Abstract
Noradrenaline modulates global brain states and diverse behaviors through what is traditionally believed to be a homogeneous cell population in the brainstem locus coeruleus (LC). However, it is unclear how LC coordinates disparate behavioral functions. We report a modular LC organization in rats, endowed with distinct neural projection patterns and coding properties for flexible specification of opposing behavioral learning states. LC projection mapping revealed functionally distinct cell modules with specific anatomical connectivity. An amygdala-projecting ensemble promoted aversive learning, while an independent medial prefrontal cortex-projecting ensemble extinguished aversive responses to enable flexible behavior. LC neurons displayed context-dependent inter-relationships, with moderate, discrete activation of distinct cell populations by fear or safety cues and robust, global recruitment of most cells by strong aversive stimuli. These results demonstrate a modular organization in LC in which combinatorial activation modes are coordinated with projection- and behavior-specific cell populations, enabling adaptive tuning of emotional responding and behavioral flexibility.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Sara, S.J. & Bouret, S. Orienting and reorienting: the locus coeruleus mediates cognition through arousal. Neuron 76, 130–141 (2012).
McCall, J.G. et al. CRH engagement of the locus coeruleus noradrenergic system mediates stress-induced anxiety. Neuron 87, 605–620 (2015).
Valentino, R.J. & Van Bockstaele, E. Convergent regulation of locus coeruleus activity as an adaptive response to stress. Eur. J. Pharmacol. 583, 194–203 (2008).
Maren, S. & Holmes, A. Stress and fear extinction. Neuropsychopharmacology 41, 58–79 (2016).
Arnsten, A.F. Stress signalling pathways that impair prefrontal cortex structure and function. Nat. Rev. Neurosci. 10, 410–422 (2009).
Berridge, C.W. & Waterhouse, B.D. The locus coeruleus-noradrenergic system: modulation of behavioral state and state-dependent cognitive processes. Brain Res. Brain Res. Rev. 42, 33–84 (2003).
Aston-Jones, G. & Cohen, J.D. An integrative theory of locus coeruleus-norepinephrine function: adaptive gain and optimal performance. Annu. Rev. Neurosci. 28, 403–450 (2005).
Schwarz, L.A. et al. Viral-genetic tracing of the input-output organization of a central noradrenaline circuit. Nature 524, 88–92 (2015).
Nakamura, S. & Iwama, K. Antidromic activation of the rat locus coeruleus neurons from hippocampus, cerebral and cerebellar cortices. Brain Res. 99, 372–376 (1975).
Nagai, T., Satoh, K., Imamoto, K. & Maeda, T. Divergent projections of catecholamine neurons of the locus coeruleus as revealed by fluorescent retrograde double labeling technique. Neurosci. Lett. 23, 117–123 (1981).
Room, P., Postema, F. & Korf, J. Divergent axon collaterals of rat locus coeruleus neurons: demonstration by a fluorescent double labeling technique. Brain Res. 221, 219–230 (1981).
Foote, S.L., Bloom, F.E. & Aston-Jones, G. Nucleus locus ceruleus: new evidence of anatomical and physiological specificity. Physiol. Rev. 63, 844–914 (1983).
Chandler, D.J., Gao, W.J. & Waterhouse, B.D. Heterogeneous organization of the locus coeruleus projections to prefrontal and motor cortices. Proc. Natl. Acad. Sci. USA 111, 6816–6821 (2014).
Li, Y. et al. Retrograde optogenetic characterization of the pontospinal module of the locus coeruleus with a canine adenoviral vector. Brain Res. 1641, 274–290 (2016).
Bouret, S. & Richmond, B.J. Relation of locus coeruleus neurons in monkeys to Pavlovian and operant behaviors. J. Neurophysiol. 101, 898–911 (2009).
Kalwani, R.M., Joshi, S. & Gold, J.I. Phasic activation of individual neurons in the locus ceruleus/subceruleus complex of monkeys reflects rewarded decisions to go but not stop. J. Neurosci. 34, 13656–13669 (2014).
