Abstract
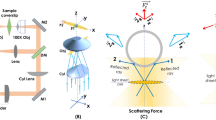
Optical tweezers are a flexible manipulation tool used to grab micro-objects at a specific point, but a controlled manipulation of objects with more complex or changing shapes is hardly possible. Here, we demonstrate, by time-sharing optical forces, that it is possible to adapt the shape of the trapping potential to the shape of an elongated helical bacterium. In contrast to most other trapped objects, this structure can continuously change its helical shape (and therefore its mechanical energy), making trapping it much more difficult than trapping tiny non-living objects. The shape deformations of the only 200-nm-thin bacterium (Spiroplasma) are measured space-resolved at 800 Hz by exploiting local phase differences in coherently scattered trapping light. By localizing each slope of the bacterium we generate high-contrast, super-resolution movies in three dimensions, without any object staining. This approach will help in investigating the nanomechanics of single wall-less bacteria while reacting to external stimuli on a broad temporal bandwidth.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Dholakia, K. & Reece, P. Optical micromanipulation takes hold. Nano Today 1, 18–27 (2006).
Hell, S. W. Far-field optical nanoscopy. Science 316, 1153–1158 (2007).
Gustafsson, M. G. L. Surpassing the lateral resolution limit by a factor of two using structured illumination microscopy. J. Microsc. 198, 82–87 (2000).
Xu, W. B., Jericho, M. H., Meinertzhagen, I. A. & Kreuzer, H. J. Digital in-line holography for biological applications. Proc. Natl Acad. Sci. USA 98, 11301–11305 (2001).
Lee, S.-H. & Grier, D. G. Holographic microscopy of holographically trapped three-dimensional structures. Opt. Express 15, 1505–1512 (2007).
Pralle, A., Prummer, M., Florin, E.-L., Stelzer, E. H. K. & Hörber, J. K. H. Three-dimensional position tracking for optical tweezers by forward scattered light. Microsc. Res. Tech. 44, 378–386 (1999).
Rohrbach, A. & Stelzer, E. H. K. Three-dimensional position detection of optically trapped dielectric particles. J. Appl. Phys. 91, 5474–5488 (2002).
Dreyer, J. K., Berg-Sorensen, K. & Oddershede, L. Improved axial position detection in optical tweezers measurements. Appl. Opt. 43, 1991–1995 (2004).
Speidel, M., Friedrich, L. & Rohrbach, A. Interferometric 3D tracking of several particles in a scanning laser focus. Opt. Express 17, 1003–1015 (2009).
Friedrich, L. & Rohrbach, A. Improved interferometric tracking of trapped particles using two frequency detuned beams. Opt. Lett. 35, 1920–1922 (2010).
Kress, H., Stelzer, E. H. K. & Rohrbach, A. Tilt angle dependent three-dimensional-position detection of a trapped cylindrical particle in a focused laser beam. Appl. Phys. Lett. 84, 4271–4273 (2004).
Irrera, A. et al. Size-scaling in optical trapping of silicon nanowires. Nano Lett. 11, 4879–4884 (2011).
Min, T. L. et al. High-resolution, long-term characterization of bacterial motility using optical tweezers. Nature Methods 6, 831–U871 (2009).
Rowe, A. D., Leake, M. C., Morgan, H. & Berry, R. M. Rapid rotation of micron and submicron dielectric particles measured using optical tweezers. J. Mod. Opt. 50, 1539–1554 (2003).
Chattopadhyay, S., Moldovan, R., Yeung, C. & Wu, X. L. Swimming efficiency of bacterium Escherichia coli. Proc. Natl Acad. Sci. USA 103, 13712–13717 (2006).
Dombrowski, C. et al. The elastic basis for the shape of Borrelia burgdorferi. Biophys. J. 96, 4409–4417 (2009).
Wang, S. Y., Arellano-Santoyo, H., Combs, P. A. & Shaevitz, J. W. Actin-like cytoskeleton filaments contribute to cell mechanics in bacteria. Proc. Natl Acad. Sci. USA 107, 9182–9185 (2010).
Ashkin, A. & Dziedzic, J. M. Optical trapping and manipulation of viruses and bacteria. Science 235, 1517–1520 (1987).
