Abstract
(+/−)-3,4-Methylenedioxymethamphetamine (MDMA, ‘Ecstasy’) abusers have persistent neuropsychiatric deficits including memory impairments after the cessation of abuse. On the other hand, cannabinoid CB1 receptors have been implicated in learning/memory, and are highly expressed in the hippocampus, a region of the brain believed to have an important function in certain forms of learning and memory. In this study, we clarified the mechanism underlying the cognitive impairment that develops during MDMA withdrawal from the standpoint of the cannabinoid CB1 receptors. Mice were administered MDMA (10 mg/kg, i.p.) once a day for 7 days. On the 7th day of withdrawal, a novel object recognition task was performed and the amount of cannabinoid CB1 receptor protein was measured with western blotting. Recognition performance was impaired on the 7th day of withdrawal. This impairment was blocked by AM251, a cannabinoid CB1 receptor antagonist, administered 30 min before the training trial or co-administered with MDMA. At this time, the level of cannabinoid CB1 receptor protein increased significantly in the hippocampus but not the prefrontal cortex or striatum. This increase of CB1 receptor protein in the hippocampus was also blocked by the co-administration of AM251. Furthermore, CB1 receptor knockout mice showed no impairment of recognition performance on the withdrawal from MDMA. The impairment of recognition memory during withdrawal from MDMA may result from the activation of cannabinoid CB1 receptors in the hippocampus.
Similar content being viewed by others
INTRODUCTION
(+/−)-3,4-Methylenedioxymethamphetamine (MDMA) is widely abused throughout the world. MDMA abusers have neuropsychiatric deficits including memory impairments (McCardle et al, 2004; Wareing et al, 2007). Recent studies suggest that this neuropsychiatric deficit persists after the cessation of abuse (Ward et al, 2006). In addition, cocaine, amphetamine, or opiate abusers also show cognitive impairment during long-term drug abstinence (Ersche et al, 2006; Pace-Schott et al, 2008).
Cannabis usage causes deficits in attention, executive functioning, and short-term memory (O'Leary et al, 2002; Medina et al, 2007). We showed earlier that repeated treatment with Δ9-tetrahydrocannabinol (Δ9-THC), the main psychoactive ingredient of marijuana (cannabis), impaired delayed matching-to-sample performance even 24 h after the administration (Miyamoto et al, 1995). An other study has also found that Δ9-THC impairs spatial memory (Lichtman and Martin, 1996). These reports suggest that the activation of the brain cannabinoid system impairs working memory. Furthermore, it has been revealed that the cannabinoid system is involved in drug dependence (Yamamoto and Takada, 2000; Yamamoto et al, 2004). A cannabinoid CB1 receptor antagonist, SR141716A, attenuated the reinstatement of methamphetamine-seeking behavior (Anggadiredja et al, 2004; Hiranita et al, 2008). Moreover, cannabinoid CB1 receptor knockout mice failed to establish cocaine, morphine, and ethanol self-administration (Cossu et al, 2001; Soria et al, 2005; Thanos et al, 2005). In a biochemical study, Gonzalez et al (2002) reported that chronic exposure to morphine increased levels of cannabinoid CB1 receptor mRNA and CB1 receptor binding in the brain. In addition, the hippocampal cannabinoid system seems to be activated during withdrawal from ethanol, because both endogenous cannabinoids and CB1 receptors levels increased (Mitrirattanakul et al, 2007). Despite the close involvement of the cannabinoid system in the reward system, it is still unclear whether the system is involved in cognitive impairment on withdrawal from chronic exposure to drugs of abuse.
Here, we clarified the role of the cannabinoid system in cognitive impairment during withdrawal from MDMA using the novel object recognition task. We also investigated that the effect of MDMA on the level of cannabinoid CB1 receptor protein correlated with a behavioral test.
