Abstract
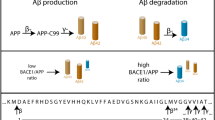
The genetic lesion underlying familial British dementia (FBD), an autosomal dominant neurodegenerative disorder, is a T–A transversion at the termination codon of the BRI gene. The mutant gene encodes BRI-L, the precursor of ABri peptides that accumulate in amyloid deposits in FBD brain. We now report that both BRI-L and its wild-type counterpart, BRI, were constitutively processed by the proprotein convertase, furin, resulting in the secretion of carboxyl-terminal peptides that encompass all or part of ABri. Elevated levels of peptides were generated from the mutant BRI precursor. Electron microscopic studies revealed that synthetic ABri peptides assembled into irregular, short fibrils. Collectively, our results support the view that enhanced furin-mediated processing of mutant BRI generates fibrillogenic peptides that initiate the pathogenesis of FBD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Plant, G. T., Revesz, T., Barnard, R. O., Harding, A. E. & Gautier-Smith, P. C. Familial cerebral amyloid angiopathy with nonneuritic amyloid plaque formation. Brain 113, 721–747 (1990).
Worster-Drought, C., Greenfield, J. G. & McMenemey, W. H. A form of familial presenile dementia with spastic paralysis (including the pathological examination of a case). Brain 63, 237–254 (1940).
Griffiths, R.A., Mortimer, T. F., Oppenheimer, D. R. & Spalding, J. M. Congophilic angiopathy of the brain: a clinical and pathological report on two siblings. J. Neurol. Neurosurg. Psychiatry 45, 396–408 (1982).
Vidal, R. et al. A stop-codon mutation in the BRI gene associated with familial British dementia. Nature 399, 776–781 (1999).
Nakayama, K. Furin: a mammalian subtilisin/Kex2p-like endoprotease involved in processing of a wide variety of precursor proteins. Biochem. J. 327, 625–635 (1997).
Molloy, S. S., Anderson, E. D., Jean, F. & Thomas, G. Bi-cycling the furin pathway: from TGN localization to pathogen activation and embryogenesis. Trends Cell Biol. 9, 28–35 (1999).
Pittois, K., Deleersnijder, W. & Merregaert, J. cDNA sequence analysis, chromosomal assignment and expression pattern of the gene coding for integral membrane protein 2B. Gene 217, 141–149 (1998).
Deleersnijder, W. et al. Isolation of markers for chondro-osteogenic differentiation using cDNA library subtraction. Molecular cloning and characterization of a gene belonging to a novel multigene family of integral membrane proteins. J. Biol. Chem. 271, 19475–19482 (1996).
Kyte, J. & Doolittle, R. F. A simple method for displaying the hydropathic character of a protein. J. Mol. Biol. 157, 105–132 (1982).
Claros, M. G. & von Heijne, G. TopPred II: an improved software for membrane protein structure predictions. Comput. Appl. Biosci. 10, 685–686 (1994).
Balch, W. E. & Rothman, J. E. Characterization of protein transport between successive compartments of the Golgi apparatus: asymmetric properties of donor and acceptor activities in a cell free system. Arch. Biochem. Biophys. 240, 413–425 (1985).
Evan, G. I., Lewis, G. K., Ramsay, G. & Bishop, J. M. Isolation of monoclonal antibodies specific for human c-myc proto-oncogene product. Mol. Cell Biol. 5, 3610–3616 (1985).
von Koch, C. S. et al. Generation of APLP2 KO mice and early postnatal lethality in APLP2/APP double KO mice. Neurobiol. Aging 18, 661–669 (1997).
Watanabe, T. et al. Sequence requirements for precursor cleavage within the constitutive secretory pathway. J. Biol. Chem. 267, 8270–8274 (1992).
Molloy, S. S., Bresnahan, P. A., Leppla, S. H., Klimpel, K. R. & Thomas, G. Human furin is a calcium-dependent serine endoprotease that recognizes the sequence Arg-X-X-Arg and efficiently cleaves anthrax toxin protective antigen. J. Biol. Chem. 267, 16396–16402 (1992).
