Abstract
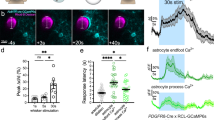
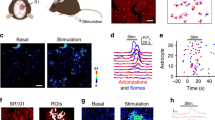
Local increase in blood flow during neural activity forms the basis for functional brain imaging, but its mechanism remains poorly defined. Here we show that cortical astrocytes in vivo possess a powerful mechanism for rapid vasodilation. We imaged the activity of astrocytes labeled with the calcium (Ca2+)-sensitive indicator rhod-2 in somatosensory cortex of adult mice. Photolysis of caged Ca2+ in astrocytic endfeet ensheathing the vessel wall was associated with an 18% increase in arterial cross-section area that corresponded to a 37% increase in blood flow. Vasodilation occurred with a latency of only 1–2 s, and both indomethacin and the cyclooxygenase-1 inhibitor SC-560 blocked the photolysis-induced hyperemia. These observations implicate astrocytes in the control of local microcirculation and suggest that one of their physiological roles is to mediate vasodilation in response to increased neural activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Iadecola, C. Neurovascular regulation in the normal brain and in Alzheimer's disease. Nat. Rev. Neurosci. 5, 347–360 (2004).
Lauritzen, M. Reading vascular changes in brain imaging: is dendritic calcium the key? Nat. Rev. Neurosci. 6, 77–85 (2005).
Chaigneau, E., Oheim, M., Audinat, E. & Charpak, S. Two-photon imaging of capillary blood flow in olfactory bulb glomeruli. Proc. Natl. Acad. Sci. USA 100, 13081–13086 (2003).
Simard, M., Arcuino, G., Takano, T., Liu, Q.S. & Nedergaard, M. Signaling at the gliovascular interface. J. Neurosci. 23, 9254–9262 (2003).
Hirase, H. A multi-photon window onto neuronal-glial-vascular communication. Trends Neurosci. 28, 217–219 (2005).
Mulligan, S.J. & MacVicar, B.A. Calcium transients in astrocyte endfeet cause cerebrovascular constrictions. Nature 431, 195–199 (2004).
Hirase, H., Qian, L., Bartho, P. & Buzsaki, G. Calcium dynamics of cortical astrocytic networks in vivo. PLoS Biol. 2, E96 (2004).
Tian, G.F. et al. An astrocytic basis of epilepsy. Nat. Med. 11, 973–981 (2005).
Filosa, J.A., Bonev, A.D. & Nelson, M.T. Calcium dynamics in cortical astrocytes and arterioles during neurovascular coupling. Circ. Res. 95, 73–81 (2004).
Ueki, M., Mies, G. & Hossmann, K.A. Effect of alpha-chloralose, halothane, pentobarbital and nitrous oxide anesthesia on metabolic coupling in somatosensory cortex of rat. Acta Anaesthesiol. Scand. 36, 318–322 (1992).
Lindauer, U., Villringer, A. & Dirnagl, U. Characterization of CBF response to somatosensory stimulation: model and influence of anesthetics. Am. J. Physiol. 264, H1223–H1228 (1993).
Gordon, E.L., Meno, J.R., Ngai, A.C., Lam, A.M. & Winn, H.R. Anesthetic-dependent pial arteriolar response to ethanol. J. Neurosurg. 83, 875–877 (1995).
Farooqui, A.A., Yang, H.C., Rosenberger, T.A. & Horrocks, L.A. Phospholipase A2 and its role in brain tissue. J. Neurochem. 69, 889–901 (1997).
Xu, J. et al. Prostaglandin E2 production in astrocytes: regulation by cytokines, extracellular ATP, and oxidative agents. Prostaglandins Leukot. Essent. Fatty Acids 69, 437–448 (2003).
Stephenson, D.T. et al. Calcium-sensitive cytosolic phospholipase A2 (cPLA2) is expressed in human brain astrocytes. Brain Res. 637, 97–105 (1994).
Xu, J., Yu, S., Sun, A.Y. & Sun, G.Y. Oxidant-mediated AA release from astrocytes involves cPLA(2) and iPLA(2). Free Radic. Biol. Med. 34, 1531–1543 (2003).
Hurley, S.D., Olschowka, J.A. & O'Banion, M.K. Cyclooxygenase inhibition as a strategy to ameliorate brain injury. J. Neurotrauma 19, 1–15 (2002).
Zonta, M. et al. Neuron-to-astrocyte signaling is central to the dynamic control of brain microcirculation. Nat. Neurosci. 6, 43–50 (2003).
Niwa, K., Haensel, C., Ross, M.E. & Iadecola, C. Cyclooxygenase-1 participates in selected vasodilator responses of the cerebral circulation. Circ. Res. 88, 600–608 (2001).