Kebschull, J.M. et al. High-throughput mapping of single-neuron projections by sequencing of barcoded RNA. Neuron 91, 975–987 (2016).
McCall, J.G. et al. Locus coeruleus to basolateral amygdala noradrenergic projections promote anxiety-like behavior. eLife 6, e18247 (2017).
Bush, D.E., Caparosa, E.M., Gekker, A. & Ledoux, J. Beta-adrenergic receptors in the lateral nucleus of the amygdala contribute to the acquisition but not the consolidation of auditory fear conditioning. Front. Behav. Neurosci. 4, 154 (2010).
Johansen, J.P. et al. Hebbian and neuromodulatory mechanisms interact to trigger associative memory formation. Proc. Natl. Acad. Sci. USA 111, E5584–E5592 (2014).
LeDoux, J.E. Coming to terms with fear. Proc. Natl. Acad. Sci. USA 111, 2871–2878 (2014).
Herry, C. & Johansen, J.P. Encoding of fear learning and memory in distributed neuronal circuits. Nat. Neurosci. 17, 1644–1654 (2014).
LeDoux, J.E. Emotion circuits in the brain. Annu. Rev. Neurosci. 23, 155–184 (2000).
Quirarte, G.L., Galvez, R., Roozendaal, B. & McGaugh, J.L. Norepinephrine release in the amygdala in response to footshock and opioid peptidergic drugs. Brain Res. 808, 134–140 (1998).
Mueller, D., Porter, J.T. & Quirk, G.J. Noradrenergic signaling in infralimbic cortex increases cell excitability and strengthens memory for fear extinction. J. Neurosci. 28, 369–375 (2008).
Hugues, S., Garcia, R. & Léna, I. Time course of extracellular catecholamine and glutamate levels in the rat medial prefrontal cortex during and after extinction of conditioned fear. Synapse 61, 933–937 (2007).
Han, X. et al. A high-light sensitivity optical neural silencer: development and application to optogenetic control of non-human primate cortex. Front. Syst. Neurosci. 5, 18 (2011).
Witten, I.B. et al. Recombinase-driver rat lines: tools, techniques, and optogenetic application to dopamine-mediated reinforcement. Neuron 72, 721–733 (2011).
Murchison, C.F. et al. A distinct role for norepinephrine in memory retrieval. Cell 117, 131–143 (2004).
McGaugh, J.L. The amygdala modulates the consolidation of memories of emotionally arousing experiences. Annu. Rev. Neurosci. 27, 1–28 (2004).
Martins, A.R. & Froemke, R.C. Coordinated forms of noradrenergic plasticity in the locus coeruleus and primary auditory cortex. Nat. Neurosci. 18, 1483–1492 (2015).
Rasmussen, K. & Jacobs, B.L. Single unit activity of locus coeruleus neurons in the freely moving cat. II. Conditioning and pharmacologic studies. Brain Res. 371, 335–344 (1986).
Sara, S.J. & Segal, M. Plasticity of sensory responses of locus coeruleus neurons in the behaving rat: implications for cognition. Prog. Brain Res. 88, 571–585 (1991).
Swanson, L.W. The locus coeruleus: a cytoarchitectonic, Golgi and immunohistochemical study in the albino rat. Brain Res. 110, 39–56 (1976).
Mattis, J. et al. Principles for applying optogenetic tools derived from direct comparative analysis of microbial opsins. Nat. Methods 9, 159–172 (2011).
Do-Monte, F.H., Allensworth, M. & Carobrez, A.P. Impairment of contextual conditioned fear extinction after microinjection of alpha-1-adrenergic blocker Prazosin into the medial prefrontal cortex. Behav. Brain Res. 211, 89–95 (2010).
