Abstract
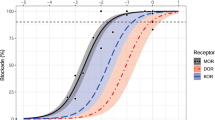
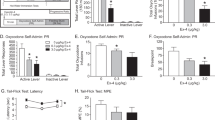
Many peptides and transmitters found within the brain also have peripheral sites of action. We now demonstrate that the brain releases functionally active neurotransmitters/neuromodulators directly from the brain into the blood through a saturable P-glycoprotein (Pgp) transport system. Downregulating Pgp1 expression with antisense reduced the brain-to-blood transport of morphine, β-endorphin and other opioids. Lowering Pgp expression significantly enhanced systemic morphine analgesia and prevented tolerance, but diminished the analgesic activity of centrally administered morphine, implying that supraspinal analgesia resulted from a combination of central and peripheral mechanisms activated by morphine transported from the brain to the blood. Similarly, mice with a disruption of the Mdr1a gene were more sensitive to systemic morphine and less sensitive to morphine given centrally. This ability of the Pgp transport system to pump functionally active compounds from the brain to periphery defines a potentially important mechanism for the central nervous system to modulate peripheral systems.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bodnar, R. J. et al. Mediation of anorexia by human, recombinant Tumor Necrosis Factor thorugh a peipheral action in the rat. Cancer Res. 49, 6280–6284 (1989).
Goodman, J. C., Robertson, C. S., Grossman, R. G. & Narayan, R. K. Elevation of tumor necrosis factor in head injury. J. Neuroimmunol. 30, 213–217 (1990).
Romero, L. I., Kakucska, I., Lechan, R. M. & Reichlin, S. Interleukin-6 (IL-6) is secreted from the brain after intracerebroventricular injection of IL-1β in rats. Am. J. Physiol. 270, R518–R524 (1996).
Banks, W. A. & Kastin, A. J. Saturable transport of peptides across the blood–brain barrier. Life Sci. 41, 1319–1338 (1987).
Banks, W. A., Kastin, A. J. & Ehrensing, C. A. Endogenous peptide Tyr-Pro-Trp-Gly-NH2 (Tyr-W-MIF-1) is transported from the brain to the blood by peptide transport system-1. J. Neurosci. Res. 35, 690–695 (1993).
Banks, W. A. & Kastin, A. J. Passage of peptides across the blood–brain barrier: pathophysiological perspectives. Life Sci. 59, 1923–1943 (1996).
Aquilante, C. L., Letrent, S. P., Pollack, G. M. & Brouwer, K. L. R. Increased brain P-glycoprotein in morphine tolerant rats. Life Sci. 66, L47–L51 (1999).
Chen, C. P. & Pollack, G. M. Enhanced antinociception of the model opioid peptide [d-Penicillamine2,5] enkephalin by P-glycoprotein modulation. Pharm. Res. 16, 296–301 (1999).
Egleton, R. D. & Davis, T. P. Transport of the δ-opioid receptor agonist [d-penicillamine2,5] enkephalin across the blood–brain barrier involves transcytosis. J. Pharm. Sci. 88, 392–397 (1999).
Jonker, J. W. et al. Role of blood–brain barrier P-glycoprotein in limiting brain accumulation and sedative side-effects of asimadoline, a peripherally acting analgaesic drug. Br. J. Pharmacol. 127, 43–50 (1999).
Letrent, S. P., Pollack, G. M., Brouwer, K. R. & Brouwer, K. L. R. Effects of a potent and specific P-glycoprotein inhibitor on the blood–brain barrier distribution and antinociceptive effect of morphine in the rat. Drug Metab. Dispos. 27, 827–834 (1999).
Schinkel, A. H.et al. Disruption of the mouse mdrla P-glycoprotein gene leads to a deficiency in the blood–brain barrier and to increased sensitivity to drugs. Cell 77, 491–502 (1994).
Xie, R. J., Hammarlund-Udenaes, M., De Boer, A. G. & De Lange, E. C. M. The role of P-glycoprotein in blood–brain barrier transport of morphine: transcortical microdialysis studies in mdr1a (−/−) and mdr1a (+/+)mice. Br. J. Pharmacol. 128, 563–568 (1999).
Cordon-Cardo, C. et al. Multidrug-resistance gene (P-glycoprotein) is expressed by endothelial cells at blood-barrier sites. Proc. Natl. Acad. Sci. USA 86, 695–698 (1989).
Beaulieu, E., Demeule, M., Ghitescu, L. & Beliveau, R. P-glycoprotein is strongly expressed in the luminal membranes of the endothelium of blood vessels in the brain. Biochemistry 326, 539–544 (1997).
