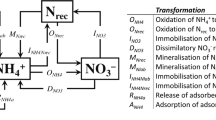
Abstract
The nitrogen (N)-use efficiency of agricultural plants is notoriously poor. Globally, about 50% of the N fertilizer applied to cropping systems is not absorbed by plants, but lost to the environment as ammonia (NH3), nitrate (NO3−), and nitrous oxide (N2O, a greenhouse gas with 300 times the heat-trapping capacity of carbon dioxide), raising agricultural production costs and contributing to pollution and climate change. These losses are driven by volatilization of NH3 and by a matrix of nitrification and denitrification reactions catalysed by soil microorganisms (chiefly bacteria and archaea). Here, we discuss mitigation of the harmful and wasteful process of agricultural N loss via biological nitrification inhibitors (BNIs) exuded by plant roots. We examine key recent discoveries in the emerging field of BNI research, focusing on BNI compounds and their specificity and transport, and discuss prospects for their role in improving agriculture while reducing its environmental impact.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Godfray, H. C. J. et al. Food security: the challenge of feeding 9 billion people. Science 327, 812–818 (2010).
Fowler, D. et al. The global nitrogen cycle in the twenty-first century. Phil. Trans. Roy. Soc. B. 368, 20130164 (2013).
Galloway, J. N. et al. Transformation of the nitrogen cycle: recent trends, questions, and potential solutions. Science 320, 889–892 (2008).
Erisman, J. W., Sutton, M. A., Galloway, J., Klimont, Z. & Winiwarter, W. How a century of ammonia synthesis changed the world. Nat. Geosci. 1, 636–639 (2008).
Cassman, K. G., Dobermann, A. & Walters, D. T. Agroecosystems, nitrogen-use efficiency, and nitrogen management. Ambio 31, 132–140 (2002).
Ladha, J. K., Pathak, H., Krupnik, T. J., Six, J. & van Kessel, C. Efficiency of fertilizer nitrogen in cereal production: retrospects and prospects. Adv. Agron. 87, 85–156 (2005).
Erisman, J. W., Galloway, J., Seitzinger, S., Bleeker, A. & Butterbach-Bahl, K. Reactive nitrogen in the environment and its effect on climate change. Curr. Opin. Environ. Sustain. 3, 281–290 (2011).
Schlesinger, W. H. On the fate of anthropogenic nitrogen. Proc. Natl Acad. Sci. USA 106, 203–208 (2009).
Tilman, D. & Isbell, F. Biodiversity: recovery as nitrogen declines. Nature 528, 336–337 (2015).
Vitousek, P. M. et al. Human alteration of the global nitrogen cycle: sources and consequences. Ecol. Appl. 7, 737–750 (1997).
Townsend, A. R. et al. Human health effects of a changing global nitrogen cycle. Front. Ecol. Environ. 1, 240–246 (2003).
Ravishankara, A. R., Daniel, J. S. & Portmann, R. W. Nitrous oxide (N2O): the dominant ozone-depleting substance emitted in the 21st century. Science 326, 123–125 (2009).
Chen, A. Q. et al. Characteristics of ammonia volatilization on rice grown under different nitrogen application rates and its quantitative predictions in Erhai Lake Watershed, China. Nutr. Cycl. Agroecosys. 101, 139–152 (2015).
Kowalchuk, G. A. & Stephen, J. R. Ammonia-oxidizing bacteria: a model for molecular microbial ecology. Ann. Rev. Microbiol. 55, 485–529 (2001).
Daims, H., Lucker, S. & Wagner, M. A new perspective on microbes formerly known as nitrite-oxidizing bacteria. Trends Microbiol. 24, 699–712 (2016).
Hayatsu, M., Tago, K. & Saito, M. Various players in the nitrogen cycle: diversity and functions of the microorganisms involved in nitrification and denitrification. Soil Sci. Plant Nutr. 54, 33–45 (2008).
