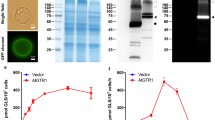
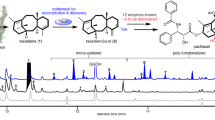
Abstract
Plants sequester intermediates of metabolic pathways into different cellular compartments, but the mechanisms by which these molecules are transported remain poorly understood. Monoterpene indole alkaloids, a class of specialized metabolites that includes the anticancer agent vincristine, antimalarial quinine and neurotoxin strychnine, are synthesized in several different cellular locations. However, the transporters that control the movement of these biosynthetic intermediates within cellular compartments have not been discovered. Here we present the discovery of a tonoplast localized nitrate/peptide family (NPF) transporter from Catharanthus roseus, CrNPF2.9, that exports strictosidine, the central intermediate of this pathway, into the cytosol from the vacuole. This discovery highlights the role that intracellular localization plays in specialized metabolism, and sets the stage for understanding and controlling the central branch point of this pharmacologically important group of compounds.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
O’Connor, S. E. & Maresh, J. J. Chemistry and biology of monoterpene indole alkaloid biosynthesis. Nat. Prod. Rep. 23, 532–547 (2006).
Keglevich, P., Hazai, L., Kalaus, G. & Szantay, C. Modifications on the basic skeletons of vinblastine and vincristine. Molecules 17, 5893–5914 (2012).
Courdavault, V. et al. A look inside an alkaloid multisite plant: the Catharanthus logistics. Curr. Opin. Plant. Biol. 19, 43–50 (2014).
McKnight, T. D., Roessner, C. A., Devagupta, R., Scott, A. I. & Nessler, C. L. Nucleotide sequence of a cDNA encoding the vacuolar protein strictosidine synthase from Catharanthus roseus. Nucleic Acids Res. 18, 4939–4939 (1990).
Guirimand, G. et al. Strictosidine activation in Apocynaceae: towards a “nuclear time bomb”? BMC Plant Biol. 10, 182 (2010).
Gongora-Castillo, E. et al. Development of transcriptomic resources for interrogating the biosynthesis of monoterpene indole alkaloids in medicinal plant species. PLoS ONE 7, e52506 (2012).
Geu-Flores, F. et al. An alternative route to cyclic terpenes by reductive cyclization in iridoid biosynthesis. Nature 492, 138–142 (2012).
Dunham, I. et al. An integrated encyclopedia of DNA elements in the human genome. Nature 489, 57–74 (2012).
Kim, D. H., Grun, D. & van Oudenaarden, A. Dampening of expression oscillations by synchronous regulation of a microRNA and its target. Nat. Genet. 45, 1337–1344 (2013).
Leran, S. et al. A unified nomenclature of NITRATE TRANSPORTER 1/PEPTIDE TRANSPORTER family members in plants. Trends Plant Sci. 19, 5–9 (2014).
Padmanabhan, M. & Dinesh-Kumar, S. P. Virus-induced gene silencing as a tool for delivery of dsRNA into plants. Cold Spring Harb. Protoc. http://dx.doi.org/10.1101/pdb.prot5139 (2009).
Gomez, C. et al. Grapevine MATE-type proteins act as vacuolar H+-dependent acylated anthocyanin transporters. Plant Physiol. 150, 402–415 (2009).
Larisch, N., Schulze, C., Galione, A. & Dietrich, P. An N-terminal dileucine motif directs two-pore channels to the tonoplast of plant cells. Traffic 13, 1012–1022 (2012).
Latz, A. et al. TPK1, a Ca2+-regulated Arabidopsis vacuole two-pore K+ channel is activated by 14-3-3 proteins. Plant J. 52, 449–459 (2007).
Marinova, K. et al. The Arabidopsis MATE transporter TT12 acts as a vacuolar flavonoid/H+-antiporter active in proanthocyanidin-accumulating cells of the seed coat. Plant Cell 19, 2023–2038 (2007).
Nelson, B. K., Cai, X. & Nebenführ, A. A multicolored set of in vivo organelle markers for co-localization studies in Arabidopsis and other plants. Plant J. 51, 1126–1136 (2007).
Komarova, N. Y., Meier, S., Meier, A., Grotemeyer, M. S. & Rentsch, D. Determinants for Arabidopsis peptide transporter targeting to the tonoplast or plasma membrane. Traffic 13, 1090–1105 (2012).
Yamada, K. et al. Functional analysis of an Arabidopsis thaliana abiotic stress-inducible facilitated diffusion transporter for monosaccharides. J. Biol. Chem. 285, 1138–1146 (2010).
