Abstract
A central unresolved problem in research on Alzheimer disease is the nature of the molecular entity causing dementia. Here we provide the first direct experimental evidence that a defined molecular species of the amyloid-β protein interferes with cognitive function. Soluble oligomeric forms of amyloid-β, including trimers and dimers, were both necessary and sufficient to disrupt learned behavior in a manner that was rapid, potent and transient; they produced impaired cognitive function without inducing permanent neurological deficits. Although β-amyloidosis has long been hypothesized to affect cognition, the abnormally folded protein species associated with this or any other neurodegenerative disease has not previously been isolated, defined biochemically and then specifically characterized with regard to its effects on cognitive function. The biochemical isolation of discrete amyloid-β moieties with pathophysiological properties sets the stage for a new approach to studying the molecular mechanisms of cognitive impairment in Alzheimer disease and related neurodegenerative disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hardy, J. & Allsop, D. Amyloid deposition as the central event in the aetiology of Alzheimer's disease. Trends Pharmacol. Sci. 12, 383–388 (1991).
Selkoe, D. The molecular pathology of Alzheimer's disease. Neuron 6, 487–498 (1991).
Price, J.L., Davis, P.B., Morris, J.C. & White, D.L. The distribution of tangles, plaques and related immunohistochemical markers in healthy aging and Alzheimer's disease. Neurobiol. Aging 12, 295–312 (1991).
Lorenzo, A. & Yankner, B.A. β-amyloid neurotoxicity requires fibril formation and is inhibited by congo red. Proc. Natl. Acad. Sci. USA 91, 12243–12247 (1994).
Cummings, B.J. & Cotman, C.W. Image analysis of β-amyloid load in Alzheimer's disease and relation to dementia severity. Lancet 346, 1524–1528 (1995).
McLean, C.A. et al. Soluble pool of Aβ amyloid as a determinant of severity of neurodegeneration in Alzheimer's disease. Ann. Neurol. 46, 860–866 (1999).
Naslund, J. et al. Correlation between elevated levels of amyloid β-peptide in the brain and cognitive decline. J. Am. Med. Assoc. 283, 1571–1577 (2000).
Pike, C.J., Walencewicz, A.J., Glabe, C.G. & Cotman, C.W. Aggregation-related toxicity of synthetic β-amyloid protein in hippocampal cultures. Eur. J. Pharmacol. 207, 367–368 (1991).
Irizarry, M.C., McNamara, M., Fedorchak, K., Hsiao, K. & Hyman, B.T. APPSw transgenic mice develop age-related A β deposits and neuropil abnormalities, but no neuronal loss in CA1. J. Neuropathol. Exp. Neurol. 56, 965–973 (1997).
Irizarry, M.C. et al. Aβ deposition is associated with neuropil changes, but not with overt neuronal loss in the human amyloid precursor protein V717F (PDAPP) transgenic mouse. J. Neurosci. 17, 7053–7059 (1997).
Moechars, D., Lorent, K., De Strooper, B., Dewachter, I. & Van Leuven, F. Expression in brain of amyloid precursor protein mutated in the α-secretase site causes disturbed behavior, neuronal degeneration and premature death in transgenic mice. EMBO J. 15, 1265–1274 (1996).
Westerman, M.A. et al. The relationship between Aβ and memory in the Tg2576 mouse model of Alzheimer's disease. J. Neurosci. 22, 1858–1867 (2002).
Terry, R.D. et al. Physical basis of cognitive alterations in Alzheimer's disease: synapse loss is the major correlate of cognitive impairment. Ann. Neurol. 30, 572–580 (1991).
Dickson, D.W. et al. Correlations of synaptic and pathological markers with cognition of the elderly. Neurobiol. Aging 16, 285–298 (1995).
Katzman, R. et al. Clinical, pathological, and neurochemical changes in dementia: a subgroup with preserved mental status and numerous neocortical plaques. Ann. Neurol. 23, 138–144 (1988).
Delaere, P. et al. Large amounts of neocortical β A4 deposits without neuritic plaques nor tangles in a psychometrically assessed, non-demented person. Neurosci. Lett. 116, 87–93 (1990).
Stern, E.A. et al. Cortical synaptic integration in vivo is disrupted by amyloid-β plaques. J. Neurosci. 24, 4535–4540 (2004).
Walsh, D.M. et al. Naturally secreted oligomers of amyloid β protein potently inhibit hippocampal long-term potentiation in vivo. Nature 416, 535–539 (2002).
Wang, H.W. et al. Soluble oligomers of β amyloid (1-42) inhibit long-term potentiation but not long-term depression in rat dentate gyrus. Brain Res. 924, 133–140 (2002).