Prosser, J. I. & Nicol, G. W. Archaeal and bacterial ammonia-oxidisers in soil: the quest for niche specialisation and differentiation. Trends Microbiol. 20, 523–531 (2012).
Daims, H. et al. Complete nitrification by Nitrospira bacteria. Nature 528, 504–509 (2015).
van Kessel, M. et al. Complete nitrification by a single microorganism. Nature 528, 555–559 (2015).
Jia, Z. & Conrad, R. Bacteria rather than archaea dominate microbial ammonia oxidation in an agricultural soil. Environ. Microbiol. 11, 1658–1671 (2009).
Leininger, S. et al. Archaea predominate among ammonia-oxidizing prokaryotes in soils. Nature 442, 806–809 (2006).
Thion, C. E. et al. Plant nitrogen-use strategy as a driver of rhizosphere archaeal and bacterial ammonia oxidiser abundance. FEMS Microbiol. Ecol. 92, fiw091 (2016).
Venter, J. C. et al. Environmental genome shotgun sequencing of the Sargasso Sea. Science 304, 66–74 (2004).
Woese, C. R. & Fox, G. E. Phylogenetic structure of prokaryotic domain – primary kingdoms. Proc. Natl Acad. Sci. USA 74, 5088–5090 (1977).
Hatzenpichler, R. Diversity, physiology, and niche differentiation of ammonia-oxidizing archaea. Appl. Environ. Microbiol. 78, 7501–7510 (2012).
Hu, H. W., Xu, Z. H. & He, J. Z. Ammonia-oxidizing archaea play a predominant role in acid soil nitrification. Adv. Agron. 125, 261–302 (2014).
Halvorson, A. D., Snyder, C. S., Blaylock, A. D. & Del Grosso, S. J. Enhanced-efficiency nitrogen fertilizers: potential role in nitrous oxide emission mitigation. Agron. J. 106, 715–722 (2014).
Pan, B. B., Lam, S. K., Mosier, A., Luo, Y. Q. & Chen, D. L. Ammonia volatilization from synthetic fertilizers and its mitigation strategies: a global synthesis. Agri. Ecosys. Environ. 232, 283–289 (2016).
Lin, B.-L., Sakoda, A., Shibasaki, R. & Suzuki, M. A modelling approach to global nitrate leaching caused by anthropogenic fertilisation. Water Res. 35, 1961–1968 (2001).
Di, H. J. & Cameron, K. C. Nitrate leaching in temperate agroecosystems: sources, factors and mitigating strategies. Nutr. Cycl. Agroecosys. 64, 237–256 (2002).
Seitzinger, S. et al. Denitrification across landscapes and waterscapes: a synthesis. Ecol. Appl. 16, 2064–2090 (2006).
Forster, P. et al. in Climate Change 2007: The Physical Science Basis (eds Solomon, S. et. al) ch. 2, 129–234 (Cambridge Univ. Press, 2007).
Linquist, B., van Groenigen, K. J., Adviento-Borbe, M. A., Pittelkow, C. & van Kessel, C. An agronomic assessment of greenhouse gas emissions from major cereal crops. Glob. Change Biol. 18, 194–209 (2012).
Turner, P. A. et al. Indirect nitrous oxide emissions from streams within the US corn belt scale with stream order. Proc. Natl Acad. Sci. USA 112, 9839–9843 (2015).
Kool, D. M., Dolfing, J., Wrage, N. & Van Groenigen, J. W. Nitrifier denitrification as a distinct and significant source of nitrous oxide from soil. Soil Biol. Biochem. 43, 174–178 (2011).
Liu, R. et al. Nitrification is a primary driver of nitrous oxide production in laboratory microcosms from different land-use soils. Front. Microbiol. 7, 1373 (2016).
Reay, D. S. et al. Global agriculture and nitrous oxide emissions. Nat. Clim. Change 2, 410–416 (2012).
