Abstract
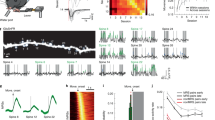
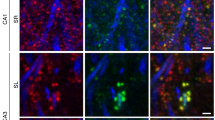
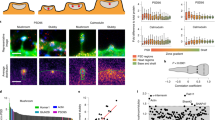
Dendritic spines appear and disappear in an experience-dependent manner. Although some new spines have been shown to contain synapses, little is known about the relationship between spine addition and synapse formation, the relative time course of these events, or whether they are coupled to de novo growth of axonal boutons. We imaged dendrites in barrel cortex of adult mice over 1 month, tracking gains and losses of spines. Using serial section electron microscopy, we analyzed the ultrastructure of spines and associated boutons. Spines reconstructed shortly after they appeared often lacked synapses, whereas spines that persisted for 4 d or more always had synapses. New spines had a large surface-to-volume ratio and preferentially contacted boutons with other synapses. In some instances, two new spines contacted the same axon. Our data show that spine growth precedes synapse formation and that new synapses form preferentially onto existing boutons.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Beaulieu, C. & Colonnier, M. A laminar analysis of the number of round-asymmetrical and flat-symmetrical synapses on spines, dendritic trunks, and cell bodies in area 17 of the cat. J. Comp. Neurol. 231, 180–189 (1985).
Harris, K.M. & Stevens, J.K. Dendritic spines of CA1 pyramidal cells in the rat hippocampus: serial electron microscopy with reference to their biophysical characteristics. J. Neurosci. 9, 2982–2997 (1989).
Nusser, Z. et al. Cell type and pathway dependence of synaptic AMPA receptor number and variability in the hippocampus. Neuron 21, 545–559 (1998).
Kharazia, V.N. & Weinberg, R.J. Immunogold localization of AMPA and NMDA receptors in somatic sensory cortex of albino rat. J. Comp. Neurol. 412, 292–302 (1999).
Takumi, Y., Ramirez-Leon, V., Laake, P., Rinvik, E. & Ottersen, O.P. Different modes of expression of AMPA and NMDA receptors in hippocampal synapses. Nat. Neurosci. 2, 618–624 (1999).
Katz, L.C. & Shatz, C.J. Synaptic activity and the construction of cortical circuits. Science 274, 1133–1138 (1996).
Chklovskii, D.B., Mel, B.W. & Svoboda, K. Cortical rewiring and information storage. Nature 431, 782–788 (2004).
Stepanyants, A., Hof, P.R. & Chklovskii, D.B. Geometry and structural plasticity of synaptic connectivity. Neuron 34, 275–288 (2002).
Knott, G.W., Quairiaux, C., Genoud, C. & Welker, E. Formation of dendritic spines with GABAergic synapses induced by whisker stimulation in adult mice. Neuron 34, 265–273 (2002).
Trachtenberg, J.T. et al. Long-term in vivo imaging of experience-dependent synaptic plasticity in adult cortex. Nature 420, 788–794 (2002).
Turner, A.M. & Greenough, W.T. Differential rearing effects on rat visual cortex synapses. I. Synaptic and neuronal density and synapses per neuron. Brain Res. 329, 195–203 (1985).
Yankova, M., Hart, S.A. & Woolley, C.S. Estrogen increases synaptic connectivity between single presynaptic inputs and multiple postsynaptic CA1 pyramidal cells: a serial electron-microscopic study. Proc. Natl. Acad. Sci. USA 98, 3525–3530 (2001).
Grutzendler, J., Kasthuri, N. & Gan, W.B. Long-term dendritic spine stability in the adult cortex. Nature 420, 812–816 (2002).
Holtmaat, A.J. et al. Transient and persistent dendritic spines in the neocortex in vivo. Neuron 45, 279–291 (2005).
Zuo, Y., Yang, G., Kwon, E. & Gan, W.B. Long-term sensory deprivation prevents dendritic spine loss in primary somatosensory cortex. Nature 436, 261–265 (2005).
Holtmaat, A., Wilbrecht, L., Knott, G.W., Welker, E. & Svoboda, K. Experience-dependent and cell-type-specific spine growth in the neocortex. Nature 441, 979–983 (2006).
