Abstract
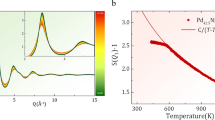
The glass transition is usually understood as a structural arrest that occurs during the cooling of liquids, trapping the system before it can crystallize. It occurs for all liquid classes, including metals. Theoretical interest has focused on the dynamical heterogeneity encountered during supercooling of ‘fragile’ liquids. Many suggest that the slow-down is caused by increasing dynamical correlation lengths. Here we report kinetics and thermodynamics of arrest in a system that disorders while in its ground state, exhibits a large heat capacity change (ΔCp=Cp,mobile−Cp,arrested) on arrest, yet clearly is characterized by a static correlation length that decreases when approaching the transition temperature Tg from above. We show that our system, the Fe50Co50 superlattice, kinetically mimics an ideal ‘strong’ liquid with a critical point. Introducing liquid critical-point simulations, we can then argue that strong liquids differ from fragile liquids by occupying opposite flanks of an underlying order–disorder transition, which can be continuous, critical or weakly first order.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Angell, C. A. Formation of glasses from liquids and biopolymers. Science 267, 1924–1935 (1995).
Busch, R, Schroers, J & Wang, W. H. Thermodynamics and kinetics of bulk metallic glass. MRS Bull. 32, 620–623 (2007).
Martinez, L. M. & Angell, C. A. A thermodynamic connection to the fragility of glass-forming liquids. Nature 410, 663–667 (2001).
Wang, L-M., Richert, R. & Angell, C. A. Fragility and thermodynamics in non-polymeric glassformers. J. Chem. Phys. 125, 074506 (2006).
Richert, R. Heterogeneous dynamics in liquids: Fluctuations in space and time. J. Phys. Condens. Matter 14, R703–R738 (2002).
Ediger, M. D. Spatially heterogeneous dynamics in supercooled liquids. Ann. Rev. Phys. Chem. 51, 99–128 (2000).
Donati, C., Glotzer, S. C. & Poole, P. H. Growing spatial correlations of particle displacements in a simulated liquid on cooling toward the glass transition. Phys. Rev. Lett. 82, 5064–5067 (1999).
Qiu, X. H. & Ediger, M. D. Lengthscale of dynamic heterogeneity in supercooled d-sorbitol: Comparison to model predictions. J. Phys. Chem. 107, 459–464 (2003).
Donati, C. et al. Growing spatial correlations of particle displacements in a simulated liquid on cooling toward the glass transition. Phys. Rev. Lett. 82, 5064–5067 (1999).
Reinsberg, S. A., Qui, X. H., Wilhelm, M., Spiess, H. W. & Ediger, M. D. Length scale of dynamic heterogeneity in supercooled glycerol near Tg . J. Chem. Phys. 114, 7299–7302 (2001).
Berthier, L. et al. Direct experimental evidence of a growing length scale accompanying the glass transition. Science 310, 1797–2000 (2005).
Moynihan, C. T. & Schroeder, J. Non-exponential structural relaxation, anomalous light scattering and nanoscale inhomogeneities in glass forming liquids. J. Non-Cryst. Solids 160, 52–59 (1993).
Tanaka, H., Kawasaki, T., Shintani, H. & Watanabe, K. Critical-like behavior of glass-forming liquids. Nature Mater. 9, 324–331 (2010).
Angell, C. A. & Ueno, K. Soft is strong. Nature 462, 45–46 (2009).
Angell, C. A. Insights into phases of liquid water from study of its unusual glass-forming properties. Science 319, 582–587 (2008).
Scheidler, P., Kob, W., Latz, A., Horbach, J. & Binder, K. Frequency-dependent specific heat of viscous silica. Phys. Rev. B 63, 104204 (2005).
Hemmati, M., Moynihan, C. T. & Angell, C. A. Interpretation of the molten BeF2 viscosity anomaly in terms of a high temperature density maximum, and other waterlike features. J. Chem. Phys. 115, 6663–6671 (2001).
Ito, K., Moynihan, C. T. & Angell, C. A. Thermodynamic determination of fragility in liquids and a fragile-to-strong liquid transition in water. Nature 398, 492–495 (1999).
Kaya, S. & Sato, H. Superstructuring in the iron–cobalt system and their magnetic properties. Proc. PhysicoMath. Soc. Jpn 25, 261–273 (1943).
Angell, C. A. Glass formation and glass transition in supercooled liquids, with insights from study of related phenomena in crystals. J. Non-Cryst. Solids 354, 4703–4712 (2008).
Moynihan, C. T., Easteal, A. J., Debolt, M. A. & Tucker, J. Dependence of fictive temperature of glass on cooling rate. J. Amer. Ceram. Soc. 59, 12–16 (1976).
Yue, Y. Z., von der Ohe, R. & Jensen, S. L. Fictive temperature, cooling rate, and viscosity of glasses. J. Chem. Phys. 120, 8053–8059 (2000).
Xu, L-M., Buldyrev, S. V., Giovambattista, N., Angell, C. A. & Stanley, H. E. A monatomic system with a liquid–liquid critical point and two glassy states. J. Chem. Phys. 130, 054505 (2009).
Wang, L. M., Velikov, V. & Angell, C. A. Direct determination of kinetic fragility indices of glassforming liquids by differential scanning calorimetry: Kinetic versus thermodynamic fragilities. J. Chem. Phys. 117, 10184–10192 (2002).
Busch, R., Bakke, E. & Johnson, W. L. Viscosity of the supercooled liquid and relaxation at the glass transition of the Zr46.75Ti8.25Cu7.5Ni10Be27.5 bulk metallic glass forming alloy. Acta Mater. 46, 4725–4732 (1998).