Wolfenstetter, S., Wirsching, P., Dotzauer, D., Schneider, S. & Sauer, N. Routes to the tonoplast: the sorting of tonoplast transporters in Arabidopsis mesophyll protoplasts. Plant Cell 24, 215–232 (2012).
Carter, C. et al. The vegetative vacuole proteorne of Arabidopsis thaliana reveals predicted and unexpected proteins. Plant Cell 16, 3285–3303 (2004).
Gomes, D. et al. Aquaporins are multifunctional water and solute transporters highly divergent in living organisms. Biochim. Biophys. Acta 1788, 1213–1228 (2009).
Asada, K. et al. A 7-deoxyloganetic acid glucosyltransferase contributes a key step in secologanin biosynthesis in Madagascar Periwinkle. Plant Cell 25, 4123–4134 (2013).
Miettinen, K. et al. The seco-iridoid pathway from Catharanthus roseus. Nat. Commun. 5, 3606 (2014).
Salim, V., Wiens, B., Masada-Atsumi, S., Yu, F. & De Luca, V. 7-Deoxyloganetic acid synthase catalyzes a key 3 step oxidation to form 7-deoxyloganetic acid in Catharanthus roseus iridoid biosynthesis. Phytochemistry 101, 23–31 (2014).
Salim, V., Yu, F., Altarejos, J. & De Luca, V. Virus-induced gene silencing identifies Catharanthus roseus 7-deoxyloganic acid-7-hydroxylase, a step in iridoid and monoterpene indole alkaloid biosynthesis. Plant J. 76, 754–765 (2013).
Yu, F. & De Luca, V. ATP-binding cassette transporter controls leaf surface secretion of anticancer drug components in Catharanthus roseus. Proc. Natl Acad. Sci. USA 110, 15830–15835 (2013).
Carqueijeiro, I., Noronha, H., Duarte, P., Geros, H. & Sottomayor, M. Vacuolar transport of the medicinal alkaloids from Catharanthus roseus is mediated by a proton-driven antiport. Plant Physiol. 162, 1486–1496 (2013).
Nour-Eldin, H. H. et al. NRT/PTR transporters are essential for translocation of glucosinolate defence compounds to seeds. Nature 488, 531–534 (2012).
Krouk, G. et al. Nitrate-regulated auxin transport by NRT1.1 defines a mechanism for nutrient sensing in plants. Dev. Cell 18, 927–937 (2010).
Kanno, Y. et al. Identification of an abscisic acid transporter by functional screening using the receptor complex as a sensor. Proc. Natl Acad. Sci. USA 109, 9653–9658 (2012).
Tal, I. et al. The Arabidopsis NPF3 protein is a GA transporter. Nat. Commun. 7, 11 (2016).
Weichert, A. et al. AtPTR4 and AtPTR6 are differentially expressed, tonoplast-localized members of the peptide transporter/nitrate transporter 1 (PTR/NRT1) family. Planta 235, 311–323 (2012).
Newstead, S. et al. Crystal structure of a prokaryotic homologue of the mammalian oligopeptide-proton symporters, PepT1 and PepT2. EMBO J. 30, 417–426 (2011).
Jorgensen, M. E. et al. A functional EXXEK motif is essential for proton coupling and active glucosinolate transport by NPF2.11. Plant Cell Physiol. 56, 2340–2350 (2015).
Parker, J. L. & Newstead, S. Molecular basis of nitrate uptake by the plant nitrate transporter NRT1.1. Nature 507, 68–72 (2014).
Morita, M. et al. Vacuolar transport of nicotine is mediated by a multidrug and toxic compound extrusion (MATE) transporter in Nicotiana tabacum. Proc. Natl Acad. Sci. USA 106, 2447–2452 (2009).
Shitan, N. et al. Involvement of the leaf-specific multidrug and toxic compound extrusion (MATE) transporter Nt-JAT2 in vacuolar sequestration of nicotine in Nicotiana tabacum. PLoS ONE 9, e108789 (2014).
Shoji, T. et al. Multidrug and toxic compound extrusion-type transporters implicated in vacuolar sequestration of nicotine in tobacco roots. Plant Physiol. 149, 708–718 (2009).
Hildreth, S. B. et al. Tobacco nicotine uptake permease (NUP1) affects alkaloid metabolism. Proc. Natl Acad. Sci. USA 108, 18179–18184 (2011).
Brown, S., Clastre, M., Courdavault, V. & O’ Connor, S. E. De novo production of the plant-derived alkaloid strictosidine in yeast. Proc. Natl Acad. Sci. USA 112, 3205–3210 (2015).