Smith, P. et al. in Climate change 2007: Mitigation: Contribution of Working Group III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change ( eds Metz, B., Davidson, O. R., Bosch, P. R., Dave, R. & Meyer, L. A. ) Ch. 8, 497–540 (Cambridge Univ. Press, 2007).
Prasad, R. & Power, J. Nitrification inhibitors for agriculture, health, and the environment. Adv. Agron. 54, 233–281 (1995).
Abalos, D., Jeffery, S., Sanz-Cobena, A., Guardia, G. & Vallejo, A. Meta-analysis of the effect of urease and nitrification inhibitors on crop productivity and nitrogen use efficiency. Agri. Ecosys. Environ. 189, 136–144 (2014).
Qiu, H., Sun, D., Gunatilake, S. R., She, J. & Mlsna, T. E. Analysis of trace dicyandiamide in stream water using solid phase extraction and liquid chromatography UV spectrometry. J. Environ. Sci. 35, 38–42 (2015).
Fillery, I. R. Plant-based manipulation of nitrification in soil: a new approach to managing N loss? Plant Soil 294, 1–4 (2007).
Subbarao, G. V. et al. Scope and strategies for regulation of nitrification in agricultural systems — challenges and opportunities. Crit. Rev. Plant Sci. 25, 303–335 (2006).
Akiyama, H., Yan, X. & Yagi, K. Evaluation of effectiveness of enhanced-efficiency fertilizers as mitigation options for N2O and NO emissions from agricultural soils: meta-analysis. Glob. Change Biol. 16, 1837–1846 (2010).
Wedin, D. A. & Tilman, D. Species effects on nitrogen cycling: a test with perennial grasses. Oecologia 84, 433–441 (1990).
Bardgett, R. D., Mommer, L. & De Vries, F. T. Going underground: root traits as drivers of ecosystem processes. Trends Ecol. Evol. 29, 692–699 (2014).
Rice, E. L. & Pancholy, S. K. Inhibition of nitrification by climax ecosystems. III. Inhibitors other than tannins. Am. J. Bot. 61, 1095–1103 (1974).
Basaraba, J. Influence of vegetable tannins on nitrification in soil. Plant Soil 21, 8–16 (1964).
Subbarao, G. et al. A bioluminescence assay to detect nitrification inhibitors released from plant roots: a case study with Brachiaria humidicola. Plant Soil 288, 101–112 (2006).
Subbarao, G. V. et al. Evidence for biological nitrification inhibition in Brachiaria pastures. Proc. Natl Acad. Sci. USA 106, 17302–17307 (2009).
Rice, E. L. & Pancholy, S. K. Inhibition of nitrification by climax ecosystems. Am. J. Bot. 59, 1033–1040 (1972).
Britto, D. T. & Kronzucker, H. J. Ecological significance and complexity of N-source preference in plants. Ann. Bot. 112, 957–963 (2013).
Lata, J. C. et al. Grass populations control nitrification in savanna soils. Funct. Ecol. 18, 605–611 (2004).
Boudsocq, S., Lata, J. C., Mathieu, J., Abbadie, L. & Barot, S. Modelling approach to analyse the effects of nitrification inhibition on primary production. Funct. Ecol. 23, 220–230 (2009).
Sylvester-Bradley, R., Mosquera, D. & Mendez, J. E. Inhibition of nitrate accumulation in tropical grassland soils – effect of nitrogen-fertilization and soil disturbance. J. Soil Sci. 39, 407–416 (1988).
Ishikawa, T., Subbarao, G. V., Ito, O. & Okada, K. Suppression of nitrification and nitrous oxide emission by the tropical grass Brachiaria humidicola. Plant Soil 255, 413–419 (2003).
Gopalakrishnan, S. et al. Nitrification inhibitors from the root tissues of Brachiaria humidicola, a tropical grass. J. Agri. Food Chem. 55, 1385–1388 (2007).