Soudais, C., Laplace-Builhe, C., Kissa, K. & Kremer, E.J. Preferential transduction of neurons by canine adenovirus vectors and their efficient retrograde transport in vivo. FASEB J. 15, 2283–2285 (2001).
Mahn, M., Prigge, M., Ron, S., Levy, R. & Yizhar, O. Biophysical constraints of optogenetic inhibition at presynaptic terminals. Nat. Neurosci. 19, 554–556 (2016).
Ozawa, T. et al. A feedback neural circuit for calibrating aversive memory strength. Nat. Neurosci. 20, 90–97 (2017).
Warden, M.R. et al. A prefrontal cortex-brainstem neuronal projection that controls response to behavioural challenge. Nature 492, 428–432 (2012).
Uematsu, A., Tan, B.Z. & Johansen, J.P. Projection specificity in heterogeneous locus coeruleus cell populations: implications for learning and memory. Learn. Mem. 22, 444–451 (2015).
Latagliata, E.C., Saccoccio, P., Milia, C. & Puglisi-Allegra, S. Norepinephrine in prelimbic cortex delays extinction of amphetamine-induced conditioned place preference. Psychopharmacology (Berl.) 233, 973–982 (2016).
Yu, A.J. & Dayan, P. Uncertainty, neuromodulation, and attention. Neuron 46, 681–692 (2005).
Schiff, H.C. et al. β-Adrenergic receptors regulate the acquisition and consolidation phases of aversive memory formation through distinct, temporally regulated signaling pathways. Neuropsychopharmacology 42, 895–903 (2017).
Fitzgerald, P.J., Giustino, T.F., Seemann, J.R. & Maren, S. Noradrenergic blockade stabilizes prefrontal activity and enables fear extinction under stress. Proc. Natl. Acad. Sci. USA 112, E3729–E3737 (2015).
Tully, K., Li, Y., Tsvetkov, E. & Bolshakov, V.Y. Norepinephrine enables the induction of associative long-term potentiation at thalamo-amygdala synapses. Proc. Natl. Acad. Sci. USA 104, 14146–14150 (2007).
Faber, E.S. et al. Modulation of SK channel trafficking by beta adrenoceptors enhances excitatory synaptic transmission and plasticity in the amygdala. J. Neurosci. 28, 10803–10813 (2008).
Burgos-Robles, A., Vidal-Gonzalez, I., Santini, E. & Quirk, G.J. Consolidation of fear extinction requires NMDA receptor-dependent bursting in the ventromedial prefrontal cortex. Neuron 53, 871–880 (2007).
Giustino, T.F., Fitzgerald, P.J. & Maren, S. Revisiting propranolol and PTSD: memory erasure or extinction enhancement? Neurobiol. Learn. Mem. 130, 26–33 (2016).
Belkin, M.R. & Schwartz, T.L. Alpha-2 receptor agonists for the treatment of posttraumatic stress disorder. Drugs Context 4, 212286 (2015).
Wickersham, I.R., Finke, S., Conzelmann, K.-K. & Callaway, E.M. Retrograde neuronal tracing with a deletion-mutant rabies virus. Nat. Methods 4, 47–49 (2007).
Mebatsion, T., Konig, M. & Conzelmann, K.K. Budding of rabies virus particles in the absence of the spike glycoprotein. Cell 84, 941–951 (1996).
Finke, S. & Conzelmann, K.K. Virus promoters determine interference by defective RNAs: selective amplification of mini-RNA vectors and rescue from cDNA by a 3′ copy-back ambisense rabies virus. J. Virol. 73, 3818–3825 (1999).
