Abstract
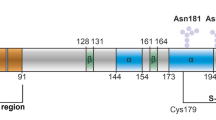
In the more than 20 years since its discovery, both the phylogenetic origin and cellular function of the prion protein (PrP) have remained enigmatic. Insights into the function of PrP may be obtained through a characterization of its molecular neighborhood. Quantitative interactome data revealed the spatial proximity of a subset of metal ion transporters of the ZIP family to mammalian prion proteins. A subsequent bioinformatic analysis revealed the presence of a prion-like protein sequence within the N-terminal, extracellular domain of a phylogenetic branch of ZIPs. Additional structural threading and ortholog sequence alignment analyses consolidated the conclusion that the prion protein gene family is phylogenetically derived from a ZIP-like ancestor molecule. Our data explain structural and functional features found within mammalian prion proteins as elements of an ancient involvement in the transmembrane transport of divalent cations. The connection to ZIP proteins is expected to open new avenues to elucidate the biology of the prion protein in health and disease.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schmitt-Ulms, G., Ehsani, S., Watts, J. et al. Evolutionary descent of prion genes from a ZIP metal ion transport ancestor. Nat Prec (2009). https://doi.org/10.1038/npre.2009.2867.1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/npre.2009.2867.1