Iizumi, T., Mizumoto, M. & Nakamura, K. A bioluminescence assay using Nitrosomonas europaea for rapid and sensitive detection of nitrification inhibitors. Appl. Environ. Microbiol. 64, 3656–3662 (1998).
Nardi, P., Akutsu, M., Pariasca-Tanaka, J. & Wissuwa, M. Effect of methyl 3-4-hydroxyphenyl propionate, a sorghum root exudate, on N dynamic, potential nitrification activity and abundance of ammonia-oxidizing bacteria and archaea. Plant Soil 367, 627–637 (2013).
Subbarao, G. V. et al. Biological nitrification inhibition (BNI) activity in sorghum and its characterization. Plant Soil 366, 243–259 (2013).
Zakir, H. et al. Detection, isolation and characterization of a root-exuded compound, methyl 3-(4-hydroxyphenyl) propionate, responsible for biological nitrification inhibition by sorghum (Sorghum bicolor). New Phytol. 180, 442–451 (2008).
Subbarao, G. V. et al. Biological nitrification inhibition (BNI) – is it a widespread phenomenon? Plant Soil 294, 5–18 (2007).
de Boer, A. H. & de Vries- van Leeuwen, I. J. Fusicoccanes: diterpenes with surprising biological functions. Trends Plant Sci. 17, 360–368 (2012).
Sun, L., Lu, Y. F., Yu, F. W., Kronzucker, H. J. & Shi, W. M. Biological nitrification inhibition by rice root exudates and its relationship with nitrogen-use efficiency. New Phytol. 212, 646–656 (2016).
Subbarao, G. V. et al. Can biological nitrification inhibition (BNI) genes from perennial Leymus racemosus (Triticeae) combat nitrification in wheat farming? Plant Soil 299, 55–64 (2007).
Subbarao, G. V. et al. Biological nitrification inhibition (BNI) – is there potential for genetic interventions in the Triticeae? Breed. Sci. 59, 529–545 (2009).
O'sullivan, C. A., Fillery, I. R. P., Roper, M. M. & Richards, R. A. Identification of several wheat landraces with biological nitrification inhibition capacity. Plant Soil 404, 61–74 (2016).
Tanaka, J. P., Nardi, P. & Wissuwa, M. Nitrification inhibition activity, a novel trait in root exudates of rice. AoB Plants 2010, plq014 (2010).
White, C. S. Nitrification inhibition by monoterpenoids – theoretical mode of action based on molecular structures. Ecology 69, 1631–1633 (1988).
McConn, M. & Browse, J. The critical requirement for linolenic acid is pollen development, not photosynthesis, in an Arabidopsis mutant. Plant Cell 8, 403–416 (1996).
Dayan, F. E. et al. Sorgoleone. Phytochem. 71, 1032–1039 (2010).
Kodama, O., Miyakawa, J., Akatsuka, T. & Kiyosawa, S. Sakuranetin, a flavanone phytoalexin from ultraviolet-irradiated rice leaves. Phytochem. 31, 3807–3809 (1992).
Liu, Y. et al. The nitrification inhibitor methyl 3-(4-hydroxyphenyl) propionate modulates root development by interfering with auxin signaling via the NO/ROS pathway. Plant Physiol. 171, 1686–1703 (2016).
McCarty, G. W. Modes of action of nitrification inhibitors. Biol. Fert. Soils 29, 1–9 (1999).
Subbarao, G. V., Wang, H. Y., Ito, O., Nakahara, K. & Berry, W. L. NH4+ triggers the synthesis and release of biological nitrification inhibition compounds in Brachiaria humidicola roots. Plant Soil 290, 245–257 (2007).
Zeng, H. Q., Di, T. J., Zhu, Y. Y. & Subbarao, G. V. Transcriptional response of plasma membrane H+-ATPase genes to ammonium nutrition and its functional link to the release of biological nitrification inhibitors from sorghum roots. Plant Soil 398, 301–312 (2016).