Engert, F. & Bonhoeffer, T. Dendritic spine changes associated with hippocampal long-term synaptic plasticity. Nature 399, 66–70 (1999).
Maletic-Savatic, M., Malinow, R. & Svoboda, K. Rapid dendritic morphogenesis in CA1 hippocampal dendrites induced by synaptic activity. Science 283, 1923–1927 (1999).
Toni, N., Buchs, P.A., Nikonenko, I., Bron, C.R. & Muller, D. LTP promotes formation of multiple spine synapses between a single axon terminal and a dendrite. Nature 402, 421–425 (1999).
Harris, K.M. Structure, development, and plasticity of dendritic spines. Curr. Opin. Neurobiol. 9, 343–348 (1999).
Miller, M. & Peters, A. Maturation of rat visual cortex. II. A combined Golgi-electron microscope study of pyramidal neurons. J. Comp. Neurol. 203, 555–573 (1981).
Yuste, R. & Bonhoeffer, T. Genesis of dendritic spines: insights from ultrastructural and imaging studies. Nat. Rev. Neurosci. 5, 24–34 (2004).
Ziv, N.E. & Smith, S.J. Evidence for a role of dendritic filopodia in synaptogenesis and spine formation. Neuron 17, 91–102 (1996).
Feng, G. et al. Imaging neuronal subsets in transgenic mice expressing multiple spectral variants of GFP. Neuron 28, 41–51 (2000).
Denk, W., Strickler, J.H. & Webb, W.W. Two-photon laser scanning microscopy. Science 248, 73–76 (1990).
Vaughan, D.W. & Peters, A. A three dimensional study of layer I of the rat parietal cortex. J. Comp. Neurol. 149, 355–370 (1973).
Adams, I. & Jones, D.G. Quantitative ultrastructural changes in rat cortical synapses during early-, mid- and late-adulthood. Brain Res. 239, 349–363 (1982).
Colonnier, M. Synaptic patterns on different cell types in the different laminae of the cat visual cortex. An electron microscope study. Brain Res. 9, 268–287 (1968).
Okabe, S., Miwa, A. & Okado, H. Spine formation and correlated assembly of presynaptic and postsynaptic molecules. J. Neurosci. 21, 6105–6114 (2001).
Peters, A. & Kaiserman-Abramof, I.R. The small pyramidal neuron of the rat cerebral cortex. The perikaryon, dendrites and spines. Am. J. Anat. 127, 321–355 (1970).
Petrak, L.J., Harris, K.M. & Kirov, S.A. Synaptogenesis on mature hippocampal dendrites occurs via filopodia and immature spines during blocked synaptic transmission. J. Comp. Neurol. 484, 183–190 (2005).
Peters, A., Palay, S.L. & Webster, H.D. The Fine Structure of the Nervous System. (Oxford Univ. Press, New York, 1991).
Spacek, J. & Harris, K.M. Three-dimensional organization of smooth endoplasmic reticulum in hippocampal CA1 dendrites and dendritic spines of the immature and mature rat. J. Neurosci. 17, 190–203 (1997).
Mizrahi, A. & Katz, L.C. Dendritic stability in the adult olfactory bulb. Nat. Neurosci. 6, 1201–1207 (2003).
Lee, W.C. et al. Dynamic remodeling of dendritic arbors in GABAergic interneurons of adult visual cortex. PLoSBiol. 4, e29 (2006).
De Paola, V. et al. Cell type-specific structural plasticity of axonal branches and boutons in the adult neocortex. Neuron 49, 861–875 (2006).
Friedman, H.V., Bresler, T., Garner, C.C. & Ziv, N.E. Assembly of new individual excitatory synapses: time course and temporal order of synaptic molecule recruitment. Neuron 27, 57–69 (2000).
Harris, K.M., Jensen, F.E. & Tsao, B. Three-dimensional structure of dendritic spines and synapses in rat hippocampus (CA1) at postnatal day 15 and adult ages: implications for the maturation of synaptic physiology and long-term potentiation. J. Neurosci. 12, 2685–2705 (1992).
Pozzan, T., Rizzuto, R., Volpe, P. & Meldolesi, J. Molecular and cellular physiology of intracellular calcium stores. Physiol. Rev. 74, 595–636 (1994).
Braitenberg, V. & Schutz, A. Anatomy of the Cortex (Springer Verlag, Berlin, 1991).