Acknowledgements
We thank M. Iwasaki, A. Umetsu, K. Mori and A. Krejcirikova for excellent technical assistance and H. Hamanaka for setting up rabies virus production in the lab. We thank C. Yokoyama, A. Luthi, H. Schiff, T. Shimogori, S. Fujisawa, T. McHugh, A. Benucci and members of the Johansen lab for comments on earlier versions of the manuscript. We also thank J. Kleinschmidt (German Cancer Research Center) for his gift of the AAV helper vectors and the RIKEN Research Resources Center for help with viral titering. This work was supported by RIKEN SPRP (A.U. and B.Z.T.), KAKENHI 16H01291, 15H04264, 15H01301 (J.P.J.), 16K21620 (B.Z.T.), 26750380, 16H05928 (A.U.)) and the Strategic Research Program for Brain Sciences from the Ministry of Education, Culture, Sports, Science and Technology (11041047, J.P.J.).
Author information
Authors and Affiliations
Contributions
A.U., B.Z.T. and J.P.J. designed the experiments and wrote up the manuscript. A.U., B.Z.T., E.A.Y., J.S.C. and J.K. carried out the experiments. A.U., B.Z.T. and J.P.J. analyzed the results. F.J. and E.J.K. supplied viral reagents before publication and expert advice on their use. I.B.W. and K.D. supplied viral reagents and transgenic animals before publication as well as expert advice on their use.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Recording sites and example waveform clustering, as well as electrophysiological recordings of auditory CS and shock processing following overtraining.
(a) Recording sites in LC for experiments in which cells were held throughout fear conditioning and extinction. (b) Example waveforms and clusters from stereotrode recordings during pre-conditioning, fear learning and extinction experiments. (c) Experimental protocol. In overtraining experiments, all units (24 units) were optogenetically identified before CS, US, and CS-US presentations. (d,e) Perievent time histograms showing auditory CS (d) and shock US (e) evoked firing rate (y-axis) responses in example LC cells. Time is on x-axis with 0=CS or US onset time (CS period denoted by gray bar and US period denoted by orange bar). Bin size=500 ms (d), 200 ms (e). (f) Pie chart showing proportion of cells (n=24) auditory responsive (red, 45.8%), shock responsive cells (yellow inside, 79.2%) and those not exhibiting shock or auditory responding (black). 37.5% of cells responded to both CS and US, 8.3% responded to CS alone and 41.7% responded to US alone. (g) Heat plots showing auditory- (left) and shock-evoked (right) responses in all cells recorded aligned vertically by cell number. X-axis=time, 0 denotes tone CS or shock US onset, top gray bar shows tone CS duration and red bar shows US duration. Bin size=500 ms (left), 200 ms (right).
Supplementary Figure 2 Task-related changes in tonic firing rate and optogenetically identified cell characterization.
(a) Tonic firing rate (20 sec prior to first stimulus in each session) changes across pre-CS, fear conditioning (FC), early and late extinction trials for all LC neurons held across all sessions (n=37) (one-way RM ANOVA, F3, 111 = 0.90, P = 0.37). (b) Tonic firing rate changes for each auditory CS defined cell population during pre-CS, early and late extinction (two-way repeated RM ANOVA; interaction F9,63 = 0.74, P = 0.67; effect of time F3, 63 = 0.62, P = 0.60; effect of neuron type F3, 21 = 0.21, P = 0.89). (c) Baseline firing rate (60 sec prior to first stimulus in each session) changes across pre-CS, fear conditioning (FC), early and late extinction trials for all LC neurons held across all sessions (n=37) (one-way RM ANOVA, F3, 111 = 0.90, P = 0.37). (d) Baseline firing rate changes for each auditory CS defined cell population during pre-CS, early and late extinction (two-way repeated RM ANOVA; interaction F9,63 = 0.87, P = 0.56; effect of time F3, 63 = 1.06, P = 0.37; effect of neuron type F3, 21 = 0.44, P = 0.73). (e) Schematic showing optogenetic identification and electrophysiological recording experimental approach. (f) Example perievent time histogram of cell showing laser inhibition of an LC neuron for optogenetic identification. (g) Pie chart showing proportion of optogenetically identified LC cells recorded across all tasks (n=10) which were classified as pre-CS (gray, 20%), ‘Fear’ (red, 10%), ‘Fear & Extinction’ (purple, 10%), ‘Extinction’ (blue, 30%) or not responsive (black, 30%). Yellow inside chart shows proportion of shock responsive cells in each cell classification. (h) Population averaged auditory CS-evoked responses (y-axis) at pre-CS, early and late extinction for each cell type showing a similar trend to that seen when analyzing all LC cells recorded (see Fig. 2g for comparison). Data represent the mean ± SEM.
