Abstract
Positron emission tomography (PET) and the high affinity D2/3 radiotracer [18F]fallypride allow the assessment of D2/3 receptor occupancy of antipsychotic drugs in striatal and extrastriatal brain regions. We measured regional occupancy attained across a range of clinical dosing by the partial D2 agonist aripiprazole using these methods. Twenty-eight PET scans were acquired on the ECAT EXACT HR+ camera in 19 patients with schizophrenia or schizoaffective disorder. Daily aripiprazole doses ranged from 2 to 40 mg, with a minimum of 10 days on steady dose. Mean regional occupancies, a model-independent estimate of aripiprazole effect on pituitary binding, and PANSS ratings changes were evaluated. Occupancy levels were high across regions of interest, ranging from 71.6±5.5% at 2 mg/day to 96.8±5.3% at 40 mg/day. Occupancy levels were higher in extrastriatal than striatal regions. Pituitary measures of aripiprazole effect correlated with doses and were unrelated to prolactin levels, which remained within the normal range under medication. PANSS positive (but not negative) symptom improvement correlated with striatal but not extrastriatal occupancies. These data show, for the first time, D2 occupancy by aripiprazole in treated patients with schizophrenia in extrastriatal as well as striatal regions, with high occupancy for all doses. We discuss possible explanations for higher extrastriatal than striatal occupancy. Correlations of ratings of clinical improvement with regional occupancy suggest that aripiprazole, as do other antipsychotics, benefits positive symptoms of schizophrenia most directly through its modulation of striatal rather than cortical or other extrastriatal dopamine activity.
Similar content being viewed by others
INTRODUCTION
Neuroreceptor imaging has been widely used in the last two decades to assess D2 receptor occupancy achieved by antipsychotic drugs (Kapur et al, 1999; Nyberg et al, 1998) and has contributed some fundamental observations to our understanding of the mechanisms underlying their effects. These studies have established the threshold of occupancy of striatal D2 receptors (about 80%) above which extrapyramidal side effects (EPS) are likely to occur (Farde et al, 1992) and have suggested that 50–60% occupancy was required to observe a rapid clinical response (Kapur et al, 2000; Nordstrom et al, 1993).
More recently, the development of high affinity D2/3 radiotracers allowed assessment of occupancy by antipsychotics in extrastriatal areas where receptors are present in lower density. Pilowsky et al (1997) initially reported lower occupancy of striatal D2 receptors compared to temporal cortex D2 receptors with clozapine, using the high affinity SPECT ligand [123I]epidepride. In contrast, typical antipsychotics were reported to achieve similar occupancy in striatal and extrastriatal areas, as measured with [11C]FLB 457 (Farde et al, 1997) or [123I]epidepride (Bigliani et al, 1999). Occupancy studies performed with [76Br]FLB 457 reported higher occupancies in cortex compared to striatum for a number of antipsychotic drugs, including typical antipsychotics (Xiberas et al, 2001). However, these very high affinity ligands do not allow accurate determination of D2 receptor availability in the striatum due to lack of equilibrium within the timeframe of the experiment (Olsson and Farde, 2001). A study combining [11C]FLB 457 imaging for extrastriatal D2 receptors and [11C]raclopride imaging for striatal D2 receptors suggested similar occupancy of D2 receptors in both regions for both typical and atypical antipsychotic drugs (Talvik et al, 2001). Conversely, [18F]fallypride enables accurate determination of D2 receptor availability in both striatal and extrastriatal areas (Kessler et al, 2000). Occupancy studies using [18F]fallypride reported that clozapine and quetiapine, but not olanzapine or haloperidol, achieved higher D2 receptor occupancy in temporal compared to striatal regions (Grunder et al, 2006; Kessler et al, 2005, 2006). Thus, there is evidence that many antipsychotics achieve higher occupancies in temporal cortex than striatum, although this phenomenon has not been universally observed. Factors underlying this difference remain to be elucidated. Furthermore, D2 receptor occupancy levels in striatum were observed to be more predictive of therapeutic response than in temporal cortex (Agid et al, 2007). Thus, the observation that, in a restricted dose range, D2 receptor occupancy by antipsychotic drugs is higher in temporal cortex than in striatum does not necessarily imply that the temporal cortex is the therapeutic site of action of these agents. In addition, the most robust preclinical evidence regarding the site of therapeutic effect of antipsychotic drugs points toward the ventral striatum (Deutch, 1992; Robertson et al, 1994), while the imaging studies reviewed above contrasted striatal versus mesotemporal D2 receptor binding.
Aripiprazole is a new antipsychotic used for treatment of schizophrenia with a distinct pharmacological profile due to its partial D2 agonism (Kessler, 2007). Two recent studies have assessed the occupancy of striatal D2 receptors by various doses of the drug after at least 2 weeks administration, one in healthy subjects (Yokoi et al, 2002) and the other in patients with schizophrenia (Mamo et al, 2007). The radioligand used for D2 receptor assessment in those studies was [11C]raclopride, which cannot provide quantitative information about D2 receptor binding outside the striatum. Yokoi et al (2002) reported striatal occupancy levels ranging from 60 to 95% without notable EPS, contrasting with the usual presence of EPS at occupancy levels exceeding 80% for D2 antagonists (Farde et al, 1992; Farde et al, 1988). Similarly, Mamo et al (2007) found striatal occupancy levels ranging from 85 to 93% with minimal occurrence of EPS only at the highest occupancy levels. Another characteristic of this drug is the absence of prolactin elevation seen commonly with antipsychotic treatment, consistent with partial agonism of aripiprazole at pituitary D2 receptors. Indeed, a recent study has found adjunctive treatment with aripiprazole to be efficacious in antipsychotic-induced hyperprolactinemia (Shim et al, 2007).
It is unclear to what extent aripiprazole, at therapeutic doses, occupies extrastriatal D2 receptors and the role this occupancy may play in the therapeutic effect. We used [18F]fallypride to measure striatal and extrastriatal D2 receptor occupancy as well as the effect on pituitary binding attained across a range of clinically determined dosing with aripiprazole in patients with schizophrenia. We also investigated the relationship between therapeutic response measured with the PANSS (Kay et al, 1987) and regional occupancy measurements to assess the likely site of antipsychotic effect.
METHODS
Subjects
The study was approved by the Institutional Review Boards of the New York State Psychiatric Institute (NYSPI) and Columbia University. Patients were recruited from the Schizophrenia Research Unit, NYSPI or the affiliated outpatient research clinic and provided written informed consent. Capacity to provide informed consent was evaluated by a psychiatrist not associated with the study. Patients were medically healthy and met inclusion criteria for schizophrenia or schizoaffective disorder, but no other DSM-IV (2000) Axis I diagnosis and no substance abuse by history confirmed with negative urine drug screens, and were free of any psychotropic medication for at least 21 days before the baseline scans and any psychotropic medication other than aripiprazole for at least 21 days before the occupancy scans (with the exception of lorazepam, which was allowed at a maximal dose of 3 mg/day up to 24 h before the study). There were no occurrences of relapse during the medication-free period.
