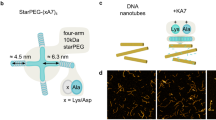
Abstract
The sarcomere of muscle is composed of tens of thousands of myosin motors that self-assemble into thick filaments and interact with surrounding actin-based thin filaments in a dense, near-crystalline hexagonal lattice1. Together, these actin–myosin interactions enable large-scale movement and force generation, two primary attributes of muscle. Research on isolated fibres has provided considerable insight into the collective properties of muscle, but how actin–myosin interactions are coordinated in an ensemble remains poorly understood2. Here, we show that artificial myosin filaments, engineered using a DNA nanotube scaffold, provide precise control over motor number, type and spacing. Using both dimeric myosin V- and myosin VI-labelled nanotubes, we find that neither myosin density nor spacing has a significant effect on the gliding speed of actin filaments. This observation supports a simple model of myosin ensembles as energy reservoirs that buffer individual stochastic events to bring about smooth, continuous motion. Furthermore, gliding speed increases with cross-bridge compliance, but is limited by Brownian effects. As a first step to reconstituting muscle motility, we demonstrate human β-cardiac myosin-driven gliding of actin filaments on DNA nanotubes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Huxley, H. E. The mechanism of muscular contraction. Science 164, 1356–1365 (1969).
Guérin, T., Prost, J., Martin, P. & Joanny, J. F. Coordination and collective properties of molecular motors: theory. Curr. Opin. Cell Biol. 22, 14–20 (2010).
De La Cruz, E. M. & Ostap, E. M. Relating biochemistry and function in the myosin superfamily. Curr. Opin. Cell Biol. 16, 61–67 (2004).
Uyeda, T. Q., Kron, S. J. & Spudich, J. A. Myosin step size. Estimation from slow sliding movement of actin over low densities of heavy meromyosin. J. Mol. Biol. 214, 699–710 (1990).
Sommese, R. F. et al. Molecular consequences of the R453C hypertrophic cardiomyopathy mutation on human β-cardiac myosin motor function. Proc. Natl Acad. Sci. USA 110, 12607–12612 (2013).
Harris, D. E. & Warshaw, D. M. Smooth and skeletal muscle myosin both exhibit low duty cycles at zero load in vitro. J. Biol. Chem. 268, 14764–14768 (1993).
Piazzesi, G. et al. Skeletal muscle performance determined by modulation of number of myosin motors rather than motor force or stroke size. Cell 131, 784–795 (2007).
Spudich, J. A. Hypertrophic and dilated cardiomyopathy: four decades of basic research on muscle lead to potential therapeutic approaches to these devastating genetic diseases. Biophys. J. 106, 1236–1249 (2014).
Baker, J. E., Brosseau, C., Joel, P. B. & Warshaw, D. M. The biochemical kinetics underlying actin movement generated by one and many skeletal muscle myosin molecules. Biophys. J. 82, 2134–2147 (2002).
Derr, N. D. et al. Tug-of-war in motor protein ensembles revealed with a programmable DNA origami scaffold. Science 338, 662–665 (2012).
Hariadi, R. F., Cale, M. & Sivaramakrishnan, S. Myosin lever arm directs collective motion on cellular actin network. Proc. Natl Acad. Sci. USA 111, 4091–4096 (2014).
Walcott, S., Warshaw, D. M. & Debold, E. P. Mechanical coupling between myosin molecules causes differences between ensemble and single-molecule measurements. Biophys. J. 103, 501–510 (2012).
Rock, R. S., Rief, M., Mehta, A. D. & Spudich, J. A. In vitro assays of processive myosin motors. Methods 22, 373–381 (2000).
Lu, H. et al. Collective dynamics of elastically coupled myosin V motors. J. Biol. Chem. 287, 27753–27761 (2012).
Yin, P. et al. Programming DNA tube circumferences. Science 321, 824–826 (2008).
Ali, M. Y. et al. Myosin Va and myosin VI coordinate their steps while engaged in an in vitro tug of war during cargo transport. Proc. Natl Acad. Sci. USA 108, E535–E541 (2011).
