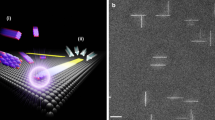
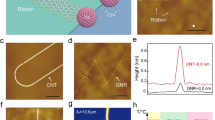
Abstract
Assembling inorganic nanomaterials on graphene1,2,3 is of interest in the development of nanodevices and nanocomposite materials, and the ability to align such inorganic nanomaterials on the graphene surface is expected to lead to improved functionalities4, as has previously been demonstrated with organic nanomaterials epitaxially aligned on graphitic surfaces5,6,7,8,9,10. However, because graphene is chemically inert, it is difficult to precisely assemble inorganic nanomaterials on pristine graphene2,11,12,13,14,15,16. Previous techniques2,3 based on dangling bonds of damaged graphene11,17,18,19,20, intermediate seed materials11,15,16,21,22 and vapour-phase deposition at high temperature12,13,14,23,24,25 have only formed randomly oriented or poorly aligned inorganic nanostructures. Here, we show that inorganic nanowires of gold(I) cyanide can grow directly on pristine graphene, aligning themselves with the zigzag lattice directions of the graphene. The nanowires are synthesized through a self-organized growth process in aqueous solution at room temperature, which indicates that the inorganic material spontaneously binds to the pristine graphene surface. First-principles calculations suggest that this assembly originates from lattice matching and π interaction to gold atoms. Using the synthesized nanowires as templates, we also fabricate nanostructures with controlled crystal orientations such as graphene nanoribbons with zigzag-edged directions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Geim, A. K. & Grigorieva, I. V. Van der Waals heterostructures. Nature 499, 419–425 (2013).
Georgakilas, V. et al. Functionalization of graphene: covalent and non-covalent approaches, derivatives and applications. Chem. Rev. 112, 6156–6214 (2012).
Huang, X., Qi, X., Boey, F. & Zhang, H. Graphene-based composites. Chem. Soc. Rev. 41, 666–686 (2012).
Barth, J. V., Costantini, G. & Kern, K. Engineering atomic and molecular nanostructures at surfaces. Nature 437, 671–679 (2005).
Hong, G. et al. Recent progress in organic molecule/graphene interfaces. Nano Today 8, 388–402 (2013).
Kim, D. W., Kim, Y. H., Jeong, H. S. & Jung, H-T. Direct visualization of large-area graphene domains and boundaries by optical birefringency. Nature Nanotech. 7, 29–34 (2012).
Garnica, M. et al. Long-range magnetic order in a purely organic 2D layer adsorbed on epitaxial graphene. Nature Phys. 9, 368–374 (2013).
Colson, J. W. et al. Oriented 2D covalent organic framework thin films on single-layer graphene. Science 332, 228–231 (2011).
Zhang, F. et al. Epitaxial growth of peptide nanofilaments on inorganic surfaces: effects of interfacial hydrophobicity/hydrophilicity. Angew. Chem. Int. Ed. 45, 3611–3613 (2006).
Fukushima, T. et al. Molecular ordering of organic molten salts triggered by single-walled carbon nanotubes. Science 300, 2072–2074 (2003).
Chung, K., Lee, C-H. & Yi, G-C. Transferable GaN layers grown on ZnO-coated graphene layers for optoelectronic devices. Science 330, 655–657 (2010).
Addou, R., Dahal, A. & Batzill, M. Growth of a two-dimensional dielectric monolayer on quasi-freestanding graphene. Nature Nanotech. 8, 41–45 (2013).
Dang, W., Peng, H., Li, H., Wang, P. & Liu, Z. Epitaxial heterostructures of ultrathin topological insulator nanoplate and graphene. Nano Lett 10, 2870–2876 (2010).
Hong, Y. J. et al. Van der Waals epitaxial double heterostructure: InAs/single-layer graphene/InAs. Adv. Mater. 25, 6847–6853 (2013).
Wang, X., Tabakman, S. M. & Dai, H. Atomic layer deposition of metal oxides on pristine and functionalized graphene. J. Am. Chem. Soc. 130, 8152–8153 (2008).
Alaboson, J. M. P. et al. Templating sub-10 nm atomic layer deposited oxide nanostructures on graphene via one-dimensional organic self-assembled monolayers. Nano Lett. 13, 5763–5770 (2013).
Kim, K. et al. Selective metal deposition at graphene line defects by atomic layer deposition. Nature Commun. 5, 4781 (2014).
Liang, Y. et al. Co3O4 nanocrystals on graphene as a synergistic catalyst for oxygen reduction reaction. Nature Mater. 10, 780–786 (2011).
Si, Y. & Samulski, E. T. Exfoliated graphene separated by platinum nanoparticles. Chem. Mater. 20, 6792–6797 (2008).