Nineteen patients (see Table 1 for clinical and demographic characteristics) underwent a total of 28 [18F]fallypride scans. Patients’ participation in baseline and occupancy scans is shown schematically in Table 2. Nine patients (group A; Table 2) underwent two scans, one in the unmedicated condition (“baseline” scan) and one on the study medication aripiprazole (“occupancy” scan); two underwent only the baseline scan (group B, Table 2); and eight underwent only the occupancy scan (group C, Table 2). Thus, the 28 [18F]fallypride scans of the study consisted of 11 baseline scans and 17 occupancy scans. Pooled medication-free durations at baseline and treatment durations before occupancy scans are shown in Table 1. For the baseline scans, four group A subjects were drug-naïve and the other 5 were off medications for 30–840 (234±344) days, while the two group B subjects were off medications for 24 days and 7 years, respectively. At the time of occupancy scans, the duration of aripiprazole treatment for group A subjects was 10–49 (28±13) days and for group B subjects was 10–140 (43±44) days. The daily clinically determined doses of aripiprazole were 2 mg (n=1), 2.5 mg (n=1), 5 mg (n=3), 7.5 mg (n=1), 10 mg (n=4), 15 mg (n=3), 25 mg (n=1), 30 mg (n=2), and 40 mg (n=1). Scans were obtained at a similar time of day (mid-afternoon).
Radiochemistry
[18F]fallypride was prepared by reacting the starting material tosylate (2–3 mg) with resolubilized K[18F]F/K222 in acetonitrile (1 ml) at 80°C for 15 min. The starting material was prepared according to a modified literature procedure (Mukherjee et al, 1995). The crude reaction mixture was mixed with water (20 ml) and passed through a C-18 Sep-Pak. The Sep-Pak was washed with 20 ml of 20% aqueous ethanol and the crude product was recovered with 1.5 ml of ethanol, which was then purified by a semipreparative HPLC method. The HPLC product fraction was mixed with 100 ml of water and passed through a C-18 Sep-Pak. After 20% ethanol (10 ml) and water (10 ml) wash, the tracer was recovered from the Sep-Pak using 1 ml of absolute ethanol. The average radiochemical yield was about 30% at the end of bombardment or about 15% at the end of synthesis. A small sample from the ethanol solution was removed for the determination of specific activity, radiochemical purity, and chemical purity. The rest of the ethanol solution was diluted with saline (9 ml) and filtered through a sterile membrane filter into a vented sterile sample vial. The injected mass was 0.75±0.33 μg, specific activity was 1378±964 Ci/mmol, and activity dose was 2.39±1.38 mCi at time of injection (n=28).
PET Scanning
Positron emission tomography (PET) imaging was performed in 3D mode with the high-resolution ECAT EXACT HR+ scanner (Siemens/CTI, Knoxville, TN) (Brix et al, 1997). Ten-minute transmission scans were obtained as detailed below. [18F]fallypride was injected i.v. over 30 s. For baseline scans, emission data were acquired over 240 min as 24 frames of increasing duration (3 × 20 s, 3 × 1 min, 3 × 2 min, 2 × 5 min, and 13 × 10 min, totaling 150 min of acquisition time). During the 240 min, there were two breaks out of the camera as follows: at 50 min, a 10-min transmission scan was followed by a 20-min break and at 140 min, a 10-min transmission scan was followed by a 40-min break followed by a final 10-min transmission scan preceding the final acquisitions. Thus, emission data were obtained in three successive blocks of 50, 60, and 40 min. Occupancy scans, with their faster kinetics, were acquired over 120 min as 21 frames of increasing duration (3 × 20 s, 3 × 1 min, 3 × 2 min, 2 × 5 min, and 10 × 10 min) in one uninterrupted block. The difference in scan duration between conditions was necessary because of the longer time to peak concentration of [18F]fallypride in the baseline condition. Following aripiprazole treatment, calculated volumes of distribution were found to be time stable with 2 h of scanning in all brain regions (Laruelle et al, 2003). PET emission data were attenuation-corrected using the transmission scans, and frames were reconstructed using a Shepp filter (cutoff 0.5 cycles/projection ray).
Input Function Measurement
Arterial access was available for n=25 of the 28 scans (all baseline scans and n=14 of the 17 occupancy scans). Following radiotracer injection, arterial samples were collected and radioactivity measured as described previously (Abi-Dargham et al, 1999). Five plasma samples (collected at 2, 20, 40, 80, and 120 min) were processed by HPLC to measure the fraction of plasma radioactivity representing unmetabolized parent tracer. A three-exponential-fitted input curve for kinetic analyses was generated as described previously (Abi-Dargham et al, 1999).
Clinical Ratings and Assessment
The PANSS rating scale (Kay et al, 1987) was administered twice, at baseline (while unmedicated) and on the study dose of aripiprazole, to seven of the nine patients who underwent both [18F]fallypride baseline and aripiprazole occupancy scans. The remaining subjects had ratings only at baseline (n=4) or on treatment (n=8), that is, two of the patients were not rated on medication. Clinical assessment of extrapyramidal signs and akathisia was made at the time of the occupancy scan by the treating psychiatrist for all the 17 subjects receiving aripiprazole.
Plasma Aripiprazole Levels
Plasma aripiprazole was assayed (Analytical Psychopharmacology Laboratory, Nathan Kline Institute, Orangeburg, NY) using reverse phase liquid chromatography with ultraviolet detection. Blood for these assays was drawn at the time of PET scan just before radioligand injection and at the end of scan 2 h later. The two plasma levels for each scan were averaged and compared with the daily dose of aripiprazole (Figure 1), showing a high correlation (n=15, Pearson's R=0.814, p=0.0002). Given this high correlation, since plasma samples were unavailable for 2 of the 17 subjects participating in occupancy scans, subsequent analyses of occupancy were performed in comparison to aripiprazole dose rather than plasma levels.
PET Data Analysis
Each patient underwent a high-resolution T1-weighted magnetic resonance imaging (MRI) scan on a GE-Signa 3T system. For each PET scan, frame to frame registration (Woods et al, 1993) using the MEDx (Sensor Systems Inc., Sterling, VA) software environment was followed by co-registration to the subject's MRI using mutual information maximization as implemented in SPM2 (Friston et al, 1995). The following regions of interest (ROIs) were drawn on each subject's MRI using MEDx: cerebellum; pituitary; 5 striatal subregions (precommissural dorsal caudate, posterior caudate, precommissural dorsal putamen, posterior putamen, ventral striatum (Martinez et al, 2003)); and 7 extrastriatal regions (amygdala, thalamus, hippocampus, temporal cortex, insula, midbrain, and entorhinal cortex). Right and left regions were averaged. Outcome measures were calculated using two methods. The first of these was the simplified reference tissue model (SRTM) (Lammertsma and Hume, 1996), yielding BPND (n=17), a binding potential measure denoting the ratio at equilibrium of specifically bound radioligand to that of nondisplaceable radioligand in tissue (Innis et al, 2007). The second method was the two tissue compartment model with arterial input function (2TCM), yielding both BPND and BPP, the latter referring to the ratio at equilibrium of specifically bound radioligand to that of total parent radioligand in plasma (Innis et al, 2007) (n=14; three subjects lacked arterial access for the occupancy scan). For each of these three BP measures, occupancy by aripiprazole was computed in each region as 100%*(1−BPocc/BPbsl), where ‘occ’ refers to the scan while medicated and ‘bsl’ refers to each individual's own scan off aripiprazole for group A, or to the mean of the n=11 off-aripiprazole scans for group C. For comparison, occupancy was also calculated for subjects in group A using ‘bsl’ as the average of the 11 off-aripiprazole scans. The results of the two different ‘bsl’ methods are referred to in the Results section as ‘mixed baseline’ in the former case and ‘average baseline’ in the latter.