MATERIALS AND METHODS
Animals
Male CD1 (wild-type) mice (Charles River, Yokohama, Japan) and cannabinoid CB1 receptor knockout (CB1 KO) mice on a CD1 background, provided by Dr Catherine Ledent (Institut de Recherches en Biologie Humaine et Moléculaire, Université Libre de Bruxelles), and weighing 30–35 g, were used in the present experiment. There were 117 wild-type mice and 29 CB1 KO mice used in all experiment. We conducted each experiment with a small control group of 2–3 mice each and these control group data were combined together in the end to represent the control values. The animals were housed in plastic cages and kept in a regulated environment (23±1°C), with a 12/12 h light–dark cycle (lights on at 7:00 am). Food and water were available ad libitum. Procedures were conducted in accordance with the Guide for the Care and Use of Laboratory Animals as adopted and promulgated by the Declaration of Helsinki and Nagasaki International University Publication, enacted in 2006.
Drugs
(+/−)-3,4-Methylenedioxymethamphetamine HCl (MDMA; provided by Dr Tatsunori Iwamura, Matsuyama University) was dissolved in saline. AM251 (Sigma, St Louis, MO) was dissolved in a mixture of DMSO, Tween-80 and saline (1 : 1 : 18, respectively). All drugs were administered intraperitoneally (i.p.), and injected at a volume of 0.1 ml per 10 g of body weight. Saline or MDMA (10 mg/kg) was administered once or once daily for 7 days. AM251 (1.0 or 3.2 mg/kg) was co-administered with MDMA or singly administered 30 min before the training trial on the 7th day of withdrawal after the repeated administration of MDMA.
Behavioral Testing
Object recognition test
The object recognition test was carried out on the 1st or 7th day after the repeated administration of MDMA in separate groups. This test was performed in a Plexiglas open-field box (in cm 70 wide × 70 deep × 40 high) with black vertical walls and a floor. The objects to be discriminated were silver cone-shaped and bulb-shaped. Mice were habituated to the open field for 1 h (habituation trial). The next day, in the training trial, each mouse was placed in the open field and allowed to explore two identical objects for 10 min. The test trial was performed 3 h after the training trial. One familiar object and one novel object were placed in the same location as in the training trial. For the measurement on the 1st day of withdrawal, the habituation trial was conducted just before the last drug injection. The time spent exploring each object and the total amount of time spent exploring both objects were recorded. Exploration of an object was defined as placing the nose or a forepaw at or beyond marks put on the open-field at a distance of 1 cm from each object. A discrimination ratio was calculated as the difference in time spent exploring the novel and familiar object, expressed as a ratio of the total time spent exploring both objects in the test trial. Mice showing a total exploration time of <10 s during the training trial were excluded. The ambulation during the trial was measured with a digital tracking and computerized scoring system (LimeLight, Actimetrics). To determine whether the mice discriminated between novel and familiar objects, the discrimination ratios obtained under each condition were compared with those that would be expected by chance (ie, a ratio of 0.0), using one-sample t tests.
Biochemical Testing
Western blot analysis
Immediately after decapitation, the whole brain was removed from the skull, placed on ice, and the hippocampus, prefrontal cortex, and striatum were removed as described earlier (Yamaguchi et al, 2004). These tissues were immediately homogenized in a lysis buffer (10 mM Tris–HCl, pH 7.5, 5 mM MgCl2, 5 mM EDTA, 250 mM Sucrose, 1 mM Dithiothreitol, 1% Triton X-100, 1% sodium cholate, and protease inhibitor cocktail). All samples were subjected to a BCA assay to adjust the amount of protein loaded before the sample buffer was added. The sample (10 μg) was applied to a 10% polyacrylamide gel (BioRad, Hercules, CA), and the proteins were transferred electrophoretically to nitrocellulose membranes (Bio Rad). The membrane was blocked with TBS-Tween 20 (0.1%) and 5% nonfat dry milk and incubated with the primary antibodies [anti-cannabinoid CB1 (1 : 1000, Calbiochem, US and Canada) overnight at 4°C and anti-β-actin (1 : 2000, Sigma)] 1 h at room temperature. The antibodies were detected using HRP-conjugated anti-rabbit and anti-mouse IgG (GE Healthcare, Tokyo, Japan, 1 : 1000) secondary antibodies. The blots were detected using a chemiluminescence method (ECL system; GE Healthcare).
Data Analysis
Data are expressed as mean±SE. A one-way ANOVA was used to compare means, and Bonferroni–Dunn tests were used for post hoc analysis. p<0.05 was accepted as statistically significant.