Watanabe, T., Murakami, K. & Nakayama, K. Positional and additive effects of basic amino acids on processing of precursor proteins within the constitutive secretory pathway. FEBS Lett. 320, 215–218 (1993).
Takahashi, S., Hatsuzawa, K., Watanabe, T., Murakami, K. & Nakayama, K. Sequence requirements for endoproteolytic processing of precursor proteins by furin: transfection and in vitro experiments. J. Biochem. (Tokyo) 116, 47–52 (1994).
Moehring, J. M. & Moehring, T. J. Strains of CHO-K1 cells resistant to Pseudomonas exotoxin A and cross-resistant to diphtheria toxin and viruses. Infect. Immun. 41, 998–1009 (1983).
Moehring, J. M., Inocencio, N. M., Robertson, B. J. & Moehring, T. J. Expression of mouse furin in a Chinese hamster cell resistant to Pseudomonas exotoxin A and viruses complements the genetic lesion. J. Biol. Chem. 268, 2590–2594 (1993).
Spence, M. J., Sucic, J. F., Foley, B. T. & Moehring, T. J. Analysis of mutations in alleles of the fur gene from an endoprotease-deficient Chinese hamster ovary cell strain. Somat. Cell Mol. Genet. 21, 1–18 (1995).
Duguay, S. J., Milewski, W. M., Young, B. D., Nakayama, K. & Steiner, D. F. Processing of wild-type and mutant proinsulin-like growth factor-IA by subtilisin-related proprotein convertases. J. Biol. Chem. 272, 6663–6670 (1997).
Sisodia, S. S., Koo, E. H., Beyreuther, K., Unterbeck, A. & Price, D. L. Evidence that beta-amyloid protein in Alzheimer's disease is not derived by normal processing. Science 248, 492–495 (1990).
Yang, M., Ellenberg, J., Bonicacino, J. S. & Weissman, A. M. The transmembrane domain of a carboxyl-terminal anchored protein determines localization to the endoplasmic reticulum. J. Biol. Chem. 272, 1970–1975 (1997).
Pan, C. J., Lin, B. & Chou, J. Y. Transmembrane topology of glucose-6-phosphatase. J. Biol. Chem. 273, 6144–6148 (1998).
Wang, R., Sweeney, D., Gandy, S. E. & Sisodia, S. S. The profile of soluble amyloid beta protein in cultured cell media. Detection and quantification of amyloid beta protein and variants by immunoprecipitation-mass spectrometry. J. Biol. Chem. 271, 31894–31902 (1996).
Acknowledgements
The authors thank Anthony A. Lanahan and Paul F. Worley for pRK5 vector, Steve Duguay for the pCMV-furin construct and Thomas Moehring for RPE.40 cells. The authors also thank Takeshi Iwatsubo (University of Tokyo) for discussions. This study was supported by National Institute of Health Grants AG14248 (S.S.S.) and 5 T32 GM07281 (D.J.G.), The Alzheimer's Association (S.M., IIRG # 98 134 and R.W.) and The Stasia Borsuk Memorial Fund (R.W.; RG1-96-070).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, SH., Wang, R., Gordon, D. et al. Furin mediates enhanced production of fibrillogenic ABri peptides in familial British dementia. Nat Neurosci 2, 984–988 (1999). https://doi.org/10.1038/14783
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/14783
This article is cited by
-
The emerging role of furin in neurodegenerative and neuropsychiatric diseases
Translational Neurodegeneration (2022)
-
First identification of ITM2B interactome in the human retina
Scientific Reports (2021)
-
The role of the integral type II transmembrane protein BRI2 in health and disease
Cellular and Molecular Life Sciences (2021)
-
BRI2 as an anti-Alzheimer gene
Medical Molecular Morphology (2019)
-
Biased and unbiased perceptual decision-making on vocal emotions
Scientific Reports (2017)