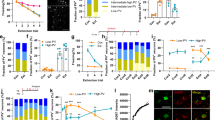
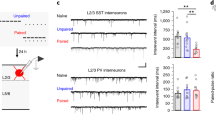
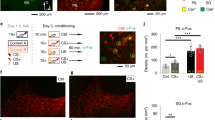
Abstract
Fear conditioning involves the induction of long-term potentiation (LTP) of excitatory synaptic transmission in the lateral amygdala, a brain structure which is tightly controlled by GABAergic inhibition. Here we show that dopamine gates the induction of LTP in the mouse lateral amygdala by suppressing feedforward inhibition from local interneurons. Our findings provide a cellular mechanism for the dopaminergic modulation of fear conditioning and indicate that suppression of feedforward inhibition represents a key mechanism for the induction of associative synaptic plasticity in the lateral amygdala.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
LeDoux, J.E. Emotion circuits in the brain. Annu. Rev. Neurosci. 23, 155–184 (2000).
Maren, S. Neurobiology of pavlovian fear conditioning. Annu. Rev. Neurosci. 24, 897–931 (2001).
Bliss, T.V.P. & Collingridge, G.L. A synaptic model of memory: long-term potentiation in the hippocampus. Nature 361, 31–39 (1993).
Blair, H.T., Schafe, G.E., Bauer, E.P., Rodrigues, S.M. & LeDoux, J.E. Synaptic plasticity in the lateral amygdala: a cellular hypothesis of fear conditioning. Learn. Mem. 8, 229–242 (2001).
Collins, D.R. & Paré, D. Differential fear conditioning induces reciprocal changes in the sensory responses of lateral amygdala neurons to the CS+ and CS−. Learn. Mem. 7, 97–103 (2000).
Inglis, F.M. & Moghaddam, B. Dopaminergic innervation of the amygdala is highly responsive to stress. J. Neurochem. 72, 1088–1094 (1999).
Guarracci, F.A., Frohardt, F.J. & Kapp, B.S. Amygdaloid D1 dopamine receptor involvement in Pavlovian fear conditioning. Brain Res. 82, 28–40 (1999).
Guarracci, F.A., Frohardt, F.J., Falls, W.A. & Kapp, B.S. The effects of intraamygdaloid infusions of a D2 dopamine receptor antagonist on Pavlovian fear conditioning. Behav. Neurosci. 114, 262–272 (2000).
Greba, Q. & Kokkinidis, L. Peripheral and intraamygdalar administration of the dopamine D1 receptor antagonist SCH23390 blocks fear-potentiated startle but not shock reactivity or the shock sensitization of acoustic startle. Behav. Neurosci. 114, 262–272 (2001).
Greba, Q., Gifkins, A. & Kokkinidis, L. Inhibition of amygdaloid dopamine D2 receptors impairs emotional learning measured with fear-potentiated startle. Brain Res. 899, 218–226 (2001).
Rosenkranz, J.A. & Grace, A.A. Dopamine-mediated modulation of odour-evoked amygdala potentials during pavlovian conditioning. Nature 417, 282–287 (2002).
Brinley-Reed, M. & McDonald, A.J. Evidence that dopaminergic axons provide a dense innervation of specific neuronal subpopulations in the rat basolateral amygdala. Brain Res. 850, 127–135 (1999).
Sugita, S., Tanaka, E. & North, R.A. Membrane properties and synaptic potentials of three types of neurones in the rat lateral amygdala. J. Physiol. (Lond.) 460, 705–718 (1993).
Li, X.F., Armony, J.L. & LeDoux, J.E. GABAA and GABAB receptors differentially regulate synaptic transmission in the auditory thalamo-amygdala pathway: An in vivo microiontophoretic study and a model. Synapse 24, 115–124 (1996).
Lang, E.J. & Paré, D. Similar inhibitory processes dominate the responses of cat lateral amygdaloid projection neurons to their various afferents. J. Neurophysiol. 77, 341–352 (1997).
Weisskopf, M.G., Bauer, E.P. & LeDoux, J.E. L-type voltage-gated calcium channels mediate NMDA-independent associative long-term potentiation at thalamic input synapses to the amygdala. J. Neurosci. 19, 10512–10519 (1999).
Rogan, M.T., Staubli, U.V. & LeDoux, J.E. Fear conditioning induces associative long-term potentiation in the amygdala. Nature 390, 604–607 (1997).
McKernan, M.G. & Shinnick-Gallagher, P. Fear conditioning induces a lasting potentiation of synaptic currents in vitro. Nature 390, 607–611 (1997).
Szinyei, C., Heinbockel, T., Montagne, J. & Pape, H.C. Putative cortical and thalamic inputs elicit convergent excitation in a population of GABAergic interneurons of the lateral amygdala. J. Neurosci. 20, 8909–8915 (2000).
Pouille, F. & Scanziani, M. Enforcement of temporal fidelity in pyramidal cells by somatic feed-forward inhibition. Science 293, 1159–1163 (2001).
Rainnie, D.G., Asprodini, E.K. & Shinnick-Gallagher, P. Excitatory transmission in the basolateral amygdala. J. Neurophysiol. 66, 986–998 (1991).
Sjöström, P.J., Turrigiano, G.G. & Nelson, S.B. Rate, timing, and cooperativity jointly determine cortical synaptic plasticity. Neuron 32, 1149–1164 (2001).
Stuart, G.J. & Häusser, M. Dendritic coincidence detection of EPSPs and action potentials. Nature Neurosci. 4, 63–71 (2001).