RESULTS
Novel Object Recognition Performance During MDMA Withdrawal
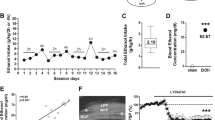
In the training trial, vehicle, single MDMA, and repeated (for 7 days) MDMA-treated mice on the 1st day after the last treatment spent 23.1±1.8, 22.0±5.6, and 21.3±2.0 s exploring objects, respectively. Meanwhile, on the 7th day after treatment, the time spent exploring objects in the vehicle, single MDMA, and repeated MDMA-treated groups was 19.3±2.7, 26.6±5.2, and 21.4±3.1 s, respectively. Hence, the time spent exploring objects on the 1st day of withdrawal in the training trial in single MDMA- and repeated MDMA-treated groups was not significantly different from that of vehicle-treated group (p=0.06 and p=0.55 vs vehicle-treated group, respectively). In addition, on the 7th day of withdrawal, there was no significant difference in the time spent exploring objects among the three groups in the training trial (p=0.28, p=0.94 vs vehicle-treated group, respectively). In the test trial, the vehicle-treated mice spent significantly longer exploring the novel object (21.3±2.3 s) than the familiar object (5.4±1.0 s) (F[1,28] =30.8, p<0.0001 vs exploration time for the familiar object). On the 1st and 7th day after a single administration of MDMA, there was no significant change in the discrimination ratio (p=0.47 and p=0.13 vs control group on the 1st and 7th day, respectively). However, the discrimination ratio significantly decreased on the 1st and 7th days of withdrawal from repeated administration of MDMA (0.597±0.071–0.26±0.106%: F[1,19]=5.3, p<0.05 and 0.633±0.048–0.048±0.049%: F[1,27]=70.2, p<0.001 vs control group on the 1st and 7th days, respectively) (Figure 1). Discrimination ratios were significantly above chance in all groups except for mice on the 7th days of withdrawal from repeated MDMA (p<0.001; control group on the 1st and 7th day, p<0.01; on the 1st and 7th day of withdrawal from single MDMA, p<0.05; on the 1st day of withdrawal from repeated MDMA). In this test trial, ambulation did not differ between the MDMA-treated and vehicle-treated groups (p=0.21 and p=0.61 on the 1st and 7th days, respectively). The decrease in the discrimination ratio on the 7th day of withdrawal was prevented by the co-administration of AM251, a cannabinoid CB1 receptor antagonist, with MDMA in a dose-dependent manner (0.048±0.049 to 0.592±0.067%: F[1,22] = 48.5, p<0.001 vs MDMA group) (Figure 2a). However, ambulation in AM251 co-administered group (4269±269 cm) did not differ from ambulation in vehicle (4578±354 cm) or MDMA (4334±310 cm) groups (p=0.51 and p=0.55 vs vehicle and MDMA alone, respectively). On the other hand, a single administration of AM251 30 min before the training trial on the 7th day of withdrawal from repeated MDMA treatment stopped the reduction in the discrimination ratio in a dose-dependent manner (0.048±0.049 to 0.661±0.074%: F[1,18] =54.7, p<0.001 vs MDMA group) (Figure 2b). A single administration of AM251 on 7th day of withdrawal from repeated MDMA had no effect on ambulation (p=0.19 and p=0.3 vs vehicle and MDMA alone, respectively). Discrimination ratios were significantly above chance in mice co-administered and singly administered AM251 (p<0.001). While, there was no significant difference in the time spent exploring objects and the discrimination ratio between vehicle-treated wild-type and CB1 KO mice in the test trial. However, CB1 KO mice did not exhibit a reduction in the discrimination ratio on both 1st and 7th day of withdrawal from repeated MDMA treatment (Figure 3). Discrimination ratios were significantly above chance in all groups of CB1 KO mice (p<0.001).
Novel object recognition performance in wild-type mice on the 1st or 7th day of withdrawal after single or repeated (daily for 7 day) MDMA treatment (10 mg/kg, i.p.). Each graph shows the discrimination ratio in the test trial. Data represent the mean±SEM (n=5–15). *p<0.05, ***p<0.001 vs vehicle-treated mice. Vehicle includes results for mice administered saline once or repeatedly for 7 days.
