Abstract
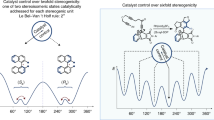
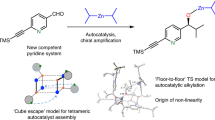
It is argued that the titled non-linear effects (NLE) may arise whenever the order of the reaction in the chiral catalyst in greater than 1. In a fundamental departure from previous approaches, this is mathematically elaborated for the second order case. (NLE may also be observed if the chiral catalyst forms non-reacting dimers in a competing equilibrium; practically, however, this implies the in situ resolution of the catalyst.) The amplification of enantiomeric excess by NLE implies a relative (although modest) reduction in the entropy of mixing. The consequent increase in free energy apparently indicates a non-equilibrium process. It is suggested, based on arguments involving the chemical potential, that kinetically-controlled reactions lead to a state of “quasi-equilibrium”: in this, although overall equilibrium is attained, the product-spread is far from equilibrium. Thus, both the linear and NLE cases of chiral catalysis represent departures from equilibrium (which requires that the product e.e. = 0). Interesting similarities exist with models of non-equilibrium systems, the NLE cases apparently being analogs of open systems just after the bifurcation point has been crossed.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chandrasekhar, S. Non-linear Effects in Asymmetric Catalysis: Whys and Wherefores. Nat Prec (2012). https://doi.org/10.1038/npre.2012.6947.1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/npre.2012.6947.1