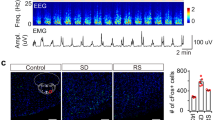
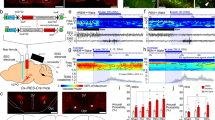
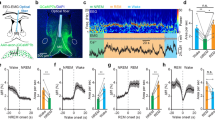
Abstract
An unknown aspect of behavioral state regulation is how the circadian oscillator of the suprachiasmatic nucleus (SCN) regulates sleep and waking. In this report, we describe the necessary elements for a circuit that provides circadian regulation of arousal. Trans-synaptic retrograde tracing revealed a prominent indirect projection from the SCN to the noradrenergic nucleus locus coeruleus (LC), a brain arousal system. Double-labeling experiments revealed several possible links between the SCN and the LC, including the dorsomedial (DMH) and paraventricular hypothalamic nuclei (PVN), as well as medial and ventrolateral pre-optic areas. Lesion studies confirmed that the DMH is a substantial relay in this circuit. Next, neurophysiology experiments revealed circadian variations in LC impulse activity. Lesions of the DMH eliminated these circadian changes in LC activity, confirming the functionality of the SCN–DMH–LC circuit. These results reveal mechanisms for regulation of circadian and sleep–waking functions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Klein, D. C., Moore, R. Y. & Reppert, S. M. Suprachiasmatic Nucleus–The Mind's Clock (Oxford Univ. Press, New York, 1991).
Gillette, M. U. & Tischkau, S. A. Suprachiasmatic nucleus: the brain's circadian clock. Recent Prog. Horm. Res. 54, 33–58 (1999).
Steriade, M. & McCarley, R. W. Brainstem Control of Wakefulness and Sleep (Plenum, New York, London, 1990).
Sherin, J. E., Shiromani, P. J., Mc Carley, R. W. & Saper, C. B. Activation of ventrolateral preoptic neurons during sleep. Science 271, 216–219 (1996).
Edgar, D. M., Dement, W. C. & Fuller, C. A. Effect of SCN lesions on sleep in squirrel monkeys: evidence for opponent processes in sleep-wake regulation. J. Neurosci. 13, 1065–1079 (1993).
Achermann, P. & Borbely, A. A. Simulation of daytime vigilance by the additive interaction of a homeostatic and a circadian process. Biol. Cybern. 71, 115–121 (1994).
Hobson, J. A., McCarley, R. W. & Wyzinski, P. W. Sleep cycle oscillation: reciprocal discharge by two brainstem neuronal groups. Science 189, 55–58 (1975).
Aston-Jones, G. & Bloom, F. E. Activity of norepinephrine-containing locus coeruleus neurons in behaving rats anticipates fluctuations in the sleep-waking cycle. J. Neurosci. 1, 876–886 (1981).
Berridge, C. W. & Foote, S. L. Effects of locus coeruleus activation on electroencephalographic activity in the neocortex and hippocampus. J. Neurosci. 11, 3135–3145 (1991).
Card, J. P. et al. Pseudorabies virus infection of the rat central nervous system: ultrastructural characterization of viral replication, transport, and pathogenesis. J. Neurosci. 13, 2515–2539 (1993).
Chen, S., Ming, X., Miselis, R. & Aston-Jones, G. Characterization of transsynaptic tracing with central application of Pseudorabies virus. Brain Res. 838, 171–183 (1999).
Aston-Jones, G. & Card, J. P. Use of Pseudorabies virus to delineate multisynaptic circuits in brain: opportunities and limitations. J. Neurosci. Methods 103, 51–61 (2000).
Jasmin, L., Burkey, A. R., Card, J. P. & Basbaum, A. I. Transneuronal labeling of a nociceptive pathway, the spino- (trigeminal-) parabrachio-amygdaloid, in the rat. J. Neurosci. 17, 3751–3765 (1997).