Effect of a cannabinoid CB1 receptor antagonist, AM251, on cognitive impairment on the 7th day of MDMA withdrawal (10 mg/kg, i.p., daily for 7 days) in wild-type mice. (a) AM251 (1.0 or 3.2 mg/kg, i.p.) was co-administered with MDMA. Data represent the mean±SEM (n=8–15). ***p<0.001 vs vehicle-treated mice; ###p<0.001 vs MDMA (10 mg/kg)-treated mice. Vehicle means results for mice administered saline. (b) AM251 (1.0 or 3.2 mg/kg, i.p.) was administered 30 min before the training trial. Data represent the mean±SEM (n=5–15). ***p<0.001 vs vehicle-treated mice; ###p<0.001 vs MDMA-treated mice. Vehicle means results for mice administered saline.
Comparison of novel object recognition performance in wild-type and CB1 receptor knockout mice on the 1st and 7th day of MDMA withdrawal (10 mg/kg, i.p., daily for 7 days). Each graph shows the discrimination ratio in the test trial. Data represent the mean±SEM (n=8–15). *p<0.05, ***p<0.001 vs vehicle-treated mice. Open and closed bars indicate wild-type and CB1 receptor knockout mice, respectively. Vehicle means results for mice administered saline.
Alteration of the Level of Cannabinoid CB1 Receptor Protein During Withdrawal from Repeated MDMA Treatment
The level of cannabinoid CB1 receptor protein did not change on the 1st day of withdrawal from repeated administration of MDMA in the hippocampus. On the 7th day of withdrawal, the level of CB1 receptor protein in the hippocampus was significantly increased (0.48±0.06–0.96±0.07, F[1,13]=28.1, p<0.001 vs vehicle-treated group) (Figure 4a). This increase was prevented by co-administration of AM251 with MDMA (0.96±0.07–0.60±0.09, F[1,14] = 9.6, p<0.01 vs MDMA-treated group) (Figure 4b). There was no significant change in the prefrontal cortex or striatum on both 1st and 7th day of withdrawal (Figure 4a).
Effect of repeated administration of MDMA on the levels of CB1 receptor protein in the brain in wild-type mice on the 1st and 7th day of MDMA withdrawal (10 mg/kg, i.p., daily for 7 days). (a) The levels of CB1 receptor protein in the hippocampus, prefrontal cortex, and striatum were measured. Data represent the mean±SEM (n=8–11). (b) Effect of repeated administration of AM251 (3.2 mg/kg) with MDMA on MDMA-induced up-regulation of CB1 receptor protein expression in the hippocampus. Open and closed bars indicate vehicle- and repeated MDMA-treated group. Data represent the mean±SEM (n=5–11). ***p<0.001 vs vehicle-treated mice; ##p<0.01 vs MDMA-treated mice. Vehicle means results for mice administered saline.
DISCUSSION
Object recognition memory in mice was impaired on withdrawal from repeated MDMA. This impairment was prevented by the co-administration of AM251, a cannabinoid CB1 receptor antagonist, with MDMA in wild-type mice. In addition, a single treatment of AM251 on the 7th day of MDMA withdrawal ameliorated this recognition memory impairment. In CB1 KO mice, recognition memory was not impaired on withdrawal from MDMA. These results suggest that the activation of cannabinoid CB1 receptors is involved in the appearance of cognitive impairment on withdrawal from MDMA. In rats, it was also reported that object recognition memory is also impaired on withdrawal from MDMA similar to our present findings in mice (Morley et al, 2001; McGregor et al, 2003; Piper and Meyer, 2004). However, this is the first report to show the involvement of cannabinoid CB1 receptors in the appearance of cognitive impairment on withdrawal from abusive drugs. Recently, Touriño et al (2008) indicated that CB1 KO mice did not show the performance of MDMA self-administration. This finding suggests that the activation of CB1 receptors is involved in the MDMA reinforcing effect.
The level of cannabinoid CB1 receptor protein in the hippocampus was significantly increased on the 7th day of withdrawal but not on the 1st day while recognition memory was impaired on both the 1st day and 7th day of MDMA withdrawal. In this regard, cognitive impairment on the 1st day of MDMA withdrawal may be due to the increase in hippocampal endocannabinoid. Mitrirattanakul et al (2007) reported that the amount of endocannabinoid 2-AG in the hippocampus significantly increased in the early phase of ethanol withdrawal without any increase in CB1 receptor expression. Our findings may be supported by this result. On the other hand, a single treatment with CB1 receptor antagonist for MDMA withdrawal significantly ameliorated the recognition memory impairment on 7th day of MDMA withdrawal. Accordingly, CB1 receptors may be activated with the increase in their expression at this time.
