Abstract
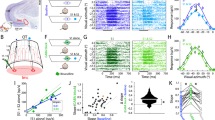
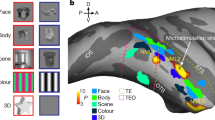
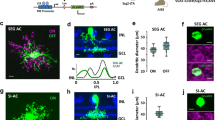
The inferotemporal cortex (area TE) of monkeys, a higher station of the visual information stream for object recognition, contains neurons selective for particular object features. Little is known about how and where this selectivity is generated. We show that blockade of inhibition mediated by γ-aminobutyric acid (GABA) markedly altered the selectivity of TE neurons by augmenting their responses to some stimuli but not to others. The effects were observed for particular groups of stimuli related to the originally effective stimuli or those that did not originally excite the neurons but activated nearby neurons. Intrinsic neuronal interactions within area TE thus determine the final characteristic of their selectivity, and GABAergic inhibition contributes to this process.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Felleman, D. J. & Van Essen, D. C. Distributed hierarchical processing in the primate cerebral cortex. Cereb. Cortex 1, 1–47 ( 1991).
Hubel, D. H. & Wiesel, T. N. Receptive fields and functional architecture of monkey striate cortex. J. Physiol. (Lond.) 195, 215–243 (1968).
Gross, C. G. in Handbook of Sensory Physiology Vol. VII, Part 3B (ed. Jung, R.) 451–482 (Springer, Berlin, 1972).
Tanaka, K. Inferotemporal cortex and object vision. Annu. Rev. Neurosci. 19, 109–139 (1996).
Logothetis, N. K. & Sheinberg, D. L. Visual object recognition. Annu. Rev. Neurosci. 19, 577 –621 (1996).
Hubel, D. H. & Wiesel, T. N. Receptive fields, binocular interaction and functional architecture of the cat's visual cortex. J. Physiol. (Lond.) 160, 106–154 ( 1962).
Vidyasagar, T. R., Pei, X. & Volgushev, M. Multiple mechanisms underlying the orientation selectivity of visual cortical neurons. Trends Neurosci. 19, 272–277 (1996).
Sompolinsky, H. & Shapley, R. New perspectives on the mechanisms for orientation selectivity. Curr. Opin. Neurobiol. 7, 514–522 ( 1997).
Sato, H., Katsuyama, N., Tamura, H., Hata, Y. & Tsumoto, T. Mechanisms underlying orientation selectivity of neurons in the primary visual cortex of the macaque. J. Physiol. (Lond.) 494, 757–771 (1996).
Crook, J. M., Kisvárday, Z. F. & Eysel, U. T. Evidence for a contribution of lateral inhibition to orientation tuning and direction selectivity in cat visual cortex: reversible inactivation of functionally characterized sites combined with neuroanatomical tracing techniques. Eur. J. Neurosci. 10, 2056–2075 (1998).
Tsumoto, T., Eckart, W. & Creutzfeldt, O. D. Modification of orientation sensitivity of cat visual cortex neurons by removal of GABA-mediated inhibition. Exp. Brain. Res. 34, 351–363 ( 1979).
Sillito, A. M., Kemp, J. A., Milson, J. A. & Berardi, N. A re-evaluation of the mechanisms underlying simple cell orientation selectivity . Brain Res. 194, 517–520 (1980).
Hendry, S. H. C., Schwark, H. D., Jones, E. G. & Yan, J. Number and proportions of GABA-immunoreactive neurons in different areas of monkey cerebral cortex. J. Neurosci. 7, 1503–1519 (1987).
Tanigawa, H., Fujita, I., Kato, M. & Ojima, H. Distribution, morphology, and γ-aminobutyric acid immunoreactivity of horizontally projecting neurons in the macaque inferior temporal cortex. J. Comp. Neurol. 401, 129–143 ( 1998).
Fujita, I., Tanaka, K., Ito, M. & Cheng, K. Columns for visual features of objects in monkey inferotemporal cortex. Nature 360, 342–346 (1992).
Wang, G., Tanaka, K. & Tanifuji, M. Optical imaging of functional organization in the monkey inferotemporal cortex. Science 272, 1665 –1668 (1996).
Fujita, I. & Fujita, T. Intrinsic connections in the macaque inferior temporal cortex. J. Comp. Neurol. 368, 467–486 (1996).
Tanaka, K., Saito, H. –A., Fukada, Y. & Moriya, M. Coding visual images of objects in the inferotemporal cortex of the macaque monkey. J. Neurophysiol. 66, 170– 189 (1991).
Kobatake, E. & Tanaka, K. Neuronal selectivities to complex object features in the ventral visual pathway of the macaque cerebral cortex . J. Neurophysiol. 71, 856– 867 (1994).
Sato, H., Katsuyama, N., Tamura, H., Hata, Y. & Tsumoto, T. Broad-tuned chromatic inputs to color-selective neurons in the monkey visual cortex. J. Neurophysiol. 72, 163–168 ( 1994).
Sato, H., Katsuyama, N., Tamura, H., Hata, Y. & Tsumoto, T. Mechanisms underlying direction selectivity of neurons in the primary visual cortex of the macaque. J. Neurophysiol. 74, 1382–1394 (1995).
Gochin, P., Miller, E. K., Gross, C. G. & Gerstein, G. L. Functional interactions among neurons in inferior temporal cortex. Exp. Brain Res. 84, 505–516 (1991).
Gawne, T. & Richmond, B. How independent are the messages carried by adjacent inferior temporal cortical neurons? J. Neurosci. 13, 2758–2771 ( 1993).
Li, B.-M., Mei, Z.-T. & Kubota, K. Multibarreled glass-coated tungsten microelectrode for both neuronal activity recording and iontophoresis in monkeys. Neurosci. Res. 8, 214–219 (1990).
Merrill., E. & Ainsworth, A. Glass-coated platinum-plated tungsten microelectrodes. Med. Biol. Eng. 10, 662 –672 (1972).
Fujita, I. & Konishi, M. The role of GABAergic inhibition in processing of interaural time difference in the owl's auditory system. J. Neurosci. 11, 722–739 (1991).
Acknowledgements
We thank Hiroshi Tamura for comments on the manuscript. This work was supported by a CREST grant from the Japan Science and Technology Corporation, and grants from the Japanese Ministry of Education, Science, Sports and Culture. In the earlier stages of this work, Y.W. was supported by a fellowship from the Uehara Memorial Foundation, and Y.M. by a fellowship from the Japan Society for the Promotion of Science for Young Scientists.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Y., Fujita, I. & Murayama, Y. Neuronal mechanisms of selectivity for object features revealed by blocking inhibition in inferotemporal cortex. Nat Neurosci 3, 807–813 (2000). https://doi.org/10.1038/77712
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/77712
This article is cited by
-
GABAergic and cholinergic modulation of repetition suppression in inferior temporal cortex
Scientific Reports (2018)
-
Increased Early Sensitivity to Eyes in Mouthless Faces: In Support of the LIFTED Model of Early Face Processing
Brain Topography (2018)
-
Auditory experience-dependent cortical circuit shaping for memory formation in bird song learning
Nature Communications (2016)
-
Basal forebrain activation controls contrast sensitivity in primary visual cortex
BMC Neuroscience (2013)
-
Age-related delay in information accrual for faces: Evidence from a parametric, single-trial EEG approach
BMC Neuroscience (2009)