O'Donnell, P. Interconnected parallel circuits between rat nucleus accumbens and thalamus revealed by retrograde transynaptic transport of pseudorabies virus. J. Neurosci. 17, 2143–2167 (1997).
Usher, M., Cohen, J. D., Rajkowski, J., Kubiak, P. & Aston-Jones, G. The role of locus coeruleus in the regulation of cognitive performance. Science 283, 549–554 (1999).
Aston-Jones, G., Rajkowski, J. & Cohen, J. Locus coeruleus and regulation of behavioral flexibility and attention. Prog. Brain Res. 126, 165–182 (2000).
Johnson, M. P. et al. Short-term memory, alertness and performance: a reappraisal of their relationship to body temperature. J. Sleep Res. 1, 24–29 (1992).
Aston-Jones, G., Ennis, M., Pieribone, V. A., Nickell, W. T. & Shipley, M. T. The brain nucleus locus coeruleus: restricted afferent control of a broad efferent network. Science 234, 734–737 (1986).
Luppi, P.-H., Aston-Jones, G., Akaoka, H., Chouvet, G. & Jouvet, M. Afferent projections to the rat locus coeruleus demonstrated by retrograde and anterograde tracing with cholera-toxin B subunit and Phaseolus vulgaris leucoagglutinin. Neuroscience 65, 119–160 (1995).
Aston-Jones, G., Shipley, M. T. & Grzanna, R. in The Rat Nervous System (ed. Paxinos, G.) 183–214 (Academic, New York, 1995).
Watts, A. G., Swanson, L. W. & Sanchez-Watts, G. Efferent projections of the suprachiasmatic nucleus: I. studies using anterograde transport of Phaseolus vulgaris leucoagglutinin in the rat. J. Comp. Neurol. 258, 204–229 (1987).
Bernardis, L. L. & Bellinger, L. L. The dorsomedial hypothalamic nucleus revisited: 1998 update. Proc. Soc. Exp. Biol. Med. 218, 284–306 (1998).
Bernardis, L. L. Ventromedial and dorsomedial hypothalamic syndromes in the weanling rat: is the “center” concept really outmoded? Brain Res. Bull. 14, 537–549 (1985).
Card, J. P., Levitt, P. & Enquist, L. W. Different patterns of neuronal infection after intracerebral injection of two strains of pseudorabies virus. J. Virol. 72, 4434–4441 (1998).
Billig, I., Foris, J., Card, J. P. & Yates, B. J. Transneuronal tracing of neural pathways controlling an abdominal muscle, rectus abdominus, in the ferret. Brain Res. 820, 31–44 (1999).
Loewy, A. D. Forebrain nuclei involved in autonomic control. Prog. Brain Res. 87, 253–268 (1991).
Morin, L. P., Goodless-Sanchez, N., Smale, L. & Moore, R. Y. Projections of the suprachiasmatic nuclei, subparaventricular zone and retrochiasmatic area in the golden hamster. Neuroscience 61, 391–410 (1994).
Buijs, R. M. The anatomical basis for the expression of circadian rhythms: the efferent projections of the suprachiasmatic nucleus. Prog. Brain Res. 111, 229–240 (1996).
Chemelli, R. M. et al. Narcolepsy in orexin knockout mice: molecular genetics of sleep regulation. Cell 98, 437–451 (1999).
Lin, L. et al. The sleep disorder canine narcolepsy is caused by a mutation in the hypocretin (orexin) receptor 2 gene. Cell 98, 365–376 (1999).
Peyron, C. et al. Neurons containing hypocretin (orexin) project to multiple neuronal systems. J. Neurosci. 18, 9996–10015 (1998).
Sakurai, T. et al. Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell 92, 573–585 (1998).
Horvath, T. L. et al. Strong hypocretin (orexin) innervation of the locus coeruleus activates noradrenergic cells. J. Comp. Neurol. 415, 145–159 (1999).
Ivanov, A. & Aston-Jones, G. Hypocretin/orexin depolarizes and decreases potassium conductance in locus coeruleus neurons. Neuroreport 11, 1755–1758 (2000).