Lightcap, I. V., Kosel, T. H. & Kamat, P. V. Anchoring semiconductor and metal nanoparticles on a two-dimensional catalyst mat. Storing and shuttling electrons with reduced graphene oxide. Nano Lett. 10, 577–583 (2010).
Choi, D. et al. Fully rollable transparent nanogenerators based on graphene electrodes. Adv. Mater. 22, 2187–2192 (2010).
Huang, X. et al. Reduced graphene oxide-templated photochemical synthesis and in situ assembly of Au nanodots to orderly patterned Au nanodot chains. Small 6, 513–516 (2010).
Kumar, B. et al. Controlled growth of semiconducting nanowire, nanowall, and hybrid nanostructures on graphene for piezoelectric nanogenerators. ACS Nano 5, 4197–4204 (2011).
Shi, Y. et al. Van der Waals epitaxy of MoS2 layers using graphene as growth templates. Nano Lett. 12, 2784–2791 (2012).
Zhou, H. et al. The transformation of a gold film on few-layer graphene to produce either hexagonal or triangular nanoparticles during annealing. Carbon 52, 379–387 (2013).
Kim, K. et al. Grain boundary mapping in polycrystalline graphene. ACS Nano 5, 2142–2146 (2011).
Hibble, S. J., Hannon, A. C. & Cheyne, S. M. Structure of AuCN determined from total neutron diffraction. Inorg. Chem. 42, 4724–4730 (2003).
Giovannetti, G. et al. Doping graphene with metal contacts. Phys. Rev. Lett. 101, 026803 (2008).
Sarkar, S. et al. Organometallic hexahapto functionalization of single layer graphene as a route to high mobility graphene devices. Adv. Mater. 25, 1131–1136 (2013).
Son, Y-W., Cohen, M. L. & Louie, S. G. Half-metallic graphene nanoribbons. Nature 444, 347–349 (2006).
Jiao, L., Zhang, L., Wang, X., Diankov, G. & Dai, H. Narrow graphene nanoribbons from carbon nanotubes. Nature 458, 877–880 (2009).
Shi, Z. et al. Patterning graphene with zigzag edges by self-aligned anisotropic etching. Adv. Mater. 23, 3061–3065 (2011).
Halas, N. J., Lal, S., Chang, W. S., Link, S. & Nordlander, P. Plasmons in strongly coupled metallic nanostructures. Chem. Rev. 111, 3913–3961 (2011).
Acknowledgements
The authors thank A.P. Alivisatos, H. Fujita, Y. Arakawa, B.J. Kim, L. Yang, J. Moon, Y. Ota, H. Suh, J. Kwon and J. Min for helpful discussions. The authors also thank J. Kim and Y. Mizutani for the AFM analysis, S. Mori and M. Onuki for technical support and A. Sato for help with graphic illustrations. This work was mainly supported by the Takeuchi Biohybrid Innovation Project, Exploratory Research for Advanced Technology (ERATO), Japan Science and Technology (JST). A.Z. and K.K. acknowledge support from the Director, Office of Energy Research, Materials Sciences and Engineering Division, of the US Department of Energy (DE-AC02-05CH11231) and from the Office of Naval Research (MURI grant N00014-09-1066). K.K. also acknowledges support from the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2014R1A1A2058178). D.A.W. and J.P. acknowledge support from the Harvard MRSEC (DMR-0820484) and Amore-Pacific. H.L. and J.K. acknowledge support from the Basic Science Research Program through the National Research Foundation of Korea, funded by the Ministry of Science, ICT & Future Planning (NRF-2015R1A1A1001583) and also acknowledge support from KISTI under the Supercomputing Applications Support Program (KSC-2013-C3-034). H.Y.J. acknowledges support from the 2012 Research Fund (1.120032.01) of UNIST.
Author information
Authors and Affiliations
Contributions
W.C.L., K.K. and J.P. conceived the design of the study. S.T., A.Z. and D.A.W. supervised the project. K.K. and J.P. initially discovered the nanowire synthesis phenomenon. W.C.L., K.K., J.P. and H.Y.J. performed all experiments. J.K. and H.L. performed first-principles calculations. W.C.L., K.K., J.P., H.L., D.A.W., A.Z. and S.T. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 14729 kb)
Supplementary Movie 1
Supplementary Movie 1 (MOV 10423 kb)
Rights and permissions
About this article
Cite this article
Lee, W., Kim, K., Park, J. et al. Graphene-templated directional growth of an inorganic nanowire. Nature Nanotech 10, 423–428 (2015). https://doi.org/10.1038/nnano.2015.36
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2015.36
This article is cited by
-
Drying-Mediated Self-Assembly of Graphene for Inkjet Printing of High-Rate Micro-supercapacitors
Nano-Micro Letters (2020)
-
Scalable salt-templated synthesis of two-dimensional transition metal oxides
Nature Communications (2016)