Supplementary Figure 3 Identification of high-firing-rate neurons using optogenetic ID of LC noradrenaline neurons in a TH-Cre rat.
(a-d) Representative waveform (a), cluster (b), interspike interval (c), and autocorrelogram (d) of high frequency firing cell. (e) Example of perievent time histogram of optogenetic inhibition in high frequency firing cell. Bin size = 100 ms. (f) Distribution of spontaneous firing rate of all optogenetically identified units in overtraining experiment. 6/24 (data from 5 rats: rat 1=6 cells, rat 2=8 cells, rat 3=7 cells, rat 4=2 cells, rat 5=1 cell) cells are defined as high firing rate based on baseline firing rates of >10 Hz. Optogenetically identified, high firing rate cells were also apparent before any behavioral manipulation (2/10 optogenetically identified cells, data from 4 rats: rat 1=1 cell, rat 2=3 cells, rat 3=4 cells, rat 4=1 cell). (g) Perievent time histograms showing shock US evoked firing rate (y-axis) responses in example LC cell. Time is on x-axis with 0=CS or US onset time (US period denoted by bar). Bin size=200 ms. (h) Pie chart showing proportion of high-frequency firing rate cells (n=6) which were identified as auditory responsive (red, 50.0%), shock responsive (yellow inside, 83.3%) or non-responsive to shocks or auditory stimuli (black). 33.3% of cells responded to both CS and US, 16.6% responded to CS alone and 50.0% responded to US alone.
Supplementary Figure 4 Quantitative differences in LC neuronal responses to aversive shocks vs. sensory predictive cues.
(a) Shock evoked responses do not differ between fear (‘Fear’ and ‘‘Fear+Extinction’ cells) and ‘Extinction’ neurons (t18 = 0.66, P = 0.52, unpaired t-test). Data represent the mean ± SEM. (b) Shock USs activate a significantly larger proportion of LC neurons compared with tone CSs presented during pre-CS, early and late extinction (Cochran’s Q test χ2(3)=37.17, P<0.0001, followed by Wilcoxon sign tests for individual comparisons ****P<0.0001, ***P<0.001, *P<0.05). (c) Shocks evoke a significantly larger response compared with auditory CS-evoked responses in LC neurons (t36 = 4.50, * P < 0.0001).
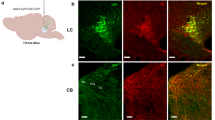
Supplementary Figure 5 Retrograde tracer labeling.
(a) Example of different populations of LA/B- (green) and IL- (blue) projecting LC neurons following injection of retrograde tracer (CTb-647) into IL and RVG-GFP into LA/B (n=3 animals). (b) Same as a except LA/B projecting cells are green (from retrograde CAV2-cre-GFP injections) and IL projecting cells are labeled red (CAV2-DsRed into IL) (n=3 animals). TH is pseudocolored in blue. (c) Example of control experiment in which green and red retrobeads were coinjected into IL resulting in a high degree of overlap in LC neurons (green+red/green=80.1%, green+red/red=81.5%) (n=3 animals). Yellow triangles denote double labeled cells. (d) Example of similar non-overlapping noradrenaline populations in mouse LC. Green and red retrobeads were injected into IL and LA/B respectively. In mice, 9.9% of LA/B projecting neurons overlapped with IL projecting, 4.9% overlap of IL projecting cells with LA/B projecting (n=3 animals). TH is pseudocolored blue. All scale bars=20 μm.