Hagan, J. J. et al. Orexin A activates locus coeruleus cell firing and increases arousal in the rat. Proc. Natl. Acad. Sci. USA 96, 10911–10916 (1999).
Semba, J.-I., Toru, M. & Mataga, N. Twenty-four hour rhythms of norepinephrine and serotonin in nucleus suprachiasmaticus, raphe nuclei, and locus coeruleus in the rat. Sleep 7, 211–218 (1984).
Foote, S. L., Bloom, F. E. & Aston-Jones, G. Nucleus locus ceruelus: new evidence of anatomical and physiological specificity. Physiol. Rev. 63, 844–914 (1983).
Berridge, C. W. & Foote, S. L. Enhancement of behavioral and electroencephalographic indices of waking following stimulation of noradrenergic B-receptors within the medial septal region of the basal forebrain. J. Neurosci. 16, 6999–7009 (1996).
Berridge, C. W. & Espana, R. A. Synergistic sedative effects of noradrenergic a1 and b-receptor blockade in forebrain electroencephalographic and behavioral indices. Neuroscience 99, 495–505 (2000).
Weitzman, E. D. Sleep and its disorders. Annu. Rev. Neurosci. 4, 381–417 (1981).
Wirz-Justice, A. & Van den Hoofdakker, R. H. Sleep deprivation in depression: what do we know, where do we go? Biol. Psychiatry 46, 445–453 (1999).
Robbins, T. W. & Everitt, B. J. in Psychopharmacology: The Fourth Generation of Progress (eds. Kupfer, D. J. & Bloom, F. E.) 363–372 (Raven, New York, 1995).
Devauges, V. & Sara, S. J. Memory retrieval enhancement by locus coeruleus stimulation: evidence for mediation by beta-receptors. Behav. Brain Res. 43, 93–97 (1991).
Aghajanian, G. K. & Gallager, D. W. Raphe origin of serotonergic nerve terminating in the cerebral ventricles. Brain Res. 88, 221–231 (1975).
Chen, S. & Aston-Jones, G. Extensive projections from the midbrain periaqueductal gray to the caudal ventrolateral medulla: A retrograde and anterograde tracing study in the rat. Neuroscience 71, 443–459 (1996).
Jodo, E., Chiang, C. & Aston-Jones, G. Potent excitatory influence of prefrontal cortex activity on noradrenergic locus coeruleus neurons. Neuroscience 83, 63–80 (1998).
Ennis, M. & Aston-Jones, G. Activation of locus coeruleus from nucleus paragigantocellularis: a new excitatory amino acid pathway in brain. J. Neurosci. 8, 3644–3657 (1988).
Swanson, L. W. Brain Maps: Structure of the Rat Brain (Elsevier, Amsterdam, 1992).
Acknowledgements
We thank R. Miselis and J. Druhan for advice and comments on the manuscript. We also thank E. Haggerty for comments as well as for assistance with monitoring locomotor activity. This work was supported by PHS grant NS24698.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aston-Jones, G., Chen, S., Zhu, Y. et al. A neural circuit for circadian regulation of arousal. Nat Neurosci 4, 732–738 (2001). https://doi.org/10.1038/89522
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/89522
This article is cited by
-
The cognitive impact of light: illuminating ipRGC circuit mechanisms
Nature Reviews Neuroscience (2024)
-
Light modulates task-dependent thalamo-cortical connectivity during an auditory attentional task
Communications Biology (2023)
-
A clock-dependent brake for rhythmic arousal in the dorsomedial hypothalamus
Nature Communications (2023)
-
Nonlinear changes in pupillary attentional orienting responses across the lifespan
GeroScience (2023)
-
Effects of Pharmacological Intervention on Recovery After Sevoflurane Anesthesia in Children: a Network Meta-analysis of Randomized Controlled Trials
Molecular Neurobiology (2023)