Niwa, K., Araki, E., Morham, S.G., Ross, M.E. & Iadecola, C. Cyclooxygenase-2 contributes to functional hyperemia in whisker-barrel cortex. J. Neurosci. 20, 763–770 (2000).
Ellis, E.F., Wei, E.P. & Kontos, H.A. Vasodilation of cat cerebral arterioles by prostaglandins D2, E2, G2, and I2. Am. J. Physiol. 237, H381–H385 (1979).
Harder, D.R., Campbell, W.B. & Roman, R.J. Role of cytochrome P-450 enzymes and metabolites of arachidonic acid in the control of vascular tone. J. Vasc. Res. 32, 79–92 (1995).
Wang, M.H. et al. Cytochrome P450-derived arachidonic acid metabolism in the rat kidney: characterization of selective inhibitors. J. Pharmacol. Exp. Ther. 284, 966–973 (1998).
Peng, X. et al. Suppression of cortical functional hyperemia to vibrissal stimulation in the rat by epoxygenase inhibitors. Am. J. Physiol. Heart Circ. Physiol. 283, H2029–H2037 (2002).
Gibson, C.L., Coughlan, T.C. & Murphy, S.P. Glial nitric oxide and ischemia. Glia 50, 417–426 (2005).
Yang, G., Chen, G., Ebner, T.J. & Iadecola, C. Nitric oxide is the predominant mediator of cerebellar hyperemia during somatosensory activation in rats. Am. J. Physiol. 277, R1760–R1770 (1999).
Cotrina, M.L. et al. Connexins regulate calcium signaling by controlling ATP release. Proc. Natl. Acad. Sci. USA 95, 15735–15740 (1998).
Dunwiddie, T.V., Diao, L. & Proctor, W.R. Adenine nucleotides undergo rapid, quantitative conversion to adenosine in the extracellular space in rat hippocampus. J. Neurosci. 17, 7673–7682 (1997).
Dunwiddie, T.V. & Masino, S.A. The role and regulation of adenosine in the central nervous system. Annu. Rev. Neurosci. 24, 31–55 (2001).
Yermakova, A.V., Rollins, J., Callahan, L.M., Rogers, J. & O'Banion, M.K. Cyclooxygenase-1 in human Alzheimer and control brain: quantitative analysis of expression by microglia and CA3 hippocampal neurons. J. Neuropathol. Exp. Neurol. 58, 1135–1146 (1999).
Sakabe, T. & Siesjo, B.K. The effect of indomethacin on the blood flow-metabolism couple in the brain under normal, hypercapnic and hypoxic conditions. Acta Physiol. Scand. 107, 283–284 (1979).
Bruhn, H., Fransson, P. & Frahm, J. Modulation of cerebral blood oxygenation by indomethacin: MRI at rest and functional brain activation. J. Magn. Reson. Imaging 13, 325–334 (2001).
Bakalova, R.A., Matsuura, T. & Kanno, I. Cyclooxygenase-pathway participates in the regulation of regional cerebral blood flow in response to neuronal activation under normo- and hypercapnia. Prostaglandins Leukot. Essent. Fatty Acids 67, 379–388 (2002).
Parri, H.R., Gould, T.M. & Crunelli, V. Spontaneous astrocytic Ca2+ oscillations in situ drive NMDAR-mediated neuronal excitation. Nat. Neurosci. 4, 803–812 (2001).
Anderson, C.M. & Nedergaard, M. Astrocyte-mediated control of cerebral microcirculation. Trends Neurosci. 26, 340–344; author reply 344–345 (2003).
del Zoppo, G.J. & Hallenbeck, J.M. Advances in the vascular pathophysiology of ischemic stroke. Thromb. Res. 98, 73–81 (2000).
Vavilala, M.S., Lee, L.A. & Lam, A.M. Cerebral blood flow and vascular physiology. Anesthesiol. Clin. N. Am. 20, 247–264 (2002).
Smith, C.D. et al. Altered brain activation in cognitively intact individuals at high risk for Alzheimer's disease. Neurology 53, 1391–1396 (1999).
Grossman, M. et al. Neural basis for verb processing in Alzheimer's disease: an fMRI study. Neuropsychology 17, 658–674 (2003).
Rombouts, S.A., Barkhof, F., Van Meel, C.S. & Scheltens, P. Alterations in brain activation during cholinergic enhancement with rivastigmine in Alzheimer's disease. J. Neurol. Neurosurg. Psychiatry 73, 665–671 (2002).
Niwa, K. et al. Cerebrovascular autoregulation is profoundly impaired in mice overexpressing amyloid precursor protein. Am. J. Physiol. Heart Circ. Physiol. 283, H315–H323 (2002).
Zlokovic, B.V. Neurovascular mechanisms of Alzheimer's neurodegeneration. Trends Neurosci. 28, 202–208 (2005).