A pituitary ROI dose–response estimate was made separately using a non-model-based approach. Tracer kinetics modeling relies on assumptions that may be unreliable for the pituitary, which is outside the blood–brain barrier (BBB): (1) the BBB excludes any radiolabeled metabolites of the parent radioligand from penetrating the ROI and contaminating the signal and (2) a reference region free of D2 receptors provides a reliable estimate of nondisplaceable binding in the modeled ROI. Therefore, a procedure for estimating the effect of aripiprazole on [18F]fallypride signal in pituitary that avoids these assumptions was used. We computed the percent change in the area under the pituitary time-activity curve (AUC), normalized to injected dose of radioligand, between the baseline and occupancy scans, for the nine subjects in group A. The AUC was sensitive to the scan truncation time and was evaluated two ways, by (1) truncating at end of scan, which was at 120 min for occupancy scans and 240 min for baseline scans and (2) truncating all scans uniformly at 120 min. The AUC more closely corresponds to the total volume of distribution VT than to BP, and no estimate of nonspecific binding was made. For this reason, the measured fractional changes following aripiprazole in pituitary were expected to be less than the occupancy measures computed in brain ROIs. For comparison, SRTM using cerebellum as a reference region was applied to these data as well.
Statistical Tests
Group means are presented as average±SD.
Regression analysis with the Pearson's correlation coefficient was used to compare plasma aripiprazole levels to aripiprazole dose, as well as to compare D2/3 receptor occupancy by aripiprazole to changes in PANSS subscale ratings.
ED80 values for each of the ROIs were obtained by use of the Prism software system (GraphPad Software Inc., San Diego, CA) to fit the dose–occupancy data for each region assuming a range from 0 to 100% occupancy. Pooled data from all regions were used to test the hypothesis of a single ED80 for all brain regions vs different ED80 values in striatal subregions and extrastriatal regions (F-test). The choice of the ED80 rather than the ED50 was based on its greater relevance to the clinical range of medication dosing, but statistical tests are unchanged, since the ED80 is a fixed multiple (four) times the ED50, affecting means and SDs equally.
RESULTS
Injected doses, masses, and specific activities of [18F]fallypride did not differ between baseline and occupancy scans (Table 3). There was no change in VND (cerebellum VT) on aripiprazole compared to baseline in the six subjects with arterial access during both scans (Table 3; p=0.72).
Regional Brain Uptake of [18F]Fallypride
Striatal and extrastriatal uptake of [18F]fallypride was in accordance with the known distribution of D2/3 receptors (Lahti et al, 1995; Rieck et al, 2004). At baseline, extrastriatal uptake was about 5% of striatal uptake (see Figure 2 and left ‘baseline’ panel of Figure 3, where the average baseline [18F]fallypride BPND computed on a voxelwise basis using the SRTM across the nine patients in group A is displayed in a common anatomic space). Similar results were obtained with 2TCM. The remaining panels in Figure 3 show the effects on [18F]fallypride binding of increasing doses of aripiprazole, which are quantified below as receptor occupancy.
Regional Receptor Occupancy
In general, for the wide range of clinically determined dosing of aripiprazole in this study, occupancy levels were high across both striatal and extrastriatal regions. Occupancy levels averaged over all subjects and all regions ranged from 71.6±5.5% at 2 mg/day dose to 96.8±5.3% at 40 mg/day based on 2TCM. However, the data showed a systematic difference between striatal and extrastriatal regions.
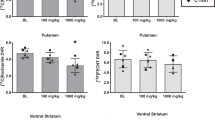
We generated dose–occupancy curves as described in the Methods section for each of the 12 regions analyzed, yielding the ED80 for each region. All available cases (groups A and C) were included for each of the three outcome measures: BPND using SRTM (n=17) and BPND as well as BPP using 2TCM (n=14 with arterial access). These different outcome measures consistently showed higher occupancy in extrastriatal compared to striatal regions for a given dose. For example, striatal occupancies were 79.8±14.8% compared with 83.1±10.9% extrastriatal at 10 mg/day and 83.5±3.2% striatal compared with 85.5±9.0% extrastriatal at 15 mg/day based on SRTM. Equivalently, the ED80 was higher in the striatum than in extrastriatal regions. As seen in Figure 4, the mean ED80 for 2TCM was 44% greater in the striatal compared with the extrastriatal regions, corresponding to higher mean extrastriatal occupancy. This difference, while corresponding to the clinically relatively small ED80 dose difference of 1.7 mg/day, is statistically significant (F-test, p=0.004). The results for all three outcome measures using ‘mixed baseline’ and ‘average baseline’ are shown in Table 4, showing similar ED80 differences with all outcome measures. Specific regional results for subjects in group A are displayed in Figure 5.
Dose–occupancy curves for striatal and extrastriatal brain regions based on 2TCM tracer kinetic modeling with arterial input function and the BPND outcome measure for n=14 subjects (3 of 17 subjects who underwent occupancy scans did not have arterial access). A nested hypothesis test on data pooled from all regions showed significant difference between ED80 in striatal and extrastriatal regions (F test, p=0.004). (a) Occupancy data from striatum (filled circles) and extrastriatal regions (open circles) and dose–occupancy curves based on the mean of 5 striatal subregions (solid curve, ED80=5.63±1.00 mg) and 7 extrastriatal subregions (dashed curve, ED80=3.90±1.32 mg). (b and c) Insets showing modeled curves from pooled data in striatum (b) and extrastriatal regions (c) with 95% prediction bands showing spread of data (dashed) and 95% confidence intervals for the curve fits (dotted). Note the ED80 from pooled data in b and c are slightly lower than the ED80 averaged across individual regions in a (5.14 in striatum, 3.34 in extrastriatal regions).
Bar charts showing occupancy by aripiprazole computed with SRTM for nine subjects in striatal subregions (anp, anterior putamen or precommissural dorsal putamen; dca, precommissural dorsal caudate; pca, posterior caudate; pop, posterior putamen; vst, ventral striatum) (a) and extrastriatal regions (ins, insula; tha, thalamus; mid, midbrain; tem, temporal cortex; amy, amygdala; ent, entorhinal cortex; hip, hippocamus) (b) at 2, 5, 10, and 30 mg/day. Occupancy was above 50% at the lowest dose of 2 mg/day of aripiprazole, and reached 75–80% at 10 mg/day. For many regions, the 30 mg/day dose resulted in occupancy in the 90% range.
Effects of Modeling Method
Occupancy computations based on all combinations of modeling method and subject grouping were in substantial agreement on the dose–occupancy relationships and the greater ED80 values in striatum compared to extrastriatal regions (Table 4). There was a tendency for SRTM to yield systematically lower values of occupancy (higher ED80) compared with 2TCM. This was also the case for baseline BPND values as shown by region in Table 5. 2TCM baseline BPND values were 27±7% higher than the SRTM values, although the two methods were highly correlated (r=1.00, p<0.0001). The ‘average baseline’ method yielded poorer statistical power to detect striatal/extrastriatal differences as expected by the sacrifice of specific knowledge of the within-subject baseline BP values for the nine cases where such data were available.
Clinical Ratings and Assessment
For the seven subjects who were rated with the PANSS scale before and after aripiprazole treatment, the relationships between the changes in the subscales and aripiprazole occupancy were evaluated. As shown in Figure 6, the positive-symptom subscale changes correlated positively with striatal subregional occupancy. The extrastriatal regions examined individually showed no such correlations, and the negative-symptom subscale changes showed no significant correlation with occupancy in striatal or extrastriatal regions.
