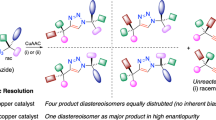
Abstract
The current theory of the titled phenomenon is apparently based on an inconsistent use of concentration units, as employed in the derivation of the fundamental equations. Thus, manifestly, whilst the relation between extent of conversion and e.e. is derived with mole fractions, the succeeding kinetic equations employ units of molarity. This invalidates the derivation in the general case. Fortuitously, however, it is applicable in the majority of simple cases, wherein the total number of moles involved in the reaction remains constant. Herein is presented a rigorous approach which is generally valid.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chandrasekhar, S. Kinetic resolution of racemates under chiral catalysis: connecting the dots . Nat Prec (2012). https://doi.org/10.1038/npre.2012.7127.1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/npre.2012.7127.1