Abstract
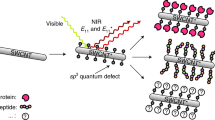
Single-walled carbon nanotubes are particularly attractive for biomedical applications, because they exhibit a fluorescent signal in a spectral region where there is minimal interference from biological media. Although single-walled carbon nanotubes have been used as highly sensitive detectors for various compounds, their use as in vivo biomarkers requires the simultaneous optimization of various parameters, including biocompatibility, molecular recognition, high fluorescence quantum efficiency and signal transduction. Here we show that a polyethylene glycol ligated copolymer stabilizes near-infrared-fluorescent single-walled carbon nanotubes sensors in solution, enabling intravenous injection into mice and the selective detection of local nitric oxide concentration with a detection limit of 1 µM. The half-life for liver retention is 4 h, with sensors clearing the lungs within 2 h after injection, thus avoiding a dominant route of in vivo nanotoxicology. After localization within the liver, it is possible to follow the transient inflammation using nitric oxide as a marker and signalling molecule. To this end, we also report a spatial-spectral imaging algorithm to deconvolute fluorescence intensity and spatial information from measurements. Finally, we demonstrate that alginate-encapsulated single-walled carbon nanotubes can function as implantable inflammation sensors for nitric oxide detection, with no intrinsic immune reactivity or other adverse response for more than 400 days.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Wray, S., Cope, M., Delpy, D. T., Wyatt, J. S. & Reynolds, E. O. Characterization of the near infrared absorption spectra of cytochrome aa3 and haemoglobin for the non-invasive monitoring of cerebral oxygenation. Biochim. Biophys. Acta 933, 184–192 (1988).
Heller, D. A. et al. Multimodal optical sensing and analyte specificity using single-walled carbon nanotubes. Nature Nanotech. 4, 114–120 (2009).
Jin, H., Heller, D. A., Kim, J. H. & Strano, M. S. Stochastic analysis of stepwise fluorescence quenching reactions on single-walled carbon nanotubes: single molecule sensors. Nano Lett. 8, 4299–4304 (2008).
Jin, H. et al. Detection of single-molecule H2O2 signalling from epidermal growth factor receptor using fluorescent single-walled carbon nanotubes. Nature Nanotech. 5, 302–309 (2010).
Kim, J. H. et al. The rational design of nitric oxide selectivity in single-walled carbon nanotube near-infrared fluorescence sensors for biological detection. Nature Chem. 1, 473–481 (2009).
Zhang, J. Q. et al. Single molecule detection of nitric oxide enabled by d(AT)15 DNA adsorbed to near infrared fluorescent single-walled carbon nanotubes. J. Am. Chem. Soc. 133, 567–581 (2011).
Liu, X. et al. Optimization of surface chemistry on single-walled carbon nanotubes for in vivo photothermal ablation of tumors. Biomaterials 32, 144–151 (2011).
Liu, Z. et al. In vivo biodistribution and highly efficient tumour targeting of carbon nanotubes in mice. Nature Nanotech. 2, 47–52 (2007).
Liu, Z. et al. Drug delivery with carbon nanotubes for in vivo cancer treatment. Cancer Res. 68, 6652–6660 (2008).
Liu, Z. et al. Circulation and long-term fate of functionalized, biocompatible single-walled carbon nanotubes in mice probed by Raman spectroscopy. Proc. Natl Acad. Sci. USA 105, 1410–1415 (2008).
Liu, Z. et al. Supramolecular stacking of doxorubicin on carbon nanotubes for in vivo cancer therapy. Angew. Chem. Int. Ed. 48, 7668–7672 (2009).
Cherukuri, P. et al. Mammalian pharmacokinetics of carbon nanotubes using intrinsic near-infrared fluorescence. Proc. Natl Acad. Sci. USA 103, 18882–18886 (2006).
Singh, R. et al. Tissue biodistribution and blood clearance rates of intravenously administered carbon nanotube radiotracers. Proc. Natl Acad. Sci. USA 103, 3357–3362 (2006).
Liu, Z., Tabakman, S., Welsher, K. & Dai, H. Carbon nanotubes in biology and medicine: in vitro and in vivo detection, imaging and drug delivery. Nano Res. 2, 85–120 (2009).
Liu, Z., Tabakman, S. M., Chen, Z. & Dai, H. Preparation of carbon nanotube bioconjugates for biomedical applications. Nature Protoc. 4, 1372–1381 (2009).
Robinson, J. T. et al. High performance in vivo near-IR (>1 µm) imaging and photothermal cancer therapy with carbon nanotubes. Nano Res. 3, 779–793 (2010).
Sato, Y. et al. Influence of length on cytotoxicity of multi-walled carbon nanotubes against human acute monocytic leukemia cell line THP-1 in vitro and subcutaneous tissue of rats in vivo. Mol. BioSyst. 1, 176–182 (2005).
Schipper, M. L. et al. A pilot toxicology study of single-walled carbon nanotubes in a small sample of mice. Nature Nanotech. 3, 216–221 (2008).
Welsher, K. et al. A route to brightly fluorescent carbon nanotubes for near-infrared imaging in mice. Nature Nanotech. 4, 773–780 (2009).
Moncada, S., Palmer, R. M. & Higgs, E. A. Nitric oxide: physiology, pathophysiology, and pharmacology. Pharmacol. Rev. 43, 109–142 (1991).
Park, S. S. et al. Real-time in vivo simultaneous measurements of nitric oxide and oxygen using an amperometric dual microsensor. Anal. Chem. 82, 7618–7624 (2010).
Thomas, D. D. et al. The chemical biology of nitric oxide: implications in cellular signaling. Free Radic. Bio. Med. 45, 18–31 (2008).
