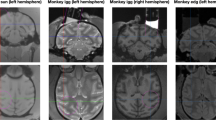
Abstract
The nature of the quantitative relationship between single-neuron recordings in monkeys and functional magnetic resonance imaging (fMRI) measurements in humans is crucial to understanding how experiments in these different species are related, yet it remains undetermined. We measured brain activity in humans attending to moving visual stimuli, using blood oxygenation level-dependent (BOLD) fMRI. Responses in V5 showed a strong and highly linear dependence on increasing strength of motion signal (coherence). These population responses in human V5 had a remarkably simple mathematical relationship to previously observed single-cell responses in macaque V5. We provided an explicit quantitative estimate for the interspecies comparison of single-neuron activity and BOLD population responses. Our data show previously unknown dissociations between the functional properties of human V5 and other human motion-sensitive areas, thus predicting similar dissociations for the properties of single neurons in homologous areas of macaque cortex.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Zeki, S. M. Functional organization of a visual area in the posterior bank of the superior temporal sulcus of the rhesus monkey. J. Physiol. (Lond.) 236, 549–573 (1974).
Allman, J. M. & Kaas, J. H. A crescent-shaped cortical visual area surrounding the middle temporal area (MT) in the owl monkey (Aotus trivirgatus). Brain Res. 81, 199– 213 (1974).
Parker, A. J. & Newsome, W. T. Sense and the single neuron: probing the physiology of perception. Annu. Rev. Neurosci. 21, 227–277 (1998).
Britten, K. H., Shadlen, M. N., Newsome, W. T. & Movshon, J. A. The analysis of visual motion: a comparison of neuronal and psychophysical performance. J. Neurosci. 12, 4745– 4765 (1992).
Newsome, W. T. & Pare, E. B. A selective impairment of motion perception following lesions of the middle temporal visual area (MT). J. Neurosci. 8, 2201–2211 (1988).
Salzman, C. D., Britten, K. H. & Newsome, W. T. Cortical microstimulation influences perceptual judgements of motion direction. Nature 346, 174– 177 (1990).
Britten, K. H., Shadlen, M. N., Newsome, W. T. & Movshon, J. A. Responses of neurons in macaque MT to stochastic motion signals. Vis. Neurosci. 10, 1157–1169 (1993).
Zeki, S. et al. A direct demonstration of functional specialization in human visual cortex. J. Neurosci. 11, 641– 649 (1991).
Tootell, R. B. et al. Functional analysis of human MT and related visual cortical areas using magnetic resonance imaging. J. Neurosci. 15, 3215–3230 (1995).
Kwong, K. K. et al. Dynamic magnetic resonance imaging of human brain activity during primary sensory stimulation. Proc. Natl. Acad. Sci. USA 89, 5675–5679 ( 1992).
Logothetis, N. K., Guggenberger, H., Peled, S. & Pauls, J. Functional imaging of the monkey brain. Nat. Neurosci. 2, 555–562 (1999).
Scannell, J. W. Young, M. P. Neuronal population activity and functional imaging. Proc. R. Soc. Lond. Biol. Sci. 266(1422), 875–881 (1999 ).
Heeger, D. J., Boynton, G. M., Demb, J. B., Seidemann, E. & Newsome, W. T. Motion opponency in visual cortex . J. Neurosci. 19, 7162– 7174 (1999).
Wandell, B. A. Computational neuroimaging of human visual cortex. Annu. Rev. Neurosci. 22, 145–173 ( 1999).
Josephs, O. & Henson, R. N. Event-related functional magnetic resonance imaging: modelling, inference and optimization. Phil. Trans. R. Soc. Lond. B Biol. Sci. 354, 1215– 1228 (1999).
Raymond, J. E. Directional anisotropy of motion sensitivity across the visual field. Vision Res. 34, 1029–1037 (1994).
Buchel, C., Wise, R. J., Mummery, C. J., Poline, J. B. & Friston, K. J. Nonlinear regression in parametric activation studies. Neuroimage 4, 60– 66 (1996).
Watson, J. D. et al. Area V5 of the human brain: evidence from a combined study using positron emission tomography and magnetic resonance imaging. Cereb. Cortex 3, 79–94 ( 1993).
Van Oostende, S., Sunaert, S., Van Hecke, P., Marchal, G. & Orban, G. A. The kinetic occipital (KO) region in man: an fMRI study. Cereb. Cortex 7, 690–701 (1997).