Schinkel, A. H., Wagenarr, E., Van, D. L., Mol, C. A. & Borst, P. Absence of the mdrla P-Glycoprotein in mice affects tissue distribution and pharmacokinetics of dexamethasone, diagoxin and cyclosporin A. J. Clin. Invest. 96, 1698–1705 (1995).
Thompson, S. J., Koszdin, K. & Bernards, C. M. Opiate-induced analgesia is increased and prolonged in mice lacking P-glycoprotein. Anesthesiology 92, 1392–1399 (2000).
Hegmann, E. J., Bauer, H. C. & Kerbel, R. S. Expression and functional activity of P-glycoprotein in cultured cerebral capillary endothelial cells. Cancer Res. 52, 6969–6975 (1992).
Chen, C. P. & Pollack, G. M. Altered disposition and antinociception of [d-Penicillamine2,5] enkephalin in mdr1a-gene-deficient mice. J. Pharmacol. Exp. Ther. 287, 545–552 (1998).
Tatsuta, T., Naito, M., Oh-hara, T., Sugawara, I. & Tsuruo, T. Functional involvement of P-glycoprotein in blood–brain barrier. J. Biol. Chem. 267, 20383–20391 (1992).
Schinkel, A. H. et al. Multidrug resistance and the role of P-glycoprotein knockout mice. Eur. J. Cancer 31A, 1295–1298 (1995).
Schinkel, A. H., Wagenaar, E., Mol, C. A. & Van Deemter, L. P-glycoprotein in the blood–brain barrier of mice influences the brain penetration and pharmacological activity of many drugs. J. Clin. Invest. 97, 25617–22524 (1996).
Regina, A. et al. Mrp1 multidrug resistance-associated protein and P-glycoprotein expression in rat brain microvessel endothelial cells. J. Neurochem. 71, 705–715 (1998).
Standifer, K. M., Chien, C.-C., Wahlestedt, C., Brown, G. P. & Pasternak, G. W. Selective loss of δ opioid analgesia and binding by antisense oligodeoxynucleotides to a δ opioid receptor. Neuron 12, 805–810 (1994).
Pan, Y.-X. et al. Cloning and functional characterization through antisense mapping of a kappa 3-related opioid receptor. Mol. Pharmacol. 47, 1180–1188 (1995).
Standifer, K. M. et al. Antisense oligodeoxynucleotides to the cloned δ receptor, DOR-1: uptake, stability and regulation of gene expression. J. Neurochem. 65, 1981–1987 (1995).
Standifer, K. M., Rossi, G. C. & Pasternak, G. W. Differential blockade of opioid analgesia by antisense oligodeoxynucleotides directed against various G-protein α subunits. Mol. Pharmacol. 50, 293–298 (1996).
Kolesnikov, Y. A. et al. Functionally differentiating two neuronal nitric oxide synthase isoforms through antisense mapping: Evidence for opposing NO actions on morphine analgesia and tolerance. Proc. Natl. Acad. Sci. USA 94, 8220–8225 (1997).
Gao, Z. Q., Gao, Z. P., Fields, J. Z. & Boman, B. M. Co-transfection of MDR1 and MRP antisense RNAs abolishes the drug resistance in multidrug-resistant human lung cancer cells. Anticancer Res. 18, 3073–3076 (1998).
Motomura S. et al. Inhibition of P-glycoprotein and recovery of drug sensitivity of human acute leukemic blast cells by multidrug resistance gene (mdr1) antisense oligonucleotides. Blood 91, 3163–3171 (1998).
Wang, L. W., Chen, L. X., Walker, V. & Jacob, T. J. C. Antisense to MDR1 mRNA reduces P-glycoprotein expression, swelling-activated Cl− current and volume regulation in bovine ciliary epithelial cells. J. Physiol. (Lond.) 511, 33–44 (1998).
Gottesman, M. M., Hrcyna, C. A., Schoenlein, P. V., Germann, U. A. & Pastan, I. Genetic analysis of the multidrug transporter. Annu. Rev. Genet. 29, 607–649 (1995).
Roninson, I. B. in Molecular and Cellular Biology of Multidrug Resistance in Tumor Cells Ch. p (ed. Roninson, I. B.) 189–211 Plenum, New York, 1991).
Kolesnikov, Y. & Pasternak, G. W. Topical opioids in mice: analgesia and reversal of tolerance by a topical N-methyl-d-aspartate antagonist. J. Pharmacol. Exp. Ther. 290, 247–252 (1999).
Kolesnikov, Y. A., Jain, S., Wilson, R. & Pasternak, G. W. Peripheral morphine analgesia: synergy with central sites and a target of morphine tolerance. J. Pharmacol. Exp. Ther. 279, 502–506 (1996).
