Abstract
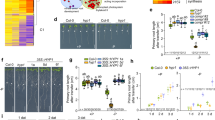
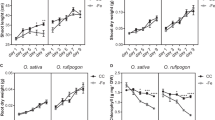
Manganese is an essential metal for plant growth. A number of transporters involved in the uptake of manganese from soils, and its translocation to the shoot, have been identified in Arabidopsis and rice. However, the transporter responsible for the radial transport of manganese out of root exodermis and endodermis cells and into the root stele remains unknown. Here, we show that metal tolerance protein 9 (MTP9), a member of the cation diffusion facilitator family, is a critical player in this process in rice (Oryza sativa). We find that MTP9 is mainly expressed in roots, and that the resulting protein is localized to the plasma membrane of exo- and endodermis cells, at the proximal side of these cell layers (opposite the manganese uptake transporter Nramp5, which is found at the distal side). We demonstrate that MTP9 has manganese transport activity by expression in proteoliposomes and yeast, and show that knockout of MTP9 in rice reduces manganese uptake and its translocation to shoots. We conclude that at least in rice MTP9 is required for manganese translocation to the root stele, and thereby manganese uptake.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Broadley, M., Brown, P., Cakmak, I., Rengel, Z. & Zhao, F. in Marschner's Mineral Nutrition of Higher Plants 3rd edn (ed. Marschner, P. ) 191–248 (Academic Press, 2012).
Pittman, J. K. Managing the manganese molecular mechanisms of manganese transport and homeostasis. New Phytol. 167, 733–742 (2005).
Williams, L. E. & Pittman, J. K. in Cell Biology of Metals and Nutrients (eds Hell, R. & Mendal, R. R. ) 95–117 (Plant Cell Monographs 17: Dissecting pathways involved in manganese homeostasis and stress in higher plant cells, Springer, 2010).
Mengel, K. & Kirkby, E. A. Principles of Plant Nutrition (International Potash Institute, 1987).
Cailliatte, R., Schikora, A., Briat, J. F., Mari, S. & Curie, C. High-affinity manganese uptake by the metal transporter NRAMP1 is essential for Arabidopsis growth in low manganese conditions. Plant Cell 22, 904–917 (2010).
Ishimaru, Y. et al. Characterizing the role of rice NRAMP5 in manganese, iron and cadmium transport. Sci. Rep. 2, 286 (2012).
Sasaki, A., Yamaji, N., Yokosho, K. & Ma, J. F. Nramp5 is a major transporter responsible for manganese and cadmium uptake in rice. Plant Cell 24, 2155–2167 (2012).
Milner, M. J., Seamon, J., Craft, E. & Kochian, L. V. Transport properties of members of the ZIP family in plants and their role in Zn and manganese homeostasis. J. Exp. Bot. 64, 369–381 (2013).
Yamaji, N., Sasaki, A., Xia, J. X., Yokosho, K. & Ma, J. F. A node-based switch for preferential distribution of manganese in rice. Nature Commun. 4, 2442 (2013).
Koike, S. et al. OsYSL2 is a rice metal-nicotianamine transporter that is regulated by iron and expressed in the phloem. Plant J. 39, 415–424 (2004).
Ishimaru, Y. et al. Rice metal-nicotianamine transporter, OsYSL2, is required for the long-distance transport of iron and manganese. Plant J. 62, 379–390 (2010).
Paulsen, I. T. & Saier, M. H. Jr A novel family of ubiquitous heavy metal ion transport proteins. J. Membr. Biol. 156, 99–103 (1997).
Eide, D. J. The molecular biology of metal ion transport in Saccharomyces cerevisiae. Ann. Rev. Nutr. 18, 441–469 (1998).
Van der Zaal, B. J. et al. Overexpression of a novel Arabidopsis gene related to putative zinc-transporter genes from animals can lead to enhanced zinc resistance and accumulation. Plant Physiol. 119, 1047–1056 (1999).
Montanini, B., Blaudez, D., Jeandroz, S., Sanders, D. & Chalot, M. Phylogenetic and functional analysis of the cation diffusion facilitator (CDF) family: improved signature and prediction of substrate specificity. BMC Genomics 8, 107 (2007).
Delhaize, E., Kataoka, T., Hebb, D. M., White, R. G. & Ryan, P. R. Genes encoding proteins of the cation diffusion facilitator family that confer manganese tolerance. Plant Cell 15, 1131–1142 (2003).
Peiter, E. et al. A secretory pathway-localized cation diffusion facilitator confers plant manganese tolerance. Proc. Natl Acad. Sci. USA 104, 8532–8537 (2007).