DeYoe, E. A. et al. Mapping striate and extrastriate visual areas in human cerebral cortex. Proc. Natl. Acad. Sci. USA 93, 2382 –2386 (1996).
Zeki, S. The response properties of cells in the middle temporal area (area MT) of owl monkey visual cortex. Proc. R. Soc. Lond. B Biol. Sci. 207, 239–248 (1980).
Malonek, D., Tootell, R. B. & Grinvald, A. Optical imaging reveals the functional architecture of neurons processing shape and motion in owl monkey area MT. Proc. R. Soc. Lond. B Biol. Sci. 258, 109– 119 (1994).
Friston, K. J., Holmes, A. P. & Worsley, K. J. How many subjects constitute a study? Neuroimage 10, 1–5 ( 1999).
Dupont, P. et al. The kinetic occipital region in human visual cortex. Cereb. Cortex 7, 283–292 (1997).
Albright, T. D., Desimone, R. & Gross, C. G. Columnar organization of directionally selective cells in visual area MT of the macaque. J. Neurophysiol. 51, 16–31 (1984).
Maunsell, J. H. & Van Essen, D. C. Functional properties of neurons in middle temporal visual area of the macaque monkey. I. Selectivity for stimulus direction, speed, and orientation. J. Neurophysiol. 49, 1127–1147 (1983).
Albright, T. D. Direction and orientation selectivity of neurons in visual area MT of the macaque. J. Neurophysiol. 52, 1106– 1130 (1984).
Britten, K. H. & Newsome, W. T. Tuning bandwidths for near-threshold stimuli in area MT. J. Neurophysiol. 80, 762–770 (1998).
Adelson, E. H. & Bergen, J. R. Spatiotemporal energy models for the perception of motion. J. Opt. Soc. Am. [A] 2, 284–299 ( 1985).
Simoncelli, E. P. & Heeger, D. J. A model of neuronal responses in visual area MT. Vision Res. 38, 743–761 (1998).
Carter, C. S. et al. Anterior cingulate cortex, error detection, and the online monitoring of performance. Science 280, 747–749 (1998).
Devinsky, O., Morrell, M. J. & Vogt, B. A. Contributions of anterior cingulate cortex to behaviour . Brain 118, 279–306 (1995).
Tootell, R. B. et al. Functional analysis of V3A and related areas in human visual cortex. J. Neurosci. 17, 7060– 7078 (1997).
Scase, M. O., Braddick, O. J. & Raymond, J. E. What is noise for the motion system? Vision Res. 36, 2579–2586 (1996).
Talairach, J. & Tournoux, P. Co-Planar Stereotaxic Atlas of the Human Brain (Georg Thieme Verlag, New York, 1988 ).
Friston, K. J. et al. Spatial realignment and normalisation of images. Hum. Brain Mapp. 2, 165–189 (1995).
Turner, R., Howseman, A., Rees, G. E., Josephs, O. & Friston, K. Functional magnetic resonance imaging of the human brain: data acquisition and analysis. Exp. Brain Res. 123, 5–12 ( 1998).
Friston, K. J. et al. Statistical parametric mapping in functional imaging: a general linear approach. Hum. Brain Mapp. 2, 189 –210 (1995).
Buchel, C., Holmes, A. P., Rees, G. & Friston, K. J. Characterizing stimulus-response functions using nonlinear regressors in parametric fMRI experiments. Neuroimage 8, 140– 149 (1998).
Draper, N. R. & Smith, H. Applied Regression Analysis (Wiley, New York, 1981).
Acknowledgements
This work was supported by the Wellcome Trust, the Keck Foundation, the National Science Foundation and the National Insitutes of Mental Health. We thank K.H. Britten for comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rees, G., Friston, K. & Koch, C. A direct quantitative relationship between the functional properties of human and macaque V5. Nat Neurosci 3, 716–723 (2000). https://doi.org/10.1038/76673
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/76673
This article is cited by
-
Transcranial static magnetic field stimulation over hMT+ inhibits visual motion discriminability
Scientific Reports (2024)
-
Amblyopia: progress and promise of functional magnetic resonance imaging
Graefe's Archive for Clinical and Experimental Ophthalmology (2023)
-
Natural scene sampling reveals reliable coarse-scale orientation tuning in human V1
Nature Communications (2022)
-
A modeling framework for determining modulation of neural-level tuning from non-invasive human fMRI data
Communications Biology (2022)
-
Deficits in the Magnocellular Pathway of People with Reading Difficulties
Current Developmental Disorders Reports (2022)