Davalos, D. et al. ATP mediates rapid microglial response to local brain injury in vivo. Nat. Neurosci. (2005).
Hademenos, G.J., Massoud, T.F. & Vinuela, F. A biomathematical model of intracranial arteriovenous malformations based on electrical network analysis: theory and hemodynamics. Neurosurgery 38, 1005–1015 (1996).
Wang, X. et al. P2X7 receptor inhibition improves recovery after spinal cord injury. Nat. Med. 10, 821–827 (2004).
Acknowledgements
We thank M. Lauritzen, B. Zlokovic and L. Bekar for comments on the manuscript; M. Schwartzman and K. O'Banion for discussions; and J.R. Falck for supplying us with MS-PPOH. This work was sponsored by the US National Institutes of Health and National Institute of Neurological Disorders and Stroke.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Rhod-2/am staining co-localizes with GFP expression in the cortex of a transgenic mouse expressing green fluorescent protein (GFP) under the GFAP promotor. (PDF 1092 kb)
Supplementary Fig. 2
Spontaneous astrocytic Ca2+ oscillations as a function of 2-photon laser power and inter-frame interval. (PDF 469 kb)
Supplementary Video 1
Photolysis of caged Ca2+ compound caused Ca2+ increase in astrocytic endfeet and vasodilation of a penetrating artery. Time-course images of cross-section of an artery were taken at 15 sec interval. The movie was presented as 5 fps. Astrocytes were loaded with the Ca2+ indicator dye, rhod-2/am (red) and DMNP-EDTA/am, whereas the vasculature was stained with FITC-dextran (green). (MOV 292 kb)
Supplementary Video 2
Vessel diameter and endfeet Ca2+ did not change in the absence of stimulation. Time-course images of cross-section of an artery were taken at 15 sec interval. The movie was presented as 5 fps. Astrocytes were loaded with the Ca2+ indicator dye, rhod-2/am (red) and DMNP-EDTA/am, whereas the vasculature was stained with FITC-dextran (green). (MOV 249 kb)
Supplementary Video 3
Photostimulation failed to trigger Ca2+ increases or vasodilation in mice not loaded with caged Ca2+. Time-course images of cross-section of an artery were taken at 15 sec interval. The movie was presented as 5 fps. Astrocytes were loaded with the Ca2+ indicator dye, rhod-2/am (red) but not DMNP-EDTA/am, whereas the vasculature was stained with FITC-dextran (green). (MOV 257 kb)
Supplementary Video 4
Photolysis of caged Ca2+ triggered Ca2+ increase in an astrocytic endfoot adjacent to a vein, but failed to evoke vasodilation. Time-course images of cross-section of an artery were taken at 15 sec interval. The movie was presented as 5 fps. Astrocytes were loaded with the Ca2+ indicator dye, rhod-2/am (red) and DMNP-EDTA/am, whereas the vasculature was stained with FITC-dextran (green). (MOV 351 kb)
Supplementary Video 5
Photolysis of caged Ca2+ triggered Ca2+ increase in astrocytic endfeet but not vasodilation of artery in the presence of indomethacin (500 μM). Time-course images of cross-section of an artery were taken at 15 sec interval. The movie was presented as 5 fps. Astrocytes were loaded with the Ca2+ indicator dye, rhod-2/am (red) and DMNP-EDTA/am, whereas the vasculature was stained with FITC-dextran (green). (MOV 268 kb)
Supplementary Video 6
Photolysis of caged Ca2+ caused Ca2+ increase in astrocytic endfeet but not arterial vasodilation in the presence of SC-560 (500 μM). Time-course images of cross-section of an artery were taken at 15 sec interval. The movie was presented as 5 fps. Astrocytes were loaded with the Ca2+ indicator dye, rhod-2/am (red) and DMNP-EDTA/am, whereas the vasculature was stained with FITC-dextran (green). (MOV 243 kb)
Rights and permissions
About this article
Cite this article
Takano, T., Tian, GF., Peng, W. et al. Astrocyte-mediated control of cerebral blood flow. Nat Neurosci 9, 260–267 (2006). https://doi.org/10.1038/nn1623
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1623
This article is cited by
-
Astrocytes modulate cerebral blood flow and neuronal response to cocaine in prefrontal cortex
Molecular Psychiatry (2024)
-
Transplantation of A2 type astrocytes promotes neural repair and remyelination after spinal cord injury
Cell Communication and Signaling (2023)
-
Astroglial Hmgb1 regulates postnatal astrocyte morphogenesis and cerebrovascular maturation
Nature Communications (2023)
-
Molecular machines stimulate intercellular calcium waves and cause muscle contraction
Nature Nanotechnology (2023)
-
Aging reduces calreticulin expression and alters spontaneous calcium signals in astrocytic endfeet of the mouse dorsolateral striatum
npj Aging (2023)