Zhu, Y. Y., Zeng, H. Q., Shen, Q. R., Ishikawa, T. & Subbarao, G. V. Interplay among NH4+ uptake, rhizosphere pH and plasma membrane H+-ATPase determine the release of BNIs in sorghum roots - possible mechanisms and underlying hypothesis. Plant Soil 358, 125–135 (2012).
Lima, J. E., Kojima, S., Takahashi, H. & von Wiren, N. Ammonium triggers lateral root branching in Arabidopsis in an AMMONIUM TRANSPORTER1;3-dependent manner. Plant Cell 22, 3621–3633 (2010).
Badri, D. V. & Vivanco, J. M. Regulation and function of root exudates. Plant Cell Environ. 32, 666–681 (2009).
Weston, L. A., Ryan, P. R. & Watt, M. Mechanisms for cellular transport and release of allelochemicals from plant roots into the rhizosphere. J. Exp. Bot. 63, 3445–3454 (2012).
Wang, M. Y., Glass, A. D. M., Shaff, J. E. & Kochian, L. V. Ammonium uptake by rice roots (III. Electrophysiology). Plant Physiol. 104, 899–906 (1994).
Britto, D. T., Siddiqi, M. Y., Glass, A. D. M. & Kronzucker, H. J. Futile transmembrane NH4+ cycling: a cellular hypothesis to explain ammonium toxicity in plants. Proc. Natl Acad. Sci. USA 98, 4255–4258 (2001).
Subbarao, G. V. et al. Suppression of soil nitrification by plants. Plant Sci. 233, 155–164 (2015).
Marre, E. Fusicoccin: a tool in plant physiology. Ann. Rev. Plant Physiol. Plant Molec. Biol. 30, 273–288 (1979).
Ullrich-Eberius, C. I., Sanz, A. & Novacky, A. J. Evaluation of arsenate-associated and vanadate-associated changes of electrical membrane potential and phosphate transport in Lemna gibba G1. J. Exp. Bot. 40, 119–128 (1989).
Cesco, S., Neumann, G., Tomasi, N., Pinton, R. & Weisskopf, L. Release of plant-borne flavonoids into the rhizosphere and their role in plant nutrition. Plant Soil 329, 1–25 (2010).
Rea, P. A. Plant ATP-binding cassette transporters. Ann. Rev. Plant Biol. 58, 347–375 (2007).
Ryan, P. R., Delhaize, E. & Jones, D. L. Function and mechanism of organic anion exudation from plant roots. Ann. Rev. Plant Physiol. Plant Molec. Biol. 52, 527–560 (2001).
Kochian, L. V., Pineros, M. A., Liu, J. P. & Magalhaes, J. V. Plant adaptation to acid soils: the molecular basis for crop aluminum resistance. Ann. Rev. Plant Biol. 66, 571–598 (2015).
Bashir, K. et al. Rice phenolics efflux transporter 2 (PEZ2) plays an important role in solubilizing apoplasmic iron. Soil Sci. Plant Nutr. 57, 803–812 (2011).
Walker, T. S., Bais, H. P., Grotewold, E. & Vivanco, J. M. Root exudation and rhizosphere biology. Plant Physiol. 132, 44–51 (2003).
Bertin, C., Yang, X. H. & Weston, L. A. The role of root exudates and allelochemicals in the rhizosphere. Plant Soil 256, 67–83 (2003).
Czarnota, M. A., Paul, R. N., Weston, L. A. & Duke, S. O. Anatomy of sorgoleone-secreting root hairs of Sorghum species. Int. J. Plant Sci. 164, 861–866 (2003).
Moreta, D. E. et al. Biological nitrification inhibition (BNI) in Brachiaria pastures: a novel strategy to improve eco-efficiency of crop-livestock systems and to mitigate climate change. Trop. Grasslands 2, 88–91 (2014).
Palmgren, M. G. et al. Are we ready for back-to-nature crop breeding? Trends Plant Sci. 20, 155–164 (2015).