Hilbert, L., Cumarasamy, S., Zitouni, N. B., Mackey, M. C. & Lauzon, A. M. The kinetics of mechanically coupled myosins exhibit group size-dependent regimes. Biophys. J. 105, 1466–1474 (2013).
Rief, M. et al. Myosin-V stepping kinetics: a molecular model for processivity. Proc. Natl Acad. Sci. USA 97, 9482–9486 (2000).
Rock, R. S. et al. Myosin VI is a processive motor with a large step size. Proc. Natl Acad. Sci. USA 98, 13655–13659 (2001).
Gebhardt, J. C. M., Clemen, A. E. M., Jaud, J. & Rief, M. Myosin-V is a mechanical ratchet. Proc. Natl Acad. Sci. USA 103, 8680–8685 (2006).
Veigel, C., Molloy, J. E., Schmitz, S. & Kendrick-Jones, J. Load-dependent kinetics of force production by smooth muscle myosin measured with optical tweezers. Nature Cell Biol. 5, 980–986 (2003).
Walsh, R., Rutland, C., Thomas, R. & Loughna, S. Cardiomyopathy: a systematic review of disease-causing mutations in myosin heavy chain 7 and their phenotypic manifestations. Cardiology 115, 49–60 (2010).
Weith, A. et al. Unique single molecule binding of cardiac myosin binding protein-C to actin and phosphorylation-dependent inhibition of actomyosin motility requires 17 amino acids of the motif domain. J. Mol. Cell. Cardiol. 52, 219–227 (2012).
Resnicow, D. I., Deacon, J. C., Warrick, H. M., Spudich, J. A. & Leinwand, L. A. Functional diversity among a family of human skeletal muscle myosin motors. Proc. Natl Acad. Sci. USA 107, 1053–1058 (2010).
Trybus, K. M., Freyzon, Y., Faust, L. Z. & Sweeney, H. L. Spare the rod, spoil the regulation: the necessity for a myosin rod. Proc. Natl Acad. Sci. USA 94, 48–52 (1997).
Huang, J., Nagy, S. S., Koide, A., Rock, R. S. & Koide, S. A peptide tag system for facile purification and single-molecule immobilization. Biochemistry 48, 11834–11836 (2009).
Acknowledgements
The authors thank M. Westfall, D. Smith and L. Hilbert for useful discussions. Research was funded by the American Heart Association Scientist Development Grant (13SDG14270009), National Institutes of Health (NIH) grants 1DP2 CA186752-01 and 1-R01-GM-105646-01-A1 to S.S. and NIH grants GM33289 and HL117138 to J.A.S. R.F.S. is a Life Sciences Research Foundation Fellow. R.E.T. is supported by the NIH (F32 HL123247-02) and A.S.A. is supported by a Lucile Packard CHRI Postdoctoral Award.
Author information
Authors and Affiliations
Contributions
R.F.H., R.F.S., A.S.A., R.E.T., J.A.S. and S.S. planned and designed experiments. R.F.H., R.F.S., A.S.A., R.E.T. and S.Su. performed experiments and analysed the results. R.F.H. and S.S. performed the mathematical modelling. R.F.H., R.F.S., J.A.S. and S.S. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary Information (PDF 1135 kb)
Supplementary Movie 1
Supplementary Movie 1 (AVI 1272 kb)
Supplementary Movie 2
Supplementary Movie 2 (AVI 2415 kb)
Supplementary Movie 3
Supplementary Movie 3 (AVI 38 kb)
Rights and permissions
About this article
Cite this article
Hariadi, R., Sommese, R., Adhikari, A. et al. Mechanical coordination in motor ensembles revealed using engineered artificial myosin filaments. Nature Nanotech 10, 696–700 (2015). https://doi.org/10.1038/nnano.2015.132
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2015.132
This article is cited by
-
Fabricating higher-order functional DNA origami structures to reveal biological processes at multiple scales
NPG Asia Materials (2023)
-
DNA origami
Nature Reviews Methods Primers (2021)
-
Growth and site-specific organization of micron-scale biomolecular devices on living mammalian cells
Nature Communications (2021)
-
Direct visualization of human myosin II force generation using DNA origami-based thick filaments
Communications Biology (2019)
-
A myosin II nanomachine mimicking the striated muscle
Nature Communications (2018)