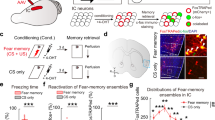
Supplementary Figure 6 Freezing responses in c-Fos experiments.
(a) Paired CS-US group showed high freezing level while unpaired control group showed low freezing to CSs during 5CS retrieval. (b) Paired, but not unpaired, CS-US group extinguished fear memory during extinction session. Data represent the mean ± SEM.
Supplementary Figure 7 Rabies virus expression in the LC.
(a,b) RVG-eArchT3.0-EGFP injected in LA/B (a) or IL (b) resulted in expression of eArchT3.0-EGFP (green cells) in LC-noradrenaline (immunostaining for TH, red) cells. Quantification of double labeled TH+&EGFP+ LC cells/total number of EGFP+ cells in LA/B- and IL-projecting cells. Data represent the mean ± SEM.
Supplementary Figure 8 Effect of inactivation of LA/B- or IL-projecting LC neurons on expression of extinction memories.
(a,b) Inactivation of LA/B (a) or IL (b) projecting LC neurons had no effect on the expression of extinction memories when light illumination occurred during the retrieval test after extinction training. (unpaired students t-tests comparing GFP to Arch groups during at the Retrieval tests in A, t16 = 0.77, P = 0.45 and B, t13 = 0.62, P = 0.54). Data represent the mean ± SEM, and the numbers in parentheses represent the number of samples.
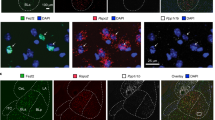
Supplementary Figure 9 Efferent mapping of LA/B- and IL-projecting terminal fields.
(a) Quantification of the pixel density of axon terminal labeling in each area (pixel count in each region/pixel count in total region) for LA/B (red) and IL (green) projecting cells. (b) Density of innervation of LA/B, IL, ACx1 and ACx2 from TH+, LA/B projecting and IL projecting LC neurons. For a and b, parentheses show number of animals used in each group. (c) Confocal images showing examples of synaptophysin-mCherry labeling of axon terminals in IL, LA/B and ACx from TH-cre expressing, LA/B projecting and IL projecting cell populations. PL, prelimbic; ACC, anterior cingulate; OFC, orbitofrontal cortex; ACx, auditory cortex; CeA/MeA, central and medial nuclei of the amygdala; IC, insula; PRh, perirhinal; BNST, bed nucleus of the stria terminalis; dHip/vHip, dorsal/ventral hippocampus; Hpt, hypothalamus; MT, medial thalamus; VPM/L, ventroposterior medial/lateral thalamus; MgM, medial geniculate thalamus; VTA, ventral tegmental area; SN, substantia nigra; v/dPAG, ventral/dorsal periaqueductal gray. For a&b, individual data-points are represented as black dots. Data represent the mean ± SEM, and the numbers in parentheses represent the number of samples. Scale bar=20 μm.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 (PDF 1467 kb)
Rights and permissions
About this article
Cite this article
Uematsu, A., Tan, B., Ycu, E. et al. Modular organization of the brainstem noradrenaline system coordinates opposing learning states. Nat Neurosci 20, 1602–1611 (2017). https://doi.org/10.1038/nn.4642
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.4642
This article is cited by
-
Compromised trigemino-coerulean coupling in migraine sensitization can be prevented by blocking beta-receptors in the locus coeruleus
The Journal of Headache and Pain (2023)
-
Sexual differences in locus coeruleus neurons and related behavior in C57BL/6J mice
Biology of Sex Differences (2023)
-
Noradrenergic projections regulate the acquisition of classically conditioned eyelid responses in wild-type and are impaired in kreisler mice
Scientific Reports (2023)
-
The tyrosine capsid mutations on retrograde adeno-associated virus accelerates gene transduction efficiency
Molecular Brain (2022)
-
Spatiotemporal dynamics of noradrenaline during learned behaviour
Nature (2022)