Oldroyd, G. E. D. & Dixon, R. Biotechnological solutions to the nitrogen problem. Curr. Opin. Biotech. 26, 19–24 (2014).
Ncube, B., Dimes, J. P., Twomlow, S. J., Mupangwa, W. & Giller, K. E. Raising the productivity of smallholder farms under semi-arid conditions by use of small doses of manure and nitrogen: a case of participatory research. Nutr. Cycl. Agroecosys. 77, 53–67 (2007).
Vitousek, P. M. et al. Nutrient imbalances in agricultural development. Science 324, 1519–1520 (2009).
Subbarao, G. V. et al. A paradigm shift towards low-nitrifying production systems: the role of biological nitrification inhibition (BNI). Ann. Bot. 112, 297–316 (2013).
Bloom, A. J. et al. CO2 enrichment inhibits shoot nitrate assimilation in C3 but not C4 plants and slows growth under nitrate in C3 plants. Ecology 93, 355–367 (2012).
Britto, D. T. & Kronzucker, H. J. NH4+ toxicity in higher plants: a critical review. J. Plant Physiol. 159, 567–584 (2002).
Britto, D. T. et al. Potassium and nitrogen poising: physiological changes and biomass gains in rice and barley. Can. J. Plant Sci. 94, 1085–1089 (2014).
Kirk, G. J. D. & Kronzucker, H. J. The potential for nitrification and nitrate uptake in the rhizosphere of wetland plants: a modelling study. Ann. Bot. 96, 639–646 (2005).
Kronzucker, H. J., Siddiqi, M. Y., Glass, A. D. M. & Kirk, G. J. D. Nitrate-ammonium synergism in rice: a subcellular flux analysis. Plant Physiol. 119, 1041–1045 (1999).
Qiao, C. L. et al. How inhibiting nitrification affects nitrogen cycle and reduces environmental impacts of anthropogenic nitrogen input. Glob. Change Biol. 21, 1249–1257 (2015).
Lam, S. K., Suter, H., Mosier, A. R. & Chen, D. Using nitrification inhibitors to mitigate agricultural N2O emission: a double-edged sword? Glob. Change Biol. 23, 485–489 (2016).
Jones, D. L., Hodge, A. & Kuzyakov, Y. Plant and mycorrhizal regulation of rhizodeposition. New Phytol. 163, 459–480 (2004).
Marschner, H. Marschner's Mineral Nutrition of Higher Plants (Academic, 2011)
Winogradsky, S. The method in soil microbiology as illustrated by studies on Azotobacter and the nitrifying organisms. Soil Sci. 40, 59–76 (1935).
Frijlink, M. J., Abee, T., Laanbroek, H. J., Deboer, W. & Konings, W. N. The bioenergetics of ammonia and hydroxylamine oxidation in Nitrosomonas europaea at acid and alkaline pH. Arch. Microbiol. 157, 194–199 (1992).
Tarre, S., Shlafman, E., Beliavski, M. & Green, M. Changes in ammonia oxidiser population during transition to low pH in a biofilm reactor starting with Nitrosomonas europaea. Water Sci. Tech. 55, 363–368 (2007).
Bardon, C. et al. Evidence for biological denitrification inhibition (BDI) by plant secondary metabolites. New Phytol. 204, 620–630 (2014).
Li, Y. L., Kronzucker, H. J. & Shi, W. M. Microprofiling of nitrogen patches in paddy soil: analysis of spatiotemporal nutrient heterogeneity at the microscale. Sci. Rep. 6, 27064 (2016).
Dinsdale, E. A. et al. Functional metagenomic profiling of nine biomes. Nature 452, 629–632 (2008).
Oita, A. et al. Substantial nitrogen pollution embedded in international trade. Nat. Geosci. 9, 111–115 (2016).
Andrews, M. & Lea, P. J. Our nitrogen ‘footprint’: the need for increased crop nitrogen use efficiency. Ann. Appl. Biol. 163, 165–169 (2013).