Gilad, R., Porat, A. & Trachtenberg, S. Motility modes of Spiroplasma melliferum BC3: a helical, wall-less bacterium driven by a linear motor. Mol. Microbiol. 47, 657–669 (2003).
Wolgemuth, C. W., Igoshin, O. & Oster, G. The motility of mollicutes. Biophys. J. 85, 828–842 (2003).
Shaevitz, J. W., Lee, J. Y. & Fletcher, D. A. Spiroplasma swim by a processive change in body helicity. Cell 122, 941–945 (2005).
Bastian, F. O. et al. Spiroplasma spp. from transmissible spongiform encephalopathy brains or ticks induce spongiform encephalopathy in ruminants. J. Med. Microbiol. 56, 1235–1242 (2007).
Trachtenberg, S. & Gilad, R. A bacterial linear motor: cellular and molecular organization of the contractile cytoskeleton of the helical bacterium Spiroplasma melliferum BC3. Mol. Microbiol. 41, 827–848 (2001).
Trachtenberg, S. The cytoskeleton of Spiroplasma: a complex linear motor. J. Mol. Microbiol. Biotechnol. 11, 265–283 (2006).
Kurner, J., Frangakis, A. S. & Baumeister, W. Cryo-electron tomography reveals the cytoskeletal structure of Spiroplasma melliferum. Science 307, 436–438 (2005).
Rohrbach, A. Stiffness of optical traps: quantitative agreement between experiment and electromagnetic theory. Phys. Rev. Lett. 95, 168102 (2005).
Faucheux, L. P., Stolovitzky, G. & Libchaber, A. Periodic forcing of a Brownian particle. Phys. Rev. E 51, 5239–5250 (1995).
Nambiar, R. & Meiners, J. C. Fast position measurements with scanning line optical tweezers. Opt. Lett. 27, 836–838 (2002).
Neuman, K. C., Chadd, E. H., Liou, G. F., Bergman, K. & Block, S. M. Characterization of photodamage to Escherichia coli in optical traps. Biophys. J. 77, 2856–2863 (1999).
Seitz, P. C., Stelzer, E. H. K. & Rohrbach, A. Interferometric tracking of optically trapped probes behind structured surfaces: a phase correction method. Appl. Opt. 45, 7903–7915 (2006).
Born, M. & Wolf, E. Principles of Optics 5th edn (Cambridge Univ. Press, 1975).
Acknowledgements
The authors thank S. Trachtenberg, F. Hamprecht, J. Huisken and L. Friedrich for stimulating discussions, as well as J. Korvink, B. Tränkle, F. Fahrbach, F. Kohler, B. Landenberger and B. Bosworth for a careful reading of the manuscript. This study was supported by the Excellence Initiative of the German Federal and State Governments (EXC 294) and by the Deutsche Forschungsgemeinschaft (DFG) (grant nos RO 3615/1 and RO 3615/2).
Author information
Authors and Affiliations
Contributions
M.K. performed experiments and simulations, analysed data and prepared all graphs. A.R. initiated and supervised the project, designed the system, developed the theory and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 1822 kb)
Supplementary Movie 1
Supplementary Movie 1 (AVI 6739 kb)
Supplementary Movie 2
Supplementary Movie 2 (AVI 6739 kb)
Supplementary Movie 3
Supplementary Movie 3 (AVI 2782 kb)
Supplementary Movie 4
Supplementary Movie 4 (AVI 41 kb)
Supplementary Movie 5
Supplementary Movie 5 (AVI 706 kb)
Rights and permissions
About this article
Cite this article
Koch, M., Rohrbach, A. Object-adapted optical trapping and shape-tracking of energy-switching helical bacteria. Nature Photon 6, 680–686 (2012). https://doi.org/10.1038/nphoton.2012.232
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nphoton.2012.232
This article is cited by
-
Towards non-blind optical tweezing by finding 3D refractive index changes through off-focus interferometric tracking
Nature Communications (2021)
-
Extending calibration-free force measurements to optically-trapped rod-shaped samples
Scientific Reports (2017)
-
Fast, label-free super-resolution live-cell imaging using rotating coherent scattering (ROCS) microscopy
Scientific Reports (2016)
-
Surface imaging beyond the diffraction limit with optically trapped spheres
Nature Nanotechnology (2015)
-
Dynamic manipulation of particles via transformative optofluidic waveguides
Scientific Reports (2015)