In addition, the activation of the brain cannabinoid system causes deficits in attention, executive functioning, and short-term memory (Lichtman and Martin, 1996; O'Leary et al, 2002; Medina et al, 2007). It is also demonstrated that object recognition memory was impaired by both endogenous cannabinoid Δ9-THC and synthetic CB1 receptor agonist WIN 55,212-2 (Schneider and Koch, 2002; Quinn et al, 2008). Our findings may be supported by the literatures above.
The important function of the hippocampus in cognitive functions including recognition memory is well established by earlier findings. Hippocampal damage from ibotenic acid disrupted recognition memory in the novel object recognition task and the visual paired comparison task (Clark et al, 2000; Broadbent et al, 2004). Furthermore, an intra-hippocampal WIN 55,212-2, also impaired performance of the novel object recognition task (Kosiorek et al, 2003; Suenaga and Ichitani, 2008).
Hampson and Deadwyler (2000) found that Δ9-THC and WIN 55,212-2 act selectively to disrupt the encoding of events in the hippocampus during memory processing, on measuring the combined simultaneous multineuron firing rate. Recently, it was also suggested that the cannabinoids Δ9-THC and a cannabinoid CB1 receptor agonist CP55940 disrupted the temporal coordination of hippocampal neurons, and that this effect may correlate with memory deficits in individuals (Robbe et al, 2006).
Enocannabinoids are known to participate in forms of synaptic plasticity (Mackie, 2008). This phenomenon associated with endocannabinoids may help explain the mechanism by which cannabinoids impair memory. Long-term potentiation (LTP) is a form of synaptic plasticity thought to have functional roles in learning and memory processes. The importance of the hippocampal LTP in learning and memory has also been shown that hippocampal LTP is facilitated after following exposure to a novel environment but not by exposure to a familiar environment (Li et al, 2003). In addition, it has been shown that the cannabinoid system affects the hippocampal LTP by chronic Δ9-THC blocking hippocampal LTP via CB1 receptors after withdrawal (Hoffman et al, 2007). It has been shown that cannabinoids inhibit neurotransmitter release via presynaptic cannabinoid CB1 receptors (Schlicker and Kathmann, 2001). Additionally, LTP disruption in the hippocampus by WIN 55,212-2 may be associated with an inhibition of hippocampal glutamatergic transmission (Misner and Sullivan, 1999). Accordingly, the appearance of cognitive impairment during MDMA withdrawal may result in dysfunction of hippocampal LTP via inhibition of glutamate release induced by an activation of CB1 receptors. These reports strongly support our present finding that the activation of the hippocampal cannabinoid system disrupts recognition memory during MDMA withdrawal.
In conclusion, our results suggest the impairment of recognition memory during withdrawal from repeated administration of MDMA to be due to the activation of cannabinoid CB1 receptors in the hippocampus. Moreover, these findings suggest that cannabinoid CB1 receptor antagonists would have a therapeutic effect on cognitive dysfunction in MDMA abusers.
References
Anggadiredja K, Nakamichi M, Hiranita T, Tanaka H, Shoyama Y, Watanabe S et al (2004). Endocannabinoid system modulates relapse to methamphetamine seeking: possible mediation by the arachidonic acid cascade. Neuropsychopharmacology 29: 1470–1478.
Broadbent NJ, Squire LR, Clark RE (2004). Spatial memory, recognition memory, and the hippocampus. Proc Natl Acad Sci USA 101: 14515–14520.
Clark RE, Zola SM, Squire LR (2000). Impaired recognition memory in rats after damage to the hippocampus. J Neurosci 20: 8853–8860.
Cossu G, Ledent C, Fattore L, Imperato A, Bohme GA, Parmentier M et al (2001). Cannabinoid CB1 receptor knockout mice fail to self-administer morphine but not other drugs of abuse. Behav Brain Res 118: 61–65.
Ersche KD, Clark L, London M, Robbins TW, Sahakian BJ (2006). Profile of executive and memory function associated with amphetamine and opiate dependence. Neuropsychopharmacology 31: 1036–1047.