Micheva, K.D. & Beaulieu, C. Quantitative aspects of synaptogenesis in the rat barrel field cortex with special reference to GABA circuitry. J. Comp. Neurol. 373, 340–354 (1996).
Jones, T.A., Klintsova, A.Y., Kilman, V.L., Sirevaag, A.M. & Greenough, W.T. Induction of multiple synapses by experience in the visual cortex of adult rats. Neurobiol. Learn. Mem. 68, 13–20 (1997).
Shepherd, G.M. & Harris, K.M. Three-dimensional structure and composition of CA3–CA1 axons in rat hippocampal slices: implications for presynaptic connectivity and compartmentalization. J. Neurosci. 18, 8300–8310 (1998).
Lu, S.M. & Lin, R.C.S. Thalamic afferents of the rat barrel cortex: a light- and electron-microscopic study using Phaseolus vulgaris leucoagglutinin as an anterograde tracer. Somatosens. Mot. Res. 10, 1–16 (1993).
Malinow, R. & Malenka, R.C. AMPA receptor trafficking and synaptic plasticity. Annu. Rev. Neurosci. 25, 103–126 (2002).
Denk, W. & Horstmann, H. Serial block-face scanning electron microscopy (SBFSEM) to reconstruct 3D tissue nanostructure. PLoSBiol. 2, e329 (2004).
Polsky, A., Mel, B.W. & Schiller, J. Computational subunits in thin dendrites of pyramidal cells. Nat. Neurosci. 7, 621–627 (2004).
Fox, K. Anatomical pathways and molecular mechanisms for plasticity in the barrel cortex. Neuroscience 111, 799–814 (2002).
Acknowledgements
We thank C. Musetti, V. DePaola and B. Burbach for help with the experiments, and M. Chklovskii, R. Weinberg, R. Weimer and K. Zito for comments on the manuscript. This work was supported by the Swiss National Science Foundation (E.W., No. 310000-108246), the Howard Hughes Medical Institute and the US National Institutes of Health (A.H., K.S. and L.W.).
Author information
Authors and Affiliations
Contributions
G.W.K. and A.H. contributed equally to this work. G.W.K., A.H. and K.S. planned the experiments. G.W.K. performed the ssEM. A.H. and L.W. performed the in vivo imaging experiments. K.S. built the custom two-photon microscope. K.S. and E.W. contributed reagents, materials and analysis tools, and provided financial support. G.W.K., A.H. and K.S. analyzed the data and wrote the paper. G.W.K., A.H., L.W., E.W. and K.S. discussed the results and commented on the manuscript.
Note: Supplementary information is available on the Nature Neuroscience website.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
The segments of dendrites imaged in vivo and reconstructed from serial electron micrographs. (PDF 834 kb)
Supplementary Fig. 2
Serial electron micrographs of spine 1 from Figure 3. (PDF 4872 kb)
Supplementary Fig. 3
Time lapse images (upper), 3d reconstruction (middle) and serial electron micrographs (lower) of 3 spines. (PDF 932 kb)
Supplementary Fig. 4
Two modes of synapse formation by spine growth. (PDF 64 kb)
Supplementary Table 1
Spine parameters. (PDF 49 kb)
Supplementary Table 2
Parameters of boutons synapsing on dendritic shafts. (PDF 50 kb)
Rights and permissions
About this article
Cite this article
Knott, G., Holtmaat, A., Wilbrecht, L. et al. Spine growth precedes synapse formation in the adult neocortex in vivo. Nat Neurosci 9, 1117–1124 (2006). https://doi.org/10.1038/nn1747
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1747
This article is cited by
-
Utilizing 2D-region-based CNNs for automatic dendritic spine detection in 3D live cell imaging
Scientific Reports (2023)
-
Filopodia are a structural substrate for silent synapses in adult neocortex
Nature (2022)
-
Learning binds new inputs into functional synaptic clusters via spinogenesis
Nature Neuroscience (2022)
-
A subpopulation of cortical VIP-expressing interneurons with highly dynamic spines
Communications Biology (2022)
-
Structure and function differences in the prelimbic cortex to basolateral amygdala circuit mediate trait vulnerability in a novel model of acute social defeat stress in male mice
Neuropsychopharmacology (2022)