Lewis, R. S., Tamir, S., Tannenbaum, S. R. & Deen, W. M. Kinetic analysis of the fate of nitric oxide synthesized by macrophages in vitro. J. Biol. Chem. 270, 29350–29355 (1995).
Dedon, P. C. & Tannenbaum, S. R. Reactive nitrogen species in the chemical biology of inflammation. Arch. Biochem. Biophys. 423, 12–22 (2004).
Coussens, L. M. & Werb, Z. Inflammation and cancer. Nature 420, 860–867 (2002).
Barone, P. W., Baik, S., Heller, D. A. & Strano, M. S. Near-infrared optical sensors based on single-walled carbon nanotubes. Nature Mater. 4, 86–U16 (2005).
Heller, D. A. et al. Optical detection of DNA conformational polymorphism on single-walled carbon nanotubes. Science 311, 508–511 (2006).
Ahn, J. H. et al. Label-free, single protein detection on a near-infrared fluorescent single-walled carbon nanotube/protein microarray fabricated by cell-free synthesis. Nano Lett. 11, 2743–2752 (2011).
Choi, J. H., Chen, K. H. & Strano, M. S. Aptamer-capped nanocrystal quantum dots: a new method for label-free protein detection. J. Am. Chem. Soc. 128, 15584–15585 (2006).
Schoppler, F. et al. Molar extinction coefficient of single-wall carbon nanotubes. J. Phys. Chem. 115, 14682–14686 (2011).
Donaldson, K. et al. Carbon nanotubes: a review of their properties in relation to pulmonary toxicology and workplace safety. Toxicol. Sci. 92, 5–22 (2006).
Lacerda, L., Bianco, A., Prato, M. & Kostarelos, K. Carbon nanotubes as nanomedicines: from toxicology to pharmacology. Adv. Drug Deliv. Rev. 58, 1460–1470 (2006).
Poland, C. A. et al. Carbon nanotubes introduced into the abdominal cavity of mice show asbestos-like pathogenicity in a pilot study. Nature Nanotech. 3, 423–428 (2008).
Gal, A., Tamir, S., Tannenbaum, S. & Wogan, G. Nitric oxide production in SJL mice bearing the RcsX lymphoma: a model for in vivo toxicological evaluation of NO. Proc. Natl Acad. Sci. USA 93, 11499–11503 (1996).
Lee, R. H., Efron, D., Tantry, U. & Barbul, A. Nitric oxide in the healing wound: a time-course study. J. Surg. Res. 101, 104–108 (2001).
Davidson, J. M. Animal models for wound repair. Arch. Dermatol. Res. 290, S1–S11 (1998).
Griveau, S. & Bedioui, F. Overview of significant examples of electrochemical sensor arrays designed for detection of nitric oxide and relevant species in a biological environment. Anal. Bioanal. Chem. 405, 3475–3488 (2013).
Bredt, D. S. & Snyder, S. H. Nitric oxide: a physiologic messenger molecule. Annu. Rev. Biochem. 63, 175–195 (1994).
Mantovani, A., Allavena, P., Sica, A. & Balkwill, F. Cancer-related inflammation. Nature 454, 436–444 (2008).
Acknowledgements
This work was supported by the National Institutes of Health (T32 Training Grant in Environmental Toxicology ES007020, to N.I.), the National Cancer Institute (grant P01 CA26731), the National Institute of Environmental Health Sciences (grant P30 ES002109), a Beckman Young Investigator Award and a National Science Foundation Presidential Early Career Award for Scientists and Engineers (to M.S.S.), the TUBITAK 2211 and 2214 Research fellowship programme (F.S. and S.S.) and the METU-DPT-OYP programme (F.S. and S.S). A Biomedical Innovation grant from Sanofi Aventis to M.S.S. is also acknowledged. The authors thank T. Grusecki for assistance with the deconvolution code, as well as R. Langer, D. Anderson and P. Dedon for helpful discussions.
Author information
Authors and Affiliations
Contributions
M.S.S. conceived of the original concept, with experimental design of in vivo experiments by G.W., M.S.S., N.I. and P.B. Sensor design and synthesis were performed by P.B., M.S., S.S., F.S., T.M., N.R. and N.I. N.I., L.T., M.S., V.I., E.A. and E.F. performed and analysed the in vivo studies. N.I., P.B., M.S., T.M. and N.R. optimized the animal imaging system. V.I. and N.I. designed the mathematical model and computer program to deconvolute the data. N.P. read and interpreted the histology slides. The manuscript was written by M.S.S and N.I. with contributions from G.W., P.B., L.T. and T.M.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary Information (PDF 1155 kb)
Rights and permissions
About this article
Cite this article
Iverson, N., Barone, P., Shandell, M. et al. In vivo biosensing via tissue-localizable near-infrared-fluorescent single-walled carbon nanotubes. Nature Nanotech 8, 873–880 (2013). https://doi.org/10.1038/nnano.2013.222
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2013.222
This article is cited by
-
Development and evaluation of an expedited system for creation of single walled carbon nanotube platforms
Carbon Letters (2024)
-
In-Vivo fluorescent nanosensor implants based on hydrogel-encapsulation: investigating the inflammation and the foreign-body response
Journal of Nanobiotechnology (2023)
-
Near-infrared luminescence high-contrast in vivo biomedical imaging
Nature Reviews Bioengineering (2023)
-
Near-infrared-II deep tissue fluorescence microscopy and application
Nano Research (2023)
-
A wavelength-induced frequency filtering method for fluorescent nanosensors in vivo
Nature Nanotechnology (2022)