Comparison of PANSS positive-symptom subscale treatment-related changes with regional aripiprazole occupancy determined by SRTM. The striatal subregion occupancies correlate with improvements in the positive-symptom subscale. The negative-symptom subscale did not show such correlations and neither subscale correlated with extrastriatal occupancy.
Pituitary
The estimation method for the pituitary showed a clear dose response, with fractional AUC decreases following aripiprazole ranging from 39.3±14.9% at 2 mg/day to 67.3±0.2% at 30 mg/day. This is consistent with the expectation that decreases in AUC would be relatively less than decreases in brain BP due to the lack of correction for nonspecific binding (Figure 7A). In addition, truncation of all integrations at 120 min further decreased the estimated change in pituitary AUC, ranging from 13.7±5.1% at 2 mg/day to 49.0±0.4% at 30 mg/day (Figure 7B). This can be understood by noting that the slower kinetics of the baseline scans results in greater contribution to the AUC after 120 min for baseline than for occupancy scans. SRTM modeling produced a nearly uniform decrease that was independent of dose (ED80=10.8 mg; Figure 7C), suggesting that pituitary data were not well characterized by this model. Qualitatively, Figure 7 suggests that pituitary D2 receptor occupancy by aripiprazole may be similar to the striatal and extrastriatal brain regions examined.
Estimated pituitary occupancy by aripiprazole computed by integrating AUC using full scan acquisition times (a), truncating all integration times to 120 min (b) and using SRTM analysis with cerebellum as reference region (c) showing results for 2, 5, 10, and 30 mg/day in the n=9 subjects with baseline and occupancy scans.
Prolactin levels were obtained on aripiprazole for n=8 of the subjects and in all cases were within the normal range for this hormone and unrelated to the extent of pituitary occupancy by aripiprazole.
DISCUSSION
These data show, for the first time, in patients with schizophrenia, simultaneous striatal and extrastriatal regional D2 receptor occupancy by a wide range of clinically relevant doses of aripiprazole. Consistent with previous findings (Mamo et al, 2007; Yokoi et al, 2002), we observed high D2 receptor occupancy by aripiprazole in the absence of EPS or prolactin elevation. These results are consistent with aripiprazole's high affinity for the D2 receptor and its behavior as a partial agonist for D2 receptor-mediated inhibition of cAMP accumulation, with intrinsic activity ∼25% that of dopamine (Burris et al, 2002). In this study, consistent with the expected effects of a partial agonist, increasing concentrations of aripiprazole blocked dopamine's action, with maximal blockade equal to the agonist effect of aripiprazole alone. In this scenario, aripiprazole would function as a D2 receptor antagonist in the presence of high dopamine stimulation, but would maintain baseline D2 receptor signaling above that of no agonist stimulation.
The notion that a single compound acts at a given receptor to produce a similar effect in all effector pathways appears to be oversimplified, and there is increasing evidence that a ligand can have different intrinsic activities at different effectors and in different cellular contexts (Mailman, 2007; Urban et al, 2007a). This mechanism has been termed ‘functional selectivity’ (Lawler et al, 1999) or ‘agonist trafficking’ (Kenakin, 1995). Indeed, aripiprazole has been found to have a complex pharmacology consistent with functional selectivity at the D2 receptor. Although in some cellular systems aripiprazole behaves as a partial agonist at cAMP accumulation, in other cellular contexts it behaves as a full antagonist (Shapiro et al, 2003). Furthermore, aripiprazole behaves as a partial agonist at calcium mobilization (Jordan et al, 2007), MAP kinase activation (Jordan et al, 2007; Urban et al, 2007b), and promotion of arachidonic acid release (Urban et al, 2007b). In addition, aripiprazole, unlike dopamine, did not lead to D2 receptor internalization, also consistent with its functional selectivity (Urban et al, 2007b). Aripiprazole's in vivo behavior as a presynaptic D2 receptor agonist and postsynaptic D2 receptor antagonist (Kikuchi et al, 1995) may result from the interplay of its functional selectivity in these different cellular contexts.
A property of G protein-coupled receptors proposed to help explain functional selectivity effects is that of constitutive activity. In the absence of ligand, receptors isomerize between different conformations, some inactive and some, by virtue of their capability to activate G-proteins, active (Samama et al, 1993). A partial agonist may be a ‘protean’ agonist in that its effect may vary depending on the level of intrinsic activity of the system. In quiescent systems, the ligand would be an agonist, whereas in a sufficiently constitutively active system it would be an inverse agonist (Kenakin, 2007). Thus, although the exact molecular mechanisms at play are under active study and not yet well understood, these studies support a unique pharmacology for aripiprazole and may provide the beginning of a molecular understanding of its ability to block some aspects of D2 receptor signaling while preserving or even enhancing others. Our findings suggest that imaging can provide a powerful approach to explore functional selectivity in vivo and to differentiate the pharmacological properties of D2 receptor ligands.
Differential Occupancy
This is the first report of combined striatal and extrastriatal D2 occupancy for aripiprazole, showing a slightly but significantly higher differential occupancy of extrastriatal vs striatal D2. This finding should be discussed in two ways: its clinical implications and its theoretical mechanistic underpinnings. Clinically, mean ED80 regional differences of about 2 mg/day are probably too small to be meaningful for clinical treatment considering that this drug is usually used in doses of 10 mg/day and higher. Nevertheless, this may be relevant for the therapeutic effect that may be observed at the lower doses of 2 and 5 mg, which will result in predominantly more extrastriatal than striatal occupancy. It may be interesting to better characterize patients who respond at this lower level in terms of their baseline pathophysiology, assessing factors such as dysregulation of baseline dopamine in extrastriatal regions and whether these predict the therapeutic response. No such evidence is available at this point, but this investigation is now possible with the use of tracers that assess extrastriatal D2 receptors, such as [18F]fallypride and [11C]FLB457, combined with acute challenges.
Mechanistically, there could be multiple explanations for this observed difference. One is a difference in regional occupancy of receptors by endogenous dopamine, either due to different dopamine levels or different average regional affinities of D2 receptors for dopamine. If the differences in regional ED80 values arise solely from binding competition between aripiprazole and endogenous dopamine, a linear relationship between the ratios of dopamine to its inhibition constant (denoted RDA, see Appendix) in extrastriatal vs striatal brain regions can be inferred. The differences in ED80 of Figure 4 give an estimate of the greater endogenous dopamine occupancy of striatal D2 receptors compared with extrastriatal regions. For example, striatal endogenous dopamine occupancy of 31% (RDA=0.44 in striatum) corresponds to negligible extrastriatal occupancy of 0% (RDA=0, extrastriatal). However, using α-MPT depletion of DA, we have previously measured baseline occupancy of D2 receptors by dopamine at levels close to 20% in patients with schizophrenia (Abi-Dargham et al, 2000). At this level, the linear model would predict negative occupancy in extrastriatal regions (Figure 8), suggesting that competition with dopamine is at best only a partial explanation of the observed difference in regional aripiprazole occupancy.
Plot of RDA,E (the ratio of extrastriatal free dopamine concentration to the dopamine-D2 receptor dissociation constant) vs RDA,S (same ratio for striatal dopamine) for a range of RDA,S values (see Appendix) using the ED80 ratio value .693 obtained with the BPND outcome measure evaluated using kinetic modeling (Figure 4). This graph shows that this ED80 ratio is compatible with dopamine RDA,S greater than .44 (the intercept on the x-axis), or striatal occupancy greater than 31%.