Galanie, S., Thodey, K., Trenchard, I. J., Interrante, M. F. & Smolke, C. D. Complete biosynthesis of opioids in yeast. Science 349, 1095–1100 (2015).
Wehrens, R. & Buydens, L. M. C. Self- and super-organizing maps in R: the kohonen package. J. Stat. Softw. 21, 1–19 (2007).
Vesanto, J. & Alhoniemi, E. Clustering of the self-organizing map. IEEE Trans. Neural Netw. 11, 586–600 (2000).
Geu-Flores, F., Nour-Eldin, H. H., Nielsen, M. T. & Halkier, B. A. USER fusion: a rapid and efficient method for simultaneous fusion and cloning of multiple PCR products. Nucleic Acids Res. 35, e55 (2007).
Nour-Eldin, H. H., Geu-Flores, F. & Halkier, B. A. USER cloning and USER fusion: the ideal cloning techniques for small and big laboratories. Methods Mol. Biol. 643, 185–200 (2010).
Liscombe, D. K. & O’ Connor, S. E. A virus-induced gene silencing approach to understanding alkaloid metabolism in Catharanthus roseus. Phytochemistry 72, 1969–1977 (2011).
Murata, J., Roepke, J., Gordon, H. & De Luca, V. The leaf epidermome of Catharanthus roseus reveals its biochemical specialization. Plant Cell 20, 524–542 (2008).
McCoy, E., Galan, M. C. & O'Connor, S. E. Substrate specificity of strictosidine synthase. Bioorg. Med. Chem. Lett. 16, 2475–2478 (2006).
Guirimand, G. et al. Optimization of the transient transformation of Catharanthus roseus cells by particle bombardment and its application to the subcellular localization of hydroxymethylbutenyl 4-diphosphate synthase and geraniol 10-hydroxylase. Plant Cell Rep. 28, 1215–1234 (2009).
Nour-Eldin, H. H., Hansen, B. G., Norholm, M. H. H., Jensen, J. K. & Halkier, B. A. Advancing uracil-excision based cloning towards an ideal technique for cloning PCR fragments. Nucleic Acids Res. 34, 8 (2006).
Acknowledgements
This work was supported by grants from the European Research Council (311363), BBSRC (BB/J004561/1) (S.E.O.), Danish National Research Foundation (DNRF99 for D.X., V.N., M.B., H.H.N., B.A.H. and 10-082858 for F.G.-F.), the Innovation Fund Denmark (j.nr. 76-2014-3) (H.H.N.) and from the Région Centre, France (ABISAL grant; E.F., A.O., T.D.D.B., V.C.). R.M.E.P. was supported by a BBSRC Studentship.
Author information
Authors and Affiliations
Contributions
R.M.E.P. made the initial discovery of CrNPF2.9 and designed and carried out all silencing experiments; D.X. and H.H.N-E designed and carried out kinetic experiments; E.F., M.I.S.T.C., T.D.B. and V.C. designed and carried out localization experiments; F.G-F., S.E.O., H.H.N-E, V.C. and B.A.H. contributed to the conception and design of the experiments; R.M.E.P., D.X., E.F., M.I.S.T.C., A.O., T.D.B, V.N., M.B., C-E.O, D.M.J., E.C.T. and A.P. contributed to the acquisition of data; R.M.E.P., D.X., E.F., M.I.S.T.C., F.G-F., V.C., H.H.N-E. and S.E.O. contributed to data analysis and interpretation; R.M.E.P. and S.E.O. drafted the manuscript and all authors critically revised and approved the final version of the manuscript for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1–5, Supplementary Tables 1–4. (PDF 6463 kb)
Rights and permissions
About this article
Cite this article
Payne, R., Xu, D., Foureau, E. et al. An NPF transporter exports a central monoterpene indole alkaloid intermediate from the vacuole. Nature Plants 3, 16208 (2017). https://doi.org/10.1038/nplants.2016.208
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nplants.2016.208
This article is cited by
-
A Cotyledon-based Virus-Induced Gene Silencing (Cotyledon-VIGS) approach to study specialized metabolism in medicinal plants
Plant Methods (2024)
-
Regulation of T16H subcellular localization for promoting its catalytic efficiency in yeast cells
Biotechnology Letters (2024)
-
Export of defensive glucosinolates is key for their accumulation in seeds
Nature (2023)
-
Single-cell multi-omics in the medicinal plant Catharanthus roseus
Nature Chemical Biology (2023)
-
Study of vacuole glycerate transporter NPF8.4 reveals a new role of photorespiration in C/N balance
Nature Plants (2023)