Wang, Q., Walsh, D.M., Rowan, M., Selkoe, D. & Anwyl, R. β-amyloid induced inhibition of LTP induction involves activation of mGluR5 and the kinases JNK, cdk5 and p38 MAPK. J. Neurosci. 24, 3370–3378 (2004).
Dudai, Y. Memory from A to Z: Keywords, Concepts and Beyond (Oxford Univ. Press, 2002).
Hsia, A.Y. et al. Plaque-independent disruption of neural circuits in Alzheimer's disease mouse models. Proc. Natl. Acad. Sci. USA 96, 3228–3233 (1999).
Kotilinek, L.A. et al. Reversible memory loss in a mouse transgenic model of Alzheimer's disease. J. Neurosci. 22, 6331–6335 (2002).
Dodart, J.C. et al. Immunization reverses memory deficits without reducing brain Aβ burden in Alzheimer's disease model. Nat. Neurosci. 5, 452–457 (2002).
Walsh, D.M., Tseng, B.P., Rydel, R.E., Podlisny, M.B. & Selkoe, D.J. The oligomerization of amyloid β-protein begins intracellularly in cells derived from human brain. Biochemistry 39, 10831–10839 (2000).
Weldon, D.T., O'Hare, E., Kuskowski, M.A., Cleary, J. & Mach, J.R. Jr. Alternating lever cyclic-ratio schedule analysis of the effects of atropine sulfate. Pharmacol. Biochem. Behav. 54, 753–757 (1996).
O'Hare, E. et al. Utilization of a novel model of food reinforced behavior involving neuropeptide Y, insulin, 2-deoxy-D-glucose and naloxone. Behav. Pharmacol. 7, 742–753 (1996).
Richardson, R.L. et al. Behavioural and histopathological analyses of ibuprofen treatment on the effect of aggregated Aβ(1-42) injections in the rat. Brain Res. 954, 1–10 (2002).
Pedersen, E.S., Krishnan, R., Harding, J.W. & Wright, J.W. A role for the angiotensin AT4 receptor subtype in overcoming scopolamine induced spatial memory deficits. Regul. Pept. 102, 147–156 (2001).
Mishima, K. et al. The scopolamine-induced impairment of spatial cognition parallels the acetylcholine release in the ventral hippocampus in rats. Jpn. J. Pharmacol. 84, 163–173 (2000).
Klein, W.L., Krafft, G.A. & Finch, C.E. Targeting small Aβ oligomers: the solution to an Alzheimer's disease conundrum? Trends Neurosci. 24, 219–224 (2001).
Selkoe, D.J. Alzheimer's disease is a synaptic failure. Science 298, 789–791 (2002).
Walsh, D.M., Hartley, D.M. & Selkoe, D.J. The many faces of Aβ: structures and function. Curr. Med. Chem. Immunol. Endocr. Metab. Agents 3, 277–291 (2003).
O'Hare, E. et al. Delayed behavioral effects following intrahippocampal injection of aggregated A β (1-42). Brain Res. 815, 1–10 (1999).
Cleary, J., Hittner, J.M., Semotuk, M., Mantyh, P. & O'Hare, E. β-amyloid(1-40) effects on behavior and memory. Brain Res. 682, 69–74 (1995).
Frautschy, S.A. et al. Phenolic anti-inflammatory antioxidant reversal of Aβ-induced cognitive deficits and neuropathology. Neurobiol. Aging 22, 993–1005 (2001).
Sweeney, W.A., Luedtke, J., McDonald, M.P. & Overmier, J.B. Intrahippocampal injections of exogenous β-amyloid induce postdelay errors in an eight-arm radial maze. Neurobiol. Learn. Mem. 68, 97–101 (1997).
McDonald, M.P., Dahl, E.E., Overmier, J.B., Mantyh, P. & Cleary, J. Effects of an exogenous β-amyloid peptide on retention for spatial learning. Behav. Neural Biol. 62, 60–67 (1994).
Geula, C. et al. Aging renders the brain vulnerable to amyloid β-protein neurotoxicity. Nat. Med. 4, 827–831 (1998).
Bookheimer, S.Y. et al. Patterns of brain activation in people at risk for Alzheimer's disease. N. Engl. J. Med. 343, 450–456 (2000).
Small, G.W. et al. Cerebral metabolic and cognitive decline in persons at genetic risk for Alzheimer's disease. Proc. Natl. Acad. Sci. USA 97, 6037–6042 (2000).
Chen, G. et al. A learning deficit related to age and β-amyloid plaques in a mouse model of Alzheimer's disease. Nature 408, 975–979 (2000).