Another possible mechanism is a higher affinity of the D2 receptor for aripiprazole in extrastriatal regions vs striatal regions so that the same dose results in a higher binding in extrastriatal regions vs striatal regions. A higher affinity in turn could result from differences in the receptor or differences in the proportion of receptors in the high affinity (D2H) vs low affinity (D2L) state for dopamine. These differences could be related to differences in baseline dopamine levels. It is known that dopamine in the striatum ranges from nM to μM levels, while in the cortex it ranges from pM to nM levels (Seamans and Yang, 2004), with the overall tissue content of dopamine in the prefrontal cortex about 100-fold less than that in the striatum (Garris et al, 1993). Higher dopamine could result in lower D2H and lower binding of a partial agonist, such as aripiprazole, thus lower occupancy. This explanation is somewhat speculative, however, as the intrinsic activity of aripiprazole is quite low and differences in its affinity for D2H and D2L are quite small in vitro (Urban et al, 2007b).
A mechanism that would cause an artifact leading to apparent occupancy differences is regionally different re-regulation (ie, upregulation) of receptors in response to aripiprazole treatment. If, for example, there were upregulation of receptors in striatum only, then apparent striatal occupancy would be artifactually diminished, due to increased BP of [18F]fallypride during the occupancy scan associated with an increased receptor pool and not directly attributable to regionally different aripiprazole occupancy. Our methods would be unable to discriminate between such an artifact and authentic regional occupancy differences.
Another source of possible artifact in the measured occupancy differential across regions is related to the kinetic interaction between aripiprazole and [18F]fallypride. The model we have used to estimate occupancy is based on the assumption that aripiprazole concentration is constant during the scanning period, but in fact it is changing, moving from peak to trough values over the course of the dosing cycle. While the terminal plasma washout half-life of steady state aripiprazole concentration has been estimated to be in excess of 60 h (Mallikaarjun et al, 2004), the measured peak to trough change is more rapid. For example, Figure 2 in Mallikaarjun et al (2004) shows measured plasma concentration changes of up to 30% over 8–9 h, corresponding to an half-life in the range of 15–16 h. If receptor occupancy by aripiprazole is changing at a similar rate during the scanning period, our method would measure a weighted average of the dynamically changing receptor occupancy. The weights would depend on the interaction between bound aripiprazole and unbound [18F]fallypride, and unbound [18F]fallypride concentration in turn varies across regions as a function of receptor density. The model and simulations we present in the Appendix suggest that this phenomenon could account for regional differences in occupancy estimates if scanning was consistently performed during the peak to trough phase of the dosing cycle, with bound aripiprazole changing at a similar rate as the plasma changes in Mallikaarjun et al (2004), but we have no evidence for the amount of aripiprazole change and scanning was performed at least 6 h after the last dose.
Another potential source of artifactual regional occupancy differences might be the presence of D2/3 receptors in the cerebellar region of reference, assumed to be devoid of these receptors. The presence of a small number of receptors in the reference region might be expected to affect lower D2/3 density ROIs more than higher density ROIs, producing a regional bias artifactually. As noted, however, we found no change in VND (cerebellum VT) due to the presence of aripiprazole compared to the baseline in the six subjects with arterial access during both scans, indicating the absence of this potential confound. Furthermore, as shown in the Appendix, the presence of receptors in the reference region would in fact bias all regions in the same way, regardless of ROI receptor density, again eliminating cerebellar D2/3 receptors as a potential confound.
The radiotracer we used does not distinguish between D2 and D3 receptors. It is known that the D3 is present in discrete regions of the brain such as the ventral striatum, globus pallidum, and thalamus. Occupancy in these regions reflects both receptor populations.
Clinical Ratings
Correlations of ratings of clinical improvement of positive symptoms with occupancy in striatal regions should be regarded as preliminary, since the number of subjects rated was small (n=7) and the relationship was significant in ventral striatum but only trend level in associative striatum. Furthermore, noisier occupancy data in extrastriatal regions arising from markedly lower D2/3 receptor density may have masked associations in these regions. However, these correlations are in agreement with one previous finding (Agid et al, 2007) and suggest that this partial agonist agent, as do D2 antagonists, benefits positive symptoms of schizophrenia through its modulation of striatal but not cortical or other extrastriatal dopamine activity. These data are consistent with the predominant involvement of limbic and associative striatum compared to sensorimotor striatum in the therapeutic effect of antipsychotics. Treatment was optimized for each patient independently of the study and the scan was obtained shortly after initiation of treatment except for four patients treated for 6 weeks or longer (for the remaining patients, treatment duration was 10–37 days, mean 23±9 days). It has been reported that most of the therapeutic response is measured in the first 4 weeks of treatment (Kerwin et al, 2007), so we predict that the treatment response measured here is likely to be close to the optimal response. The lack of relationship with improvement in negative symptoms suggests that treatment of negative symptoms is more multifactorial than that of positive symptoms and may depend on occupancy of other systems and in other regions.
Four cases were studied after a more prolonged treatment as noted. In these four cases, occupancy measures can be underestimated due to increases in receptor expression with chronic treatment. These changes are likely to be minimal in the first few weeks, and are likely to be minimal with a partial agonist (Inoue et al, 1998; Ozdemir et al, 2002).
Pituitary Gland
Aripiprazole appears to occupy a significant proportion of D2 receptors in the pituitary, with no correlation between occupancy and prolactin levels. These estimates are the first ever derived from in vivo imaging data; to our knowledge, no quantification of D2-like receptor occupancy in the pituitary has been published, although D2 radioligand binding in this region has been demonstrated qualitatively (Bergstrom et al, 1991). Measuring occupancy in the pituitary is a challenge due to the absence of applicable models. However, within these constraints, we used a straightforward method based on the AUC. As a result, the denominator in the expression for the percent changes includes nonspecific binding and possibly radiolabeled metabolites. These quantities were not expected to change across conditions, so that the observed systematically lower change compared to the occupancy condition as measured in brain was not a surprise. For the sake of comparison, we also computed the conventional occupancy measure in pituitary, using SRTM with cerebellum as reference region. While substantial apparent occupancy was observed with this approach, the results were not well correlated with dose (Figure 7c). Although it remains for future work to characterize the pharmacokinetics of this structure more precisely, the substantial pituitary occupancy observed in the absence of prolactin elevation argues for sufficient partial agonism of aripiprazole at pituitary D2 receptors to avoid the functional antagonism that leads to prolactin elevation with other antipsychotic medications.
Substantia Nigra
The substantia nigra has previously been reported to be relatively spared of D2 occupancy by atypical antipsychotic medications such as clozapine and quetiapine compared with cortical regions (Kessler et al, 2006). On inspection, we found that our midbrain ROI definition closely encompassed the substantia nigra as defined in Kessler et al (2006), and our data show no such occupancy sparing for aripiprazole in that region, with over 65% occupancy at 2 mg/day and over 90% occupancy at 30 mg/day, based on SRTM, comparable to other extrastriatal regions.
CONCLUSION
In conclusion, this study is the first report on extrastriatal and striatal D2 receptor occupancy in patients with schizophrenia by various doses of aripiprazole, a partial D2 receptor agonist, and the first estimate of pituitary D2 receptor occupancy. We confirm the in vivo partial agonism of the drug as we find high occupancies in the absence of EPS or prolactin elevations. In addition, we find higher occupancy by aripiprazole in extrastriatal compared with striatal regions, although the differential occupancy is small and the clinical improvement in positive symptoms is related to striatal rather than extrastriatal regions. Higher extrastriatal occupancy by aripiprazole may be related to higher striatal than extrastriatal dopamine concentrations but also could be an artifact derived from differential kinetics across these regions. Further studies using established methods of assessing dopamine function such as amphetamine-induced dopamine release or dopamine depletion for evaluating baseline dopamine levels are needed to investigate this finding. These studies may also clarify the issue of potential therapeutic effect of extrastriatal occupancy by providing information about baseline stimulation of the D2 receptors in these regions.