Tesfamariam, T. et al. Biological nitrification inhibition in sorghum: the role of sorgoleone production. Plant Soil 379, 325–335 (2014).
Subbarao, G. V. et al. Free fatty acids from the pasture grass Brachiaria humidicola and one of their methyl esters as inhibitors of nitrification. Plant Soil 313, 89–99 (2008).
Ladha, J. K. et al. Global nitrogen budgets in cereals: a 50-year assessment for maize, rice, and wheat production systems. Sci. Rep. 6, 19355 (2016).
Cassman, K. G., Dobermann, A., Walters, D. T. & Yang, H. Meeting cereal demand while protecting natural resources and improving environmental quality. Ann. Rev. Environ. Res. 28, 315–358 (2003).
Bouwman, A. F. et al. A global high-resolution emission inventory for ammonia. Glob. Biogeochem. Cycl. 11, 561–587 (1997).
Sommer, S. G., Schjoerring, J. K. & Denmead, O. T. Ammonia emission from mineral fertilizers and fertilized crops. Adv. Agron. 82, 557–622 (2004).
Cai, G. X. et al. Nitrogen losses from fertilizers applied to maize, wheat and rice in the North China Plain. Nutr. Cycl. Agroecosys. 63, 187–195 (2002).
Zhang, X. L. et al. In situ nitrogen mineralization, nitrification, and ammonia volatilization in maize field fertilized with urea in Huanghuaihai region of northern China. PLoS ONE 10, e0115649 (2015).
Cai, Z. C. et al. Methane and nitrous oxide emissions from rice paddy fields as affected by nitrogen fertilisers and water management. Plant Soil 196, 7–14 (1997).
Ding, W., Cai, Y., Cai, Z., Yagi, K. & Zheng, X. Nitrous oxide emissions from an intensively cultivated maize-wheat rotation soil in the North China Plain. Sci. Tot. Environ. 373, 501–511 (2007).
Zhang, Y. Y. et al. Emissions of nitrous oxide, nitrogen oxides and ammonia from a maize field in the North China Plain. Atmos. Environ. 45, 2956–2961 (2011).
Chowdary, V. M., Rao, N. H. & Sarma, P. B. S. A coupled soil water and nitrogen balance model for flooded rice fields in India. Agri. Ecosys. Environ. 103, 425–441 (2004).
Ghosh, B. C. & Bhat, R. Environmental hazards of nitrogen loading in wetland rice fields. Environ. Poll. 102, 123–126 (1998).
Tian, Y. H., Yin, B., Yang, L. Z., Yin, S. X. & Zhu, Z. L. Nitrogen runoff and leaching losses during rice-wheat rotations in Taihu Lake Region, China. Pedosphere 17, 445–456 (2007).
Acknowledgements
The authors would like to thank the Natural Sciences and Engineering Research Council of Canada (NSERC), the Strategic Priority Research Program (B)—‘Soil-microbial system function and regulation’ of the Chinese Academy of Sciences, and the National Natural Science Foundation of China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Coskun, D., Britto, D., Shi, W. et al. Nitrogen transformations in modern agriculture and the role of biological nitrification inhibition. Nature Plants 3, 17074 (2017). https://doi.org/10.1038/nplants.2017.74
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nplants.2017.74
This article is cited by
-
Intermediate soil acidification induces highest nitrous oxide emissions
Nature Communications (2024)
-
High nitrogen fertilizer input enhanced the microbial network complexity in the paddy soil
Soil Ecology Letters (2024)
-
Harnessing plant–microbe interactions to promote nitrogen use efficiency in cereal crops
Plant and Soil (2024)
-
Nitrogen absorption and mineralization change with cover crop types and greenhouse vegetable planting years in fluvo-aquic soil
Journal of Soils and Sediments (2024)
-
Greenhouse gas emissions from nitrogen fertilizers could be reduced by up to one-fifth of current levels by 2050 with combined interventions
Nature Food (2023)