Gonzalez S, Fernandez-Ruiz J, Sparpaglione V, Parolaro D, Ramos JA (2002). Chronic exposure to morphine, cocaine or ethanol in rats produced different effects in brain cannabinoid CB(1) receptor binding and mRNA levels. Drug Alcohol Depend 66: 77–84.
Hampson RE, Deadwyler SA (2000). Cannabinoids reveal the necessity of hippocampal neural encoding for short-term memory in rats. J Neurosci 20: 8932–8942.
Hiranita T, Nawata Y, Sakimura K, Yamamoto T (2008). Methamphetamine-seeking behavior is due to inhibition of nicotinic cholinergic transmission by activation of cannabinoid CB1 receptors. Neuropharmacology 55: 1300–1306.
Hoffman AF, Oz M, Yang R, Lichtman AH, Lupica CR (2007). Opposing actions of chronic Delta9-tetrahydrocannabinol and cannabinoid antagonists on hippocampal long-term potentiation. Learn Mem 14: 63–74.
Kosiorek P, Hryniewicz A, Bialuk I, Zawadzka A, Winnicka MM (2003). Cannabinoids alter recognition memory in rats. Pol J Pharmacol 55: 903–910.
Li S, Cullen WK, Anwyl R, Rowan MJ (2003). Dopamine-dependent facilitation of LTP induction in hippocampal CA1 by exposure to spatial novelty. Nat Neurosci 6: 526–531.
Lichtman AH, Martin BR (1996). Delta 9-tetrahydrocannabinol impairs spatial memory through a cannabinoid receptor mechanism. Psychopharmacology 126: 125–131.
Mackie K (2008). Signaling via CNS cannabinoid receptors. Mol Cell Endocrinol 286: S60–S65.
McCardle K, Luebbers S, Carter JD, Croft RJ, Stough C (2004). Chronic MDMA (ecstasy) use, cognition and mood. Psychopharmacology 173: 434–439.
McGregor IS, Gurtman CG, Morley KC, Clemens KJ, Blokland A, Li KM et al (2003). Increased anxiety and ‘depressive’ symptoms months after MDMA (‘ecstasy’) in rats: drug-induced hyperthermia does not predict long-term outcomes. Psychopharmacology 168: 465–474.
Medina KL, Hanson KL, Schweinsburg AD, Cohen-Zion M, Nagel BJ, Tapert SF (2007). Neuropsychological functioning in adolescent marijuana users: subtle deficits detectable after a month of abstinence. J Int Neuropsychol Soc 13: 807–820.
Misner DL, Sullivan JM (1999). Mechanism of cannabinoid effects on long-term potentiation and depression in hippocampal CA1 neurons. J Neurosci 19: 6795–6805.
Mitrirattanakul S, Lopez-Valdes HE, Liang J, Matsuka Y, Mackie K, Faull KF et al (2007). Bidirectional alterations of hippocampal cannabinoid 1 receptors and their endogenous ligands in a rat model of alcohol withdrawal and dependence. Alcohol Clin Exp Res 31: 855–867.
Miyamoto A, Yamamoto T, Watanabe S (1995). Effect of repeated administration of delta 9-tetrahydrocannabinol on delayed matching-to-sample performance in rats. Neurosci Lett 201: 139–142.
Morley KC, Gallate JE, Hunt GE, Mallet PE, McGregor IS (2001). Increased anxiety and impaired memory in rats 3 months after administration of 3,4-methylenedioxymethamphetamine (‘ecstasy’). Eur J Pharmacol 433: 91–99.
O'Leary DS, Block RI, Koeppel JA, Flaum M, Schultz SK, Andreasen NC et al (2002). Effects of smoking marijuana on brain perfusion and cognition. Neuropsychopharmacology 26: 802–816.
Pace-Schott EF, Morgan PT, Malison RT, Hart CL, Edgar C, Walker M et al (2008). Cocaine users differ from normals on cognitive tasks which show poorer performance during drug abstinence. Am J Drug Alcohol Abuse 34: 109–121.
Piper BJ, Meyer JS (2004). Memory deficit and reduced anxiety in young adult rats given repeated intermittent MDMA treatment during the periadolescent period. Pharmacol Biochem Behav 79: 723–731.