References
Abi-Dargham A, Rodenhiser J, Printz D, Zea-Ponce Y, Gil R, Kegeles LS et al (2000). Increased baseline occupancy of D2 receptors by dopamine in schizophrenia. Proc Natl Acad Sci USA 97: 8104–8109.
Abi-Dargham A, Simpson N, Kegeles L, Parsey R, Hwang D-R, Anjilvel S et al (1999). PET studies of binding competition between endogenous dopamine and the D1 radiotracer [11C]NNC 756. Synapse 32: 93–109.
Agid O, Mamo D, Ginovart N, Vitcu I, Wilson AA, Zipursky RB et al (2007). Striatal vs extrastriatal dopamine D2 receptors in antipsychotic response—a double-blind PET study in schizophrenia. Neuropsychopharmacology 32: 1209–1215.
Bergstrom M, Muhr C, Lundberg PO, Langstrom B (1991). PET as a tool in the clinical evaluation of pituitary adenomas. J Nucl Med 32: 610–615.
Bigliani V, Mulligan RS, Acton PD, Visvikis D, Ell PJ, Stephenson C et al (1999). In vivo occupancy of striatal and temporal cortical D2/D3 dopamine receptors by typical antipsychotic drugs. [123I]epidepride single photon emission tomography (SPET) study. Br J Psychiatry 175: 231–238.
Brix G, Zaers J, Adam LE, Bellemann ME, Ostertag H, Trojan H et al (1997). Performance evaluation of a whole-body PET scanner using the NEMA protocol. National Electrical Manufacturers Association. J Nucl Med 38: 1614–1623.
Burris KD, Molski TF, Xu C, Ryan E, Tottori K, Kikuchi T et al (2002). Aripiprazole, a novel antipsychotic, is a high-affinity partial agonist at human dopamine D2 receptors. J Pharmacol Exp Ther 302: 381–389.
Deutch AY (1992). The regulation of subcortical dopamine systems by the prefrontal cortex: interactions of central dopamine systems and the pathogenesis of schizophrenia. J Neural Transm Suppl 36: 61–89.
DSM-IV-TR: Diagnostic and Statistical Manual of Mental Disorders, 4th edn, Text Revision (2000). American Psychiatric Association: Washington, DC.
Farde L, Nordström AL, Wiesel FA, Pauli S, Halldin C, Sedvall G (1992). Positron emission tomography analysis of central D1 and D2 dopamine receptor occupancy in patients treated with classical neuroleptics and clozapine. Arch Gen Psychiatry 49: 538–544.
Farde L, Suhara T, Nyberg S, Karlsson P, Nakashima Y, Hietala J et al (1997). A PET-study of [11C]FLB 457 binding to extrastriatal D2-dopamine receptors in healthy subjects and antipsychotic drug-treated patients. Psychopharmacology (Berlin) 133: 396–404.
Farde L, Wiesel FA, Halldin C, Sedvall G (1988). Central D2-dopamine receptor occupancy in schizophrenic patients treated with antipsychotic drugs. Arch Gen Psychiatry 45: 71–76.
Friston KJ, Holmes AP, Worsley KJ, Poline J-P, Frith CD, Frakowiak RSJ (1995). Statistical parametric maps in functional imaging: a general linear approach. Hum Brain Mapping 2: 189–210.
Garris PA, Collins LB, Jones SR, Wightman RM (1993). Evoked extracellular dopamine in vivo in the medial prefrontal cortex. J Neurochem 61: 637–647.
Grunder G, Landvogt C, Vernaleken I, Buchholz HG, Ondracek J, Siessmeier T et al (2006). The striatal and extrastriatal D2/D3 receptor-binding profile of clozapine in patients with schizophrenia. Neuropsychopharmacology 31: 1027–1035.
Innis RB, Cunningham VJ, Delforge J, Fujita M, Gjedde A, Gunn RN et al (2007). Consensus nomenclature for in vivo imaging of reversibly binding radioligands. J Cereb Blood Flow Metab 27: 1533–1539.
Inoue A, Seto M, Sugita S, Hide I, Hirose T, Koga N et al (1998). Differential effects on D2 dopamine receptor and prolactin gene expression by haloperidol and aripiprazole in the rat pituitary. Brain Res Mol Brain Res 55: 285–292.
Jordan S, Regardie K, Johnson JL, Chen R, Kambayashi J, McQuade R et al (2007). In vitro functional characteristics of dopamine D2 receptor partial agonists in second and third messenger-based assays of cloned human dopamine D2Long receptor signalling. J Psychopharmacol 21: 620–627.
Kapur S, Zipursky R, Jones C, Remington G, Houle S (2000). Relationship between dopamine D(2) occupancy, clinical response, and side effects: a double-blind PET study of first-episode schizophrenia. Am J Psychiatry 157: 514–520.
Kapur S, Zipursky RB, Remington G (1999). Clinical and theoretical implications of 5-HT2 and D2 receptor occupancy of clozapine, risperidone, and olanzapine in schizophrenia. Am J Psychiatry 156: 286–293.
Kay SR, Fiszbein A, Opler LA (1987). The positive and negative syndrome scale (PANSS) for schizophrenia. Schiz Bull 13: 261–276.
Kenakin T (1995). Agonist-receptor efficacy. II. Agonist trafficking of receptor signals. Trends Pharmacol Sci 16: 232–238.
Kenakin T (2007). Functional selectivity through protean and biased agonism: who steers the ship? Mol Pharmacol 72: 1393–1401.
Kerwin R, Millet B, Herman E, Banki CM, Lublin H, Pans M et al (2007). A multicentre, randomized, naturalistic, open-label study between aripiprazole and standard of care in the management of community-treated schizophrenic patients Schizophrenia Trial of Aripiprazole: (STAR) study. Eur Psychiatry 22: 433–443.
Kessler RM, Ansari MS, Riccardi P, Li R, Jayathilake K, Dawant B et al (2005). Occupancy of striatal and extrastriatal dopamine D2/D3 receptors by olanzapine and haloperidol. Neuropsychopharmacology 30: 2283–2289.
Kessler RM, Ansari MS, Riccardi P, Li R, Jayathilake K, Dawant B et al (2006). Occupancy of striatal and extrastriatal dopamine D2 receptors by clozapine and quetiapine. Neuropsychopharmacology 31: 1991–2001.
Kessler RM (2007). Aripiprazole: what is the role of dopamine D(2) receptor partial agonism? Am J Psychiatry 164: 1310–1312.
Kessler RM, Mason N, Jones C, Ansari MS, Manning RF, Price RR (2000). [18F]N-allyl-5-fluoropropylepidepride (fallypride): radiation dosimetry, quantification of striatal and extrastriatal dopamine receptors in man. NeuroImage 11: S32.
Kikuchi T, Tottori K, Uwahodo Y, Hirose T, Miwa T, Oshiro Y et al (1995). 7-(4-[4-(2, 3-Dichlorophenyl)-1-piperazinyl]butyloxy)-3, 4-dihydro-2(1H)-qui nolinone (OPC-14597), a new putative antipsychotic drug with both presynaptic dopamine autoreceptor agonistic activity and postsynaptic D2 receptor antagonistic activity. J Pharmacol Exp Ther 274: 329–336.