Delhaize, E. et al. A role for the AtMTP11 gene of Arabidopsis in manganese transport and tolerance. Plant J. 51, 198–210 (2007).
Chen, Z. et al. Manganese tolerance in rice is mediated by MTP8.1, a member of the cation diffusion facilitator family. J. Exp. Bot. 64, 4375–4387 (2013).
Gustin, J. L., Zanis, M. J., & Salt, D. E. Structure and evolution of the plant cation diffusion facilitator family of ion transporters. BMC Evol. Biol. 11, 76 (2011).
Zheng, L., Yamaji, N., Yokosho, K. & Ma, J. F. YSL16 is a phloem-localized transporter of the copper-nicotianamine complex that is responsible for copper distribution in rice. Plant Cell 24, 3767–3782 (2012).
Ishimaru, Y. et al. Rice plants take up iron as an Fe3+-phytosiderophore and as Fe2+. Plant J. 45, 335–346 (2006).
Yamaji, N. & Ma, J. F. Further characterization of a rice Si efflux transporter, Lsi2. Soil. Sci. Plant Nutr. 57, 259–264 (2011).
Yamaji, N. & Ma, J. F. Spatial distribution and temporal variation of the rice silicon transporter Lsi1. Plant Physiol. 143, 1306–1313 (2007).
Sakurai, G. et al. In silico simulation modeling reveals the importance of the Casparian strip for efficient silicon uptake in rice roots. Plant Cell Physiol. 56, 631–639 (2015).
Ma, J. F. et al. A silicon transporter in rice. Nature 440, 688–691 (2006).
Ma, J. F. et al. An efflux transporter of silicon in rice. Nature 448, 209–212 (2007).
Vlamis, J. & Williams, D. E. Manganese and silicon interaction in the gramineae. Plant Soil 27, 131–140 (1967).
Takano, J., Miwa, K., Yuan, L., von Wiren, N. & Fujiwara, T. Polar localization and degradation of Arabidopsis boron transporters through distinct trafficking pathways. Proc. Natl Acad. Sci. USA 107, 5220–5225 (2010).
Ueno, D., Yamaji, N. & Ma, J. F. Further characterization of ferric-phytosiderophore transporters ZmYS1 and HvYS1 in maize and barley. J. Exp. Bot. 60, 3513–3520 (2009).
Murata, Y. et al. A specific transporter for iron(III)-phytosiderophore in barley roots. Plant J. 46, 563–572 (2006).
Rosch, J. W., Gao, G., Ridout, G., Wang, Y. D. & Tuomanen, E. I. Role of the manganese efflux system mntE for signalling and pathogenesis in Streptococcus pneumonia. Mol. Microbiol. 72, 12–25 (2009).
Chen, S. et al. A highly efficient transient protoplast system for analyzing defence gene expression and protein-protein interactions in rice. Mol. Plant. Pathol. 7, 417–427 (2006).
Miyaji, T. et al. AtPHT4;4 is a chloroplast-localized ascorbate transporter in Arabidopsis. Nature Commun. 6, 5928 (2015).
Acknowledgements
This work was supported by a Grant-in-Aid for Scientific Research on Innovative Areas from the Ministry of Education, Culture, Sports, Science and Technology of Japan (No. 23119515 to D.U., No. 22119002 to J.F.M.), and by The Kurata Memorial Hitachi Science and Technology Foundation (No. 1044 to D.U.). We thank Kato, S. Yasutake, D. Rikiishi, A. Morita, T. Haruna and K. Ishii for their technical assistance.
Author information
Authors and Affiliations
Contributions
D.U., N.Y. and J.F.M. designed the research. D.U., A.S. and N.Y. equally contributed to the research. D.U., A.S., N.Y., Y.F., Y.T., K.I., J.C., T.M., S.M., Y.M. and J.F.M. performed the experiments, analysed data and wrote the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Ueno, D., Sasaki, A., Yamaji, N. et al. A polarly localized transporter for efficient manganese uptake in rice. Nature Plants 1, 15170 (2015). https://doi.org/10.1038/nplants.2015.170
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nplants.2015.170
This article is cited by
-
Deciphering the functional roles of transporter proteins in subcellular metal transportation of plants
Planta (2023)
-
The Implication of Manganese Surplus on Plant Cell Homeostasis: A Review
Journal of Plant Growth Regulation (2023)
-
TaNRAMP3 is essential for manganese transport in Triticum aestivum
Stress Biology (2023)
-
Metal tolerance gene family in barley: an in silico comprehensive analysis
Journal of Applied Genetics (2023)
-
Application of oyster shell powder reduces cadmium accumulation by inhibiting the expression of genes responsible for cadmium uptake and translocation in rice
Environmental Science and Pollution Research (2023)