Rosenkranz, J.A. & Grace, A.A. Modulation of basolateral amygdala neuronal firing and afferent drive by dopamine receptor activation in vivo. J. Neurosci. 19, 11027–11039 (1999).
Washburn, M.S. & Moises, H.C. Electrophysiological and morphological properties of rat basolateral amygdaloid neurons in vitro. J. Neurosci. 12, 4066–4079 (1992).
Rainnie, D.G., Asprodini, E.K. & Shinnick-Gallagher, P. Intracellular recordings from morphologically identified neurons of the basolateral amygdala. J. Neurophysiol. 69, 1350–1362 (1993).
McDonald, A.J. Neurons of the lateral and basolateral amygdaloid nuclei: a Golgi study in the rat. J. Comp. Neurol. 212, 293–312 (1982).
Lang, E.J. & Paré, D. Synaptic responsiveness of interneurons of the cat lateral amygdaloid nucleus. Neuroscience 83, 877–889 (1998).
Paré, D. & Gaudreau, H. Projection cells and interneurons of the lateral and basolateral amygdala: distinct firing patterns and differential relation to theta and delta rhythms in conscious cats. J. Neurosci. 16, 3334–3350 (1996).
Paré, D. & Collins, D.R. Neuronal correlates of fear in the lateral amygdala: multiple extracellular recordings in conscious cats. J. Neurosci. 20, 2701–2710 (2000).
Davis, M. The role of the amygdala in conditioned and unconditioned fear and anxiety. in The Amygdala (ed. Aggleton, J.P.) 231–288 (Oxford Univ. Press, Oxford, UK, 2000).
Seamans, J.K., Gorelova, N., Durstewitz, D. & Yang, C.R. Bidirectional dopamine modulation of GABAergic inhibition in prefrontal cortical pyramidal neurons. J. Neurosci. 21, 3628–3638 (2001).
McDonald, A.J. & Mascagni, F. Colocalization of calcium-binding proteins and gamma-aminobutyric acid in neurons of the rat basolateral amygdala. Neuroscience 105, 681–693 (2001).
Kemppainen, S. & Pitkänen, A. Distribution of parvalbumin, calretinin, and calbindin-D(28k) immunoreactivity in the rat amygdaloid complex and colocalization with gamma-aminobutyric acid. J. Comp. Neurol. 426, 441–467 (2000).
Wigström, H. & Gustafsson, B. Facilitated induction of hippocampal long-lasting potentiation during blockade of inhibition. Nature 301, 603–604 (1983).
Davies, C.H., Starkey, S.J., Pozza, M.F. & Collingridge, G.L. GABAB autoreceptors regulate the induction of LTP. Nature 349, 609–611 (1991).
Yamada, J., Saitow, F., Satake, S., Kiyohara, T. & Konishi, S. GABAB receptor-mediated presynaptic inhibition of glutamatergic and GABAergic transmission in the basolateral amygdala. Neuropharmacology 38, 1743–1753 (1999).
Bauer, E.P., Schafe, G.E. & LeDoux, J.E. NMDA receptors and voltage-gated calcium channels contribute to long-term potentiation and different components of fear memory formation in the lateral amygdala. J. Neurosci. 22, 5239–5249 (2002).
Rosenkranz, J.A. & Grace, A.A. Dopamine attenuates prefrontal cortical suppression of sensory inputs to the basolateral amygdala of rats. J. Neurosci. 21, 4090–4103 (2001).
Nader, K. & LeDoux, J.E. Inhibition of the mesoamygdala dopaminergic pathway impairs the retrieval of conditioned fear associations. Behav. Neurosci. 113, 891–901 (1999).
El-Ghundi, M., O'Dowd, B.F. & George, S.R. Prolonged fear responses in mice lacking dopamine D1 receptor. Brain Res. 892, 86–93 (2001).
Heidbreder, C.A. et al. Behavioral, neurochemical and endocrinological characterization of the early social isolation syndrome. Neuroscience 100, 749–768 (2000).
Tessitore, A. et al. Dopamine modulates the response of the human amygdala: a study in Parkinson's disease. J. Neurosci. 22, 9099–9103 (2002).
Klimek, V., Schenck, J.E., Han, H., Stockmeier, C.A. & Ordway, G.A. Dopaminergic abnormalities in amygdaloid nuclei in major depression: a postmortem study. Biol. Psychiatry 52, 740–748 (2002).
Anderson, W.W. & Collingridge, G.L. The LTP program: A data acquisition program for on-line analysis of long-term potentiation and other synaptic events. J. Neurosci. Methods 108, 71–83 (2001).
Acknowledgements
We are grateful to B. Gähwiler, M. Scanziani and E. Seifritz for helpful discussions and comments on the manuscript. Supported by the Borderline Personality Disorder Research Foundation and the Swiss National Science Foundation (3100-067749.02).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Bissière, S., Humeau, Y. & Lüthi, A. Dopamine gates LTP induction in lateral amygdala by suppressing feedforward inhibition. Nat Neurosci 6, 587–592 (2003). https://doi.org/10.1038/nn1058
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1058
This article is cited by
-
Dopamine projections to the basolateral amygdala drive the encoding of identity-specific reward memories
Nature Neuroscience (2024)
-
Dendrites help mitigate the plasticity-stability dilemma
Scientific Reports (2023)
-
Plasticity of neuronal dynamics in the lateral habenula for cue-punishment associative learning
Molecular Psychiatry (2023)
-
Closed-loop brain stimulation augments fear extinction in male rats
Nature Communications (2023)
-
Insights into the Involvement and Therapeutic Target Potential of the Dopamine System in the Posttraumatic Stress Disorder
Molecular Neurobiology (2023)