation variable positions in CpG islands by Pyrosequencing. Biotechniques 35, 152–156 (2003).
Colella, S., Shen, L., Baggerly, K.A., Issa, J.P. & Krahe, R. Sensitive and quantitative universal Pyrosequencing methylation analysis of CpG sites. Biotechniques 35, 146–150 (2003).
Laird, C.D. et al. Hairpin-bisulfite PCR: assessing epigenetic methylation patterns on complementary strands of individual DNA molecules. Proc. Natl. Acad. Sci. USA 101, 204–209 (2004).
Schatz, P., Dietrich, D. & Schuster, M. Rapid analysis of CpG methylation patterns using RNase T1 cleavage and MALDI-TOF. Nucleic Acids Res. 32, e167 (2004).
Ehrich, M. et al. Quantitative high-throughput analysis of DNA methylation patterns by base-specific cleavage and mass spectrometry. Proc. Natl. Acad. Sci. USA 102, 15785–15790 (2005).
Worm, J., Aggerholm, A. & Guldberg, P. In-tube DNA methylation profiling by fluorescence melting curve analysis. Clin. Chem. 47, 1183–1189 (2001).
Guldberg, P., Worm, J. & Gronbaek, K. Profiling DNA methylation by melting analysis. Methods 27, 121–127 (2002).
Yang, I. et al. Rapid quantification of DNA methylation through dNMP analysis following bisulfite-PCR. Nucleic Acids Res. 34, e61 (2006).
Yamamoto, T. et al. Methylation assay by nucleotide incorporation: a quantitative assay for regional CpG methylation density. Biotechniques 36, 846–50 852, 854 (2004).
Maekawa, M. et al. DNA methylation analysis using bisulfite treatment and PCR-single-strand conformation polymorphism in colorectal cancer showing microsatellite instability. Biochem. Biophys. Res. Commun. 262, 671–676 (1999).
Bianco, T., Hussey, D. & Dobrovic, A. Methylation-sensitive, single-strand conformation analysis (MS-SSCA): A rapid method to screen for and analyze methylation. Hum. Mutat. 14, 289–293 (1999).
Baumer, A., Wiedemann, U., Hergersberg, M. & Schinzel, A. A novel MSP/DHPLC method for the investigation of the methylation status of imprinted genes enables the molecular detection of low cell mosaicisms. Hum. Mutat. 17, 423–430 (2001).
Sadri, R. & Hornsby, P.J. Rapid analysis of DNA methylation using new restriction enzyme sites created by bisulfite modification. Nucleic Acids Res. 24, 5058–5059 (1996).
Xiong, Z. & Laird, P.W. COBRA: a sensitive and quantitative DNA methylation assay. Nucleic Acids Res. 25, 2532–2534 (1997).
Eads, C.A. & Laird, P.W. Combined bisulfite restriction analysis (COBRA). Methods Mol. Biol. 200, 71–85 (2002).
Gonzalgo, M.L. & Jones, P.A. Quantitative methylation analysis using methylation-sensitive single-nucleotide primer extension (Ms-SNuPE). Methods 27, 128–133 (2002).
Kaminsky, Z.A., Assadzadeh, A., Flanagan, J. & Petronis, A. Single nucleotide extension technology for quantitative site-specific evaluation of metC/C in GC-rich regions. Nucleic Acids Res. 33, e95 (2005).
El-Maarri, O., Herbiniaux, U., Walter, J. & Oldenburg, J. A rapid, quantitative, non-radioactive bisulfite-SNuPE- IP RP HPLC assay for methylation analysis at specific CpG sites. Nucleic Acids Res. 30, e25 (2002).
Gitan, R.S., Shi, H., Chen, C.M., Yan, P.S. & Huang, T.H. Methylation-specific oligonucleotide microarray: a new potential for high-throughput methylation analysis. Genome Res. 12, 158–164 (2002).
Adorjan, P. et al. Tumour class prediction and discovery by microarray-based DNA methylation analysis. Nucleic Acids Res. 30, e21 (2002).
Thomassin, H., Kress, C. & Grange, T. MethylQuant: a sensitive method for quantifying methylation of specific cytosines within the genome. Nucleic Acids Res. 32, e168 (2004).
Bibikova, M. et al. High-throughput DNA methylation profiling using universal bead arrays. Genome Res. 16, 383–393 (2006).
Herman, J.G., Graff, J.R., Myohanen, S., Nelkin, B.D. & Baylin, S.B. Methylation-specific PCR: a novel PCR assay for methylation status of CpG islands. Proc. Natl. Acad. Sci. USA 93, 9821–9826 (1996).
Eads, C.A. et al. MethyLight: a high-throughput assay to measure DNA methylation. Nucleic Acids Res. 28, E32 (2000).
Rand, K., Qu, W., Ho, T., Clark, S.J. & Molloy, P. Conversion-specific detection of DNA methylation using real-time polymerase chain reaction (ConLight-MSP) to avoid false positives. Methods 27, 114–120 (2002).
Zeschnigk, M. et al. A novel real-time PCR assay for quantitative analysis of methylated alleles (QAMA): analysis of the retinoblastoma locus. Nucleic Acids Res. 32, e125 (2004).
Cottrell, S.E. et al. A real-time PCR assay for DNA-methylation using methylation-specific blockers. Nucleic Acids Res. 32, e10 (2004).
Rand, K.N. et al. Headloop suppression PCR and its application to selective amplification of methylated DNA sequences. Nucleic Acids Res. 33, e127 (2005).
Shaw, R.J., Akufo-Tetteh, E.K., Risk, J.M., Field, J.K. & Liloglou, T. Methylation enrichment pyrosequencing: combining the specificity of MSP with validation by pyrosequencing. Nucleic Acids Res. 34, e78 (2006).
Rand, K., Mitchell, S., Clark, S. & Molloy, P. Bisulphite differential denaturation PCR for analysis of DNA methylation. Epigenetics 1, 94–100 (2006).
Warnecke, P.M. et al. Detection and measurement of PCR bias in quantitative methylation analysis of bisulphite-treated DNA. Nucleic Acids Res. 25, 4422–4426 (1997).
Acknowledgements
The authors thank Rebecca Hinshelwood for help with the figures. The work was partly supported by grants from NH and MRC to S.J.C. (ID9293811, ID293810, ID325622).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Clark, S., Statham, A., Stirzaker, C. et al. DNA methylation: Bisulphite modification and analysis. Nat Protoc 1, 2353–2364 (2006). https://doi.org/10.1038/nprot.2006.324
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2006.324
This article is cited by
-
Profiling the epigenetic landscape of the antigen receptor repertoire: the missing epi-immunogenomics data
Nature Methods (2023)
-
Accurate, non-destructive, and high-throughput age estimation for Golden perch (Macquaria ambigua spp.) using DNA methylation
Scientific Reports (2023)
-
Bisulfite profiling of the MGMT promoter and comparison with routine testing in glioblastoma diagnostics
Clinical Epigenetics (2022)
-
Methylation-mediated silencing of miR-125a-5p facilitates breast cancer progression by inducing autophagy
Molecular Biology Reports (2022)
-
Downregulation of epithelial sodium channel (ENaC) activity in cystic fibrosis cells by epigenetic targeting
Cellular and Molecular Life Sciences (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.