Lahti RA, Roberts RC, Tamminga CA (1995). D2-family receptor distribution in human postmortem tissue: an autoradiographic study. Neuroreport 6: 2505–2512.
Lammertsma AA, Hume SP (1996). Simplified reference tissue model for PET receptor studies. Neuroimage 4: 153–158.
Laruelle M, Slifstein M, Huang Y (2003). Relationships between radiotracer properties and image quality in molecular imaging of the brain with positron emission tomography. Mol Imaging Biol 5: 363–375.
Lawler CP, Prioleau C, Lewis MM, Mak C, Jiang D, Schetz JA et al (1999). Interactions of the novel antipsychotic aripiprazole (OPC-14597) with dopamine and serotonin receptor subtypes. Neuropsychopharmacology 20: 612–627.
Mailman RB (2007). GPCR functional selectivity has therapeutic impact. Trends Pharmacol Sci 28: 390–396.
Mallikaarjun S, Salazar DE, Bramer SL (2004). Pharmacokinetics, tolerability, and safety of aripiprazole following multiple oral dosing in normal healthy volunteers. J Clin Pharmacol 44: 179–187.
Mamo D, Graff A, Mizrahi R, Shammi CM, Romeyer F, Kapur S (2007). Differential effects of aripiprazole on D(2), 5-HT(2), and 5-HT(1A) receptor occupancy in patients with schizophrenia: a triple tracer PET study. Am J Psychiatry 164: 1411–1417.
Martinez D, Slifstein M, Broft A, Mawlawi O, Hwang DR, Huang Y et al (2003). Imaging human mesolimbic dopamine transmission with positron emission tomography. Part II: amphetamine-induced dopamine release in the functional subdivisions of the striatum. J Cereb Blood Flow Metab 23: 285–300.
Mukherjee J, Yang ZY, Das MK, Brown T (1995). Fluorinated benzamide neuroleptics--III. Development of (S)-N-[(1-allyl-2-pyrrolidinyl)methyl]-5-(3-[18F]fluoropropyl)-2, 3-dimethoxybenzamide as an improved dopamine D-2 receptor tracer. Nucl Med Biol 22: 283–296.
Nordstrom AL, Farde L, Wiesel FA, Forslund K, Pauli S, Halldin C et al (1993). Central D2-dopamine receptor occupancy in relation to antipsychotic drug effects: a double-blind PET study of schizophrenic patients. Biol Psychiatry 33: 227–235.
Nyberg S, Dencker SJ, Malm U, Dahl ML, Svenson JO, Halldin C et al (1998). D(2)- and 5-HT(2) receptor occupancy in high-dose neuroleptic-treated patients. Int J Neuropsychopharmacol 1: 95–101.
Olsson H, Farde L (2001). Potentials and pitfalls using high affinity radioligands in PET and SPET determinations on regional drug induced D2 receptor occupancy—a simulation study based on experimental data. Neuroimage 14: 936–945.
Ozdemir V, Fourie J, Ozdener F (2002). Aripiprazole (Otsuka Pharmaceutical Co). Curr Opin Investig Drugs 3: 113–120.
Pilowsky LS, Mulligan RS, Acton PD, Ell PJ, Costa DC, Kerwin RW (1997). Limbic selectivity of clozapine. Lancet 350: 490–491.
Rieck RW, Ansari MS, Whetsell Jr WO, Deutch AY, Kessler RM (2004). Distribution of dopamine D2-like receptors in the human thalamus: autoradiographic and PET studies. Neuropsychopharmacology 29: 362–372.
Robertson GS, Matsumura H, Fibiger HC (1994). Induction patterns of Fos-like immunoreactivity in the forebrain as predictors of atypical antipsychotic activity. J Pharmacol Exp Ther 271: 1058–1066.
Samama P, Cotecchia S, Costa T, Lefkowitz RJ (1993). A mutation-induced activated state of the beta 2-adrenergic receptor. Extending the ternary complex model. J Biol Chem 268: 4625–4636.
Seamans JK, Yang CR (2004). The principal features and mechanisms of dopamine modulation in the prefrontal cortex. Prog Neurobiol 74: 1–58.
Shapiro DA, Renock S, Arrington E, Chiodo LA, Liu LX, Sibley DR et al (2003). Aripiprazole, a novel atypical antipsychotic drug with a unique and robust pharmacology. Neuropsychopharmacology 28: 1400–1411.
Shim JC, Shin JG, Kelly DL, Jung DU, Seo YS, Liu KH et al (2007). Adjunctive treatment with a dopamine partial agonist, aripiprazole, for antipsychotic-induced hyperprolactinemia: a placebo-controlled trial. Am J Psychiatry 164: 1404–1410.
Talvik M, Nordstrom AL, Nyberg S, Olsson H, Halldin C, Farde L (2001). No support for regional selectivity in clozapine-treated patients: a PET study with [(11)C]raclopride and [(11)C]FLB 457. Am J Psychiatry 158: 926–930.
Urban JD, Clarke WP, von Zastrow M, Nichols DE, Kobilka B, Weinstein H et al (2007a). Functional selectivity and classical concepts of quantitative pharmacology. J Pharmacol Exp Ther 320: 1–13.
Urban JD, Vargas GA, von Zastrow M, Mailman RB (2007b). Aripiprazole has functionally selective actions at dopamine D2 receptor-mediated signaling pathways. Neuropsychopharmacology 32: 67–77.
Woods RP, Mazziotta JC, Cherry SR (1993). MRI-PET registration with automated algorithm. J Comput Assist Tomogr 17: 536–546.
Xiberas X, Martinot JL, Mallet L, Artiges E, Canal M, Loc’h C et al (2001). In vivo extrastriatal and striatal D2 dopamine receptor blockade by amisulpride in schizophrenia. J Clin Psychopharmacol 21: 207–214.
Yokoi F, Grunder G, Biziere K, Stephane M, Dogan AS, Dannals RF et al (2002). Dopamine D2 and D3 receptor occupancy in normal humans treated with the antipsychotic drug aripiprazole (OPC 14597): a study using positron emission tomography and [11C]raclopride. Neuropsychopharmacology 27: 248–259.
Acknowledgements
We thank Bristol-Myers Squibb for financial support; Erica Scher, Elisa Reich, and Erica Meyers for technical support; and Jonathan Javitch for fruitful scientific discussions.
Author information
Authors and Affiliations
Corresponding author
Additional information
DISCLOSURE/CONFLICTS OF INTEREST
Dr Kegeles has received research support from Pfizer. Dr Slifstein has received research support from GSK and is a consultant for GSK and Amgen. Dr Frankle has received research support from GSK and Sepracor, is a consultant or advisory board member for Sepracor, BMS, Transcept, Eli Lilly, and member of the Speaker's Bureau for BMS and Otsuka pharmaceuticals. Dr Abi-Dargham has received research support from Eli Lilly, BMS, and GSK, is a consultant or advisory board member for Sanofi-Aventis, BMS, Wanda, Eli Lilly, Intracellular Therapeutics, and member of the Speaker's Bureau for Sanofi-Aventis, BMS and, Otsuka pharmaceuticals.
Appendix
Appendix
Throughout the Appendix, the nomenclature of Innis et al (2007) is used: CS (specifically bound compartment concentration), CND (nondisplaceable compartment concentration), and fND (tissue free fraction) are as defined in that reference and BPP and BPND are as defined in the reference and in the Methods section above.