Pardridge, W. M., Golden, P. L., Kang, Y. S. & Bickel, U. Brain microvascular and astrocyte localization of P-glycoprotein. J. Neurochem. 68, 1278–1285 (1997).
Roerig, S. C., O'Brien, S. M., Fujimoto, J. A. & Wilcox, G. L. Tolerance to morphine analgesia: decreased multiplicative interaction between spinal and supraspinal sites. Brain Res. 308, 360–363 (1984).
Mathis, J. P. et al. Biochemical evidence for orphanin FQ/nociceptin receptor heterogeneity in mouse brain. Biochem. Biophys. Res. Commun. 230, 462–465 (1997).
Pan, Y.-X. et al. Dissociation of affinity and efficacy in KOR-3 chimeras. FEBS Lett. 395, 207–210 (1996).
Rossi, G. C., Pasternak, G. W. & Bodnar, R. J. μ and δ opioid synergy between the periaqueductal gray and the rostro–ventral medulla. Brain Res. 665, 85–93 (1994).
Rossi, G. C., Pasternak, G. W. & Bodnar, R. J. Synergistic brainstem interactions for morphine analgesia. Brain Res. 624, 171–180 (1993).
Ling, G. S. F., Spiegel, K., Lockhart, S. H. & Pasternak, G. W. Separation of opioid analgesia from respiratory depression: evidence for different receptor mechanisms. J. Pharmacol. Exp. Ther. 232, 149–155 (1985).
Ling, G. S. F., Spiegel, K., Nishimura, S. & Pasternak, G. W. Dissociation of morphine's analgesic and respiratory depressant actions. Eur. J. Pharmacol. 86, 487–488 (1983).
Pan, Y.-X., Cheng, J., Xu, J. & Pasternak, G. W. Cloning, expression and classification of a kappa3-related opioid receptor using antisense oligodeoxynucleotides. Regul. Pept. 54, 217–218 (1994).
Rohde, D. S., McKay, W. R., Chang, D. S., Abbadie, C. & Basbaum, A. I. The contribution of supraspinal, peripheral and intrinsic spinal circuits to the pattern and magnitude of Fos-like immunoreactivity in the lumbar spinal cord of the rat withdrawing from morphine. Neuroscience 80, 599–612 (1997).
Rossi, G., Leventhal, L., Bolan, E. A. & Pasternak, G. W. Pharmacological characterization of orphanin FQ/nociceptin and its fragments. J. Pharmacol. Exp. Ther. 282, 858–865 (1997).
Rossi, G. C., Brown, G. P., Leventhal, L., Yang, K. & Pasternak, G. W. Novel receptor mechanisms for heroin and morphine-6β-glucuronide analgesia. Neurosci. Lett. 216, 1–4 (1996).
King, M. A., Rossi, G. C., Chang, A. H., Williams, L. & Pasternak, G. W. Spinal analgesic activity of orphanin FQ/nociceptin and its fragments. Neurosci. Lett. 223, 113–116 (1997).
Kolesnikov, Y. A. & Pasternak, G. W. Peripheral blockade of topical morphine tolerance by ketamine. Eur. J. Pharmacol. 374, R1–R2 (1999).
Tallarida, R. J. & Murray, R. B. Manual of Pharmacological Calculations with Computer Programs (Springer, New York, 1987).
Acknowledgements
This work was supported, in part, by research grants (DA02615 and DA07242) and a Senior Scientist Award (DA00220) to G. W. P. and a training grant (T32DA07274) to M. K. and W. S. from the National Institute on Drug Abuse, and a core grant from the National Cancer Institute (CA08748) to M.S.K.C.C.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
King, M., Su, W., Chang, A. et al. Transport of opioids from the brain to the periphery by P-glycoprotein: peripheral actions of central drugs. Nat Neurosci 4, 268–274 (2001). https://doi.org/10.1038/85115
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/85115
This article is cited by
-
Essential role of P-glycoprotein in the mechanism of action of oliceridine
Neuropsychopharmacology (2023)
-
Synthesis of the Mechanisms of Opioid Tolerance: Do We Still Say NO?
Cellular and Molecular Neurobiology (2021)
-
Candidate gene analyses for acute pain and morphine analgesia after pediatric day surgery: African American versus European Caucasian ancestry and dose prediction limits
The Pharmacogenomics Journal (2019)
-
An observational study examining the effects of a surgically induced inflammatory response on the distribution of morphine and its metabolites into cerebrospinal fluid
Canadian Journal of Anesthesia/Journal canadien d'anesthésie (2017)