Koistinaho, M. et al. Specific spatial learning deficits become severe with age in β-amyloid precursor protein transgenic mice that harbor diffuse β-amyloid deposits but do not form plaques. Proc. Natl. Acad. Sci. USA 98, 14675–14680 (2001).
Palop, J.J. et al. Neuronal depletion of calcium-dependent proteins in the dentate gyrus is tightly linked to Alzheimer's disease-related cognitive deficits. Proc. Natl. Acad. Sci. USA 100, 9572–9577 (2003).
Hsiao, K. et al. Correlative memory deficits, Aβ elevation, and amyloid plaques in transgenic mice. Science 274, 99–102 (1996).
Johnson-Wood, K. et al. Amyloid precursor protein processing and A β42 deposition in a transgenic mouse model of Alzheimer disease. Proc. Natl. Acad. Sci. USA 94, 1550–1555 (1997).
Acknowledgements
We thank S. Lesné, J. Huard and M. Podlisny for critical discussions. We are grateful to J. Fadeeva and V. Betts for technical assistance. These studies were funded by the Minnesota Medical Foundation (K.H.A.), the Mayo Medical Foundation (K.H.A.), the US National Institutes of Health (K.H.A., D.J.S.) and the Wellcome Trust (D.M.W.). K.H.A. is a member of the Scientific Advisory Board of Acumen Pharmaceuticals.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Fig. 1
Running response rates at each ratio response requirement value. Running response rate is the rate of responding on either lever, in responses per minute, occurring while a subject is actively pressing levers. Time spent eating and post-reinforcement pauses are not counted in this activity measure. Running response rate is a direct function of the size of the response requirement. No significant differences in running response rate were found under ICV injections of 7PA2 CM (triangles) or A βoligomers (squares) when tested against baseline running response rates (diamonds) at any ratio value. Overall response rates (total responses / session time) were also not significantly different (data not shown). Brackets are 1 SEM. (PDF 219 kb)
Supplementary Fig. 2
(A) 7PA2 and CHO cells were metabolically labelled with 35S-methionine for 16 h and the CM collected, cleared of cells and used for R1282 IP and autoradiography. Only p3, A β monomer, dimers and trimers are specifically detected in 7PA2, and not CHO CM. (B) Full gel view of our original Fig. 1 (A). 7PA2 and CHO CM were IP’d with R1282, the IP stored and the supernatant IP’d a second time. The first and second IPs were then examined by Western blotting using 6E10. As in (A), A β monomer, dimers and trimers are specifically detected in 7PA2, and not CHO CM. (PDF 248 kb)
Supplementary Fig. 3
(A) Incubation of 7PA2 CM in CSF at 37°C for 150? minutes results in no significant decrease in 7PA2-derived monomeric Aβ. 7PA2 cells were metabolically labeled with 35S-methionine for 16 h and the conditioned medium (CM) collected and cleared of cells. This CM was used as a source of labeled Aβ: aliquots of 35S-met 7PA2 CM (1 ml) were added to human cerebrospinal fluid (CSF) (20 ml) in the presence of proteases inhibitors and incubated at 4°C or else incubated without protease inhibitors at 37°C. After 160 min the CM/CSF mixtures were then IP’d with R1282 and the immunoprecipitate electrophoresed on 10-20% tricine gels and subjected to autoradiography. (B Incubation of CSF at 37°C for 150 minutes results in no significant decrease in endogenous monomeric Aβ. Human (4 ml) or ovine (2.5 ml) CSF was incubated at either 4°C or 37°C for 150 min and monomeric Aβ content determined by immunoprecipitation with R1282 and subsequent Western blotting with 6E10. Artificial CSF (ACSF), which does not contain Aβ was used as a negative control (PDF 453 kb)
Rights and permissions
About this article
Cite this article
Cleary, J., Walsh, D., Hofmeister, J. et al. Natural oligomers of the amyloid-β protein specifically disrupt cognitive function. Nat Neurosci 8, 79–84 (2005). https://doi.org/10.1038/nn1372
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1372
This article is cited by
-
Early onset diagnosis in Alzheimer’s disease patients via amyloid-β oligomers-sensing probe in cerebrospinal fluid
Nature Communications (2024)
-
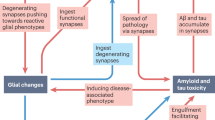
Synaptic degeneration in Alzheimer disease
Nature Reviews Neurology (2023)
-
A common pathway for detergent-assisted oligomerization of Aβ42
Communications Biology (2023)
-
Upregulation of extracellular proteins in a mouse model of Alzheimer’s disease
Scientific Reports (2023)
-
A phase 1b randomized clinical trial of CT1812 to measure Aβ oligomer displacement in Alzheimer’s disease using an indwelling CSF catheter
Translational Neurodegeneration (2023)