Quinn HR, Matsumoto I, Callaghan PD, Long LE, Arnold JC, Gunasekaran N et al (2008). Adolescent rats find repeated Delta(9)-THC less aversive than adult rats but display greater residual cognitive deficits and changes in hippocampal protein expression following exposure. Neuropsychopharmacology 33: 1113–1126.
Robbe D, Montgomery SM, Thome A, Rueda-Orozco PE, McNaughton BL, Buzsaki G (2006). Cannabinoids reveal importance of spike timing coordination in hippocampal function. Nat Neurosci 9: 1526–1533.
Schlicker E, Kathmann M (2001). Modulation of transmitter release via presynaptic cannabinoid receptors. Trends Pharmacol Sci 22: 565–572.
Schneider M, Koch M (2002). The cannabinoid agonist WIN 55,212-2 reduces sensorimotor gating and recognition memory in rats. Behav Pharmacol 13: 29–37.
Soria G, Mendizabal V, Tourino C, Robledo P, Ledent C, Parmentier M et al (2005). Lack of CB1 cannabinoid receptor impairs cocaine self-administration. Neuropsychopharmacology 30: 1670–1680.
Suenaga T, Ichitani Y (2008). Effects of hippocampal administration of a cannabinoid receptor agonist WIN 55,212-2 on spontaneous object and place recognition in rats. Behav Brain Res 190: 248–252.
Thanos PK, Dimitrakakis ES, Rice O, Gifford A, Volkow ND (2005). Ethanol self-administration and ethanol conditioned place preference are reduced in mice lacking cannabinoid CB1 receptors. Behav Brain Res 164: 206–213.
Touriño C, Ledent C, Maldonado R, Valverde O (2008). CB1 cannabinoid receptor modulates 3,4-methylenedioxymethamphetamine acute responses and reinforcement. Biol Psychiatry 63: 1030–1038.
Ward J, Hall K, Haslam C (2006). Patterns of memory dysfunction in current and 2-year abstinent MDMA users. J Clin Exp Neuropsychol 28: 306–324.
Wareing M, Fisk JE, Montgomery C, Murphy PN, Chandler MD (2007). Information processing speed in ecstasy (MDMA) users. Hum Psychopharmacol 22: 81–88.
Yamaguchi T, Kubota T, Watanabe S, Yamamoto T (2004). Activation of brain prostanoid EP3 receptors via arachidonic acid cascade during behavioral suppression induced by Delta8-tetrahydrocannabinol. J Neurochem 88: 148–154.
Yamamoto T, Takada K (2000). Role of cannabinoid receptor in the brain as it relates to drug reward. Jpn J Pharmacol 84: 229–236.
Yamamoto T, Anggadiredja K, Hiranita T (2004). New perspectives in the studies on endocannabinoid and cannabis: a role for the endocannabinoid-arachidonic acid pathway in drug reward and long-lasting relapse to drug taking. J Pharmacol Sci 96: 382–388.
Acknowledgements
We thank Dr Catherine Ledent (Université Libre de Bruxelles) for donating CB1 receptor knockout mice and Dr Tatsunori Iwamura (Matsuyama University) for providing MDMA. This study was supported by a grant from the Ministry of Health, Labor, and Welfare of Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
DISCLOSURE
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Nawata, Y., Hiranita, T. & Yamamoto, T. A Cannabinoid CB1 Receptor Antagonist Ameliorates Impairment of Recognition Memory on Withdrawal from MDMA (Ecstasy). Neuropsychopharmacol 35, 515–520 (2010). https://doi.org/10.1038/npp.2009.158
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/npp.2009.158
Keywords
This article is cited by
-
Gene expression analysis indicates reduced memory and cognitive functions in the hippocampus and increase in synaptic reorganization in the frontal cortex 3 weeks after MDMA administration in Dark Agouti rats
BMC Genomics (2018)
-
Peripheral endocannabinoid concentrations are not associated with verbal memory impairment during MDMA intoxication
Psychopharmacology (2018)
-
RETRACTED ARTICLE: Gene expression analysis indicates CB1 receptor upregulation in the hippocampus and neurotoxic effects in the frontal cortex 3 weeks after single-dose MDMA administration in Dark Agouti rats
BMC Genomics (2013)