A.1: DERIVATION OF DOPAMINE OCCUPANCY RATIOS FROM REGIONAL DIFFERENCES IN ED80
The assumptions in this estimating procedure are that (i) aripiprazole has the same affinity for all D2-like receptors in all regions, (ii) the concentration of dopamine in the proximity of the receptors is not substantially changed following aripiprazole treatment compared to baseline, and (iii) ED80 ratios between regions are equivalent to EC80 ratios between regions.
Let RA,S, RA,E, RDA,S, and RDA,E be the ratio of concentration to inhibition constant for aripiprazole (A) and dopamine (DA) in striatum (S) and extrastriatal regions (E). Then, occupancy in both region types is equal to

Let ED80(ROI) be the estimated ED80 for a given ROI type, striatal (STR) or extrastriatal (EXT). Then, at the ED80 concentrations, RDA,E is given by the affine function (line with non-zero intercept) of RDA,S

Figure 8 is a plot of RDA,E vs RDA,S for a range of RDA,S values using the ED80 ratio value of .693 observed in this data set generated from BPND values from 2TCM (Figure 4).
A.2: DRUG OCCUPANCY MEASURED BY PET WHEN DRUG CONCENTRATION IS CHANGING DURING THE SCAN
Theory
An underlying assumption used to derive aripiprazole occupancy from the fractional change in BP is that the bound drug concentration is constant during the scanning period. If this quantity is changing during the course of the scan, the apparent occupancy will then be a weighted sum of the dynamically changing occupancy over the course of the scan, and the weights will be influenced by time-varying free concentration of the radioligand. Free radioligand concentration will be different across brain regions, due to the ability of the receptor pool to act like a capacitance, removing ligand from the free compartment by specific binding in the early scan phase when free ligand exceeds the equilibrium binding point, and returning ligand to the free compartment in the later phase when specifically bound ligand exceeds the equilibrium point. The phenomenon will be more pronounced in high receptor density regions (striatum) than in low receptor density regions (cortex and limbic regions), leading to different weights in the estimated occupancies. In particular, if receptor-bound drug was in an approximately exponentially decreasing phase during the course of the scan, the following analysis and simulation show that this phenomenon could cause apparent differences in occupancy in the absence of true differences.
In addition to the standard modeling assumptions regarding rapid equilibration of radioligand free fractions and constant VND (nonspecific distribution volume) across regions, we also assume here that arterial plasma clearance of the radioligand is the same across conditions. This is only for computational simplification, and does not materially affect the result.
In this study, occupancy is measured as

where drug and baseline refer to the scan conditions. Based on the assumptions above and the fact that compartmental distribution volumes are equivalent to the ratio of the AUC to infinity of the compartment to the AUC of the arterial plasma, and denoting the specifically bound compartment CS, the right-hand side of this expression simplifies to

This is an identity when the modeling assumptions are met exactly. If receptor availability is changing over the course of the scan during the drug condition, the equation is an approximation in the sense that the fitting procedure will find the closest curve to the data from the set of all curves that fit the model equations by least squares minimization, and AUC(CS) of this curve will be influenced by, although not necessarily identical to, AUC(CS) of the data. The AUCs can be derived from the linearized mass action laws for the conditions. Denoting the nondisplaceable compartment as CND,

The parameter k3 equals the constant fND kon Bmax during the baseline condition. Its explicit time dependence applies during the drug occupancy condition. The effect of the drug is represented as a multiplicative factor (k3(t)) due to tracer conditions for the radioligand, so that the drug concentration is not perturbed by the presence of the tracer, and tracer ‘sees’ a time-varying receptor availability. The solution to this differential equation is

for the baseline condition and

for the drug condition, where ⊗ is the convolution operator and D(t) is 1−the fractional occupancy of receptors by the drug at time t. Using the stated assumptions and the properties of convolutions, the occupancy simplifies to

For the case that the bound drug is decreasing exponentially,

where occ0 is the occupancy at the beginning of the scan and the rate constant is λ=ln(2)/τ where τ is the half-life of the drug washout from the receptors. Substituting for D and simplifying leads to

Finally, noting AUC(CND(baseline)) is equal across regions, it can be seen that it is possible for occ0 to be the same across regions, but still have regional differences in estimated occupancy due to differences in AUC(e−λtCND(drug)). Apparent occupancy will therefore be affected by three factors: occ0 and the regional difference in Bmax, each of which influences the shape of CND during the drug condition, and τ, the half-life of the drug.
Simulations
To test the predictive power of this analysis, simulations were performed. Time activity curves were generated using a plasma input function and kinetic parameters similar to human [18F]fallypride parameters in anterior putamen and hippocampus for baseline conditions and for drug occupancy with exponential washout as above. Initial occupancy was tested at 60, 75, and 90%. Based on Figure 2 in Mallikaarjun et al (2004) (also see discussion section), drug off-rates were tested in the range of 10–20 h. For each initial occupancy and washout rate combination, 1000 pairs of curves were generated in each region with Gaussian noise according to the formula C(t) × (1+0.05z), where z was sampled from a standard normal distribution and C(t) was the noise-free time activity curve. Data were then fitted and occupancy computed according to the methods used in the study. Results (Table A1) show that both for the AUC-based prediction and the simulations, striatum occupancy was slightly but consistently less than hippocampus. The results also show that the AUC analysis is numerically closer to striatum fits than hippocampus; in hippocampus, the estimated occupancies are closer to the initial values than the AUC equation predicts. The differences are greatest at high (90%) initial occupancy, where the simulated regional differences are comparable to those seen in the aripiprazole study.
A.3: EFFECT OF D2/3 RECEPTORS IN THE REFERENCE REGION
This section of the Appendix shows that the presence of D2/3 receptors in the reference region would not differentially affect ROIs of low compared with high D2/3 receptor density. Let us assume the null hypothesis that there are a small amount of D2/3 receptors in the reference region, and that after aripiprizole, all regions have the same fractional occupancy by aripiprizole. Let α=1−occupancy, that is, if the baseline binding potential is BP, then after aripiprizole, it becomes αBP. Then at baseline,

and after drug,

and the apparent occupancy is

so that there is a bias (true occupancy=1−α) but it affects all regions to the same extent.
Rights and permissions
About this article
Cite this article
Kegeles, L., Slifstein, M., Frankle, W. et al. Dose–Occupancy Study of Striatal and Extrastriatal Dopamine D2 Receptors by Aripiprazole in Schizophrenia with PET and [18F]Fallypride. Neuropsychopharmacol 33, 3111–3125 (2008). https://doi.org/10.1038/npp.2008.33
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/npp.2008.33
Keywords
This article is cited by
-
Antipsychotic dose, dopamine D2 receptor occupancy and extrapyramidal side-effects: a systematic review and dose-response meta-analysis
Molecular Psychiatry (2023)
-
Therapeutic Reference Range for Aripiprazole in Schizophrenia Revised: a Systematic Review and Metaanalysis
Psychopharmacology (2022)
-
In vivo absolute quantification of striatal and extrastriatal D2/3 receptors with [123I]epidepride SPECT
EJNMMI Research (2020)
-
A positron emission tomography occupancy study of brexpiprazole at dopamine D2 and D3 and serotonin 5-HT1A and 5-HT2A receptors, and serotonin reuptake transporters in subjects with schizophrenia
Neuropsychopharmacology (2020)
-
Switching strategies for antipsychotic monotherapy in schizophrenia: a multi-center cohort study of aripiprazole
Psychopharmacology (2020)