Fujimori, H. & Oguni, M. Correlation index (Tga−Tgb)/Tg and activation energy ratio as parameters characterising the structure of liquid and glass. Solid State Commun. 94 (1995).
Busch, R. & Johnson, W. L. The kinetic glass transition of the Zr46.75Ti8.25Cu7.5Ni10Be27.5 bulk metallic glass former-supercooled liquids on a long timescale. Appl. Phys. Lett. 72, 2695–2697 (1998).
Girifalco, L. A. Statistical Mechanics of Solids (Oxford Univ. Press, 2000).
Lipp, M. J. et al. Thermal signatures of the Kondo volume collapse in cerium. Phys. Rev. Lett 101, 165703 (2008).
Xu, L.-M. et al. Relation between the Widom line and the dynamic crossover in systems with a liquid–liquid phase transition. Proc. Natl Acad. Sci. USA 102, 16558–16562 (2005).
Angell, C. A. Water II is a strong liquid. J. Phys. Chem. 97, 6339–6341 (1993).
Mallamace, F. et al. The fragile-to-strong dynamic crossover transition in confined water: Nuclear magnetic resonance results. J. Chem. Phys. 124, 161102 (2006).
Liu, L. et al. Pressure dependence of fragile-to-strong transition and a possible second critical point in supercooled confined water. J. Chem. Phys. 95, 117802 (2005).
Zhang, Y. et al. Dynamic susceptibility of supercooled water and its relation to the dynamic crossover phenomenon. Phys. Rev. E 79, 040201 (2009).
Bosio, L., Teixeira, J. & Stanley, H. E. Enhanced density fluctuations in water analysed by neutron scattering. Phys. Rev. Lett. 46, 597–600 (1981).
Xie, Y. et al. Noncritical behaviour of density fluctuations in supercooled water. Phys. Rev. Lett. 71, 2050–2053 (1993).
Huang, C. et al. The inhomogeneous structure of water at ambient conditions. Proc. Natl Acad. Sci. USA 106, 15214–15218 (2009).
Moore, E. B. & Molinero, V. Growing correlation length in supercooled water. J. Chem. Phys. 130, 244505 (2009).
Poole, P. H., Hemmati, M. & Angell, C. A. Comparison of thermodynamic properties of simulated liquid silica and water. Phys. Rev. Lett. 79, 2281–2284 (1997).
Kim, K. Y. Calorimetric Studies on Argon and Hexafluoro Ethane and a Generalized Correlation of Maxima in Isobaric Heat Capacity 20. (1974), (Technical report, Department of Chemical Engineering, Michigan University) http://deepblue.lib.umich.edu/handle/2027.42/6003.
Bockris, J. O., McKenzie, J. D. & Kitchener, J. A. Viscous flow in silica and binary liquid silicates. Trans. Faraday Soc. 51, 1734–1748 (1955).
Saika-Voivod, I., Poole, P. H. & Sciortino, F. Fragile-to-strong transition and polyamorphism in the energy landscape of liquid silica. Nature 412, 514–517 (2001).
Way, C., Wadhwa, P. & Busch, R. Influence of shear rate and temperature on the viscosity and fragility of the Zr41.2Ti13.8Cu12.5Ni10.0Be22.5 metallic glass-forming liquid. Acta Mater. 55, 2977–2983 (2007).
Zhang, C., Hu, L., Yue, Y-Z. & Mauro, J. C. Fragile-to-strong transition in metallic glass-forming liquids. J. Chem. Phys. 133, AN014508 (2010).
Tamura, S., Yokokawa, T. & Niwa, K. J. Enthalpy of beryllium fluoride from 456 to 1,083 K by transposed temperature drop calorimetry. J. Chem. Thermodyn. 7, 633–643 (1975).
Sakaguchi, S. & Todoroki, S-I. Raleigh scattering of silica core optical fiber after heat treatment. Appl. Opt. 37, 7708–7711 (1998).
Berthier, L. Revisiting the slow dynamics of a silica melt using Monte Carlo simulations. Phys. Rev. E 76, 011507 (2007).
Matyushov, D. & Angell, C. A. Gaussian excitations model for glassformer thermodynamics and dynamics. J. Chem. Phys. 126, AN094501 (2007).
Dawson, K. J., Kearns, K. L., Yu, L. & Ediger, M. D. Physical vapor deposition as a route to hidden amorphous states. Proc. Natl Acad. Sci. USA 106, 15165–15170 (2009).
Ishii, K., Nakayama, H., Hirabayashi, S. & Moriyama, R. Anomalously high-density glass of ethylbenzene prepared by vapor deposition at temperatures close to the glass-transition temperature. Chem. Phys. Lett. 459, 109–112 (2008).
Acknowledgements
We appreciate support received from the Deutsche Forschungsgemeinschaft (DFG). C.A.A. acknowledges helpful discussions with M. D. Ediger and JY-Z. Yue.
Author information
Authors and Affiliations
Contributions
C.A.A. and R.B. conceived the project, I.G. and S.W. planned and carried out the experimental work, S.W. and I.G. analyzed the data, and C.A.A. wrote the paper with important literature and diagrammatic input from S.W. and advice from I.G. and R.B.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1149 kb)
Rights and permissions
About this article
Cite this article
Wei, S., Gallino, I., Busch, R. et al. Glass transition with decreasing correlation length during cooling of Fe50Co50 superlattice and strong liquids. Nature Phys 7, 178–182 (2011). https://doi.org/10.1038/nphys1823
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nphys1823
This article is cited by
-
Relaxation dynamics in the strong chalcogenide glass-former of Ge22Se78
Scientific Reports (2017)
-
Energy landscape-driven non-equilibrium evolution of inherent structure in disordered material
Nature Communications (2017)
-
Liquid–liquid transition in a strong bulk metallic glass-forming liquid
Nature Communications (2013)