Abstract
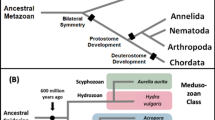
11[beta]-hydroxysteroid dehydrogenase type-2 (11[beta]-HSD2) regulates the local concentration of cortisol that can activate the glucocorticoid receptor and mineralocorticoid receptor, as well as the concentration of 11-keto-testosterone, the active androgen in fish. Similarly, 17[beta]-HSD2 regulates the levels of testosterone and estradiol that activate the androgen receptor and estrogen receptor, respectively. Interestingly, although human 11[beta]-HSD2 and 17[beta]-HSD2 act at different positions on different steroids, these enzymes are paralogs. Despite the physiological importance of 11[beta]-HSD2 and 17[beta]-HSD2, details of their origins and divergence from a common ancestor are not known. An opportunity to understand their evolution is presented by the recent sequencing of genomes from sea urchin, a basal deuterostome, and amphioxus, a basal chordate, and the availability of substantial sequence for acorn worm and elephant shark, which together provide a more complete dataset for analysis of the origins of 11[beta]-HSD2 and 17[beta]-HSD2. BLAST searches find an ancestral sequence of 17[beta]-HSD2 in sea urchin, acorn worm and amphioxus, while an ancestral sequence of 11[beta]-HSD2 first appears in sharks. Sequence analyses indicate that 17[beta]-HSD2 in sea urchin may have a non-enzymatic activity. Evolutionary analyses indicate that if acorn worm 17[beta]-HSD2 is catalytically active, then it metabolizes novel substrate(s).
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Baker, M. 11[beta]-hydroxysteroid dehydrogenase-type 2 evolved from an ancestral 17[beta]-hydroxysteroid dehydrogenase-type 2. Nat Prec (2010). https://doi.org/10.1038/npre.2010.4649.1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/npre.2010.4649.1