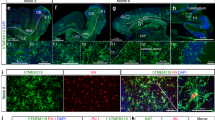
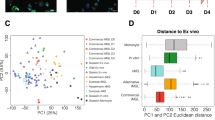
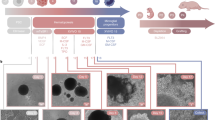
Abstract
Microglia are resident inflammatory cells of the CNS and have important roles in development, homeostasis and a variety of neurologic and psychiatric diseases. Difficulties in procuring human microglia have limited their study and hampered the clinical translation of microglia-based treatments shown to be effective in animal disease models. Here we report the differentiation of human induced pluripotent stem cells (iPSC) into microglia-like cells by exposure to defined factors and co-culture with astrocytes. These iPSC-derived microglia have the phenotype, gene expression profile and functional properties of brain-isolated microglia. Murine iPSC-derived microglia generated using a similar protocol have equivalent efficacy to primary brain-isolated microglia in treatment of murine syngeneic intracranial malignant gliomas. The ability to generate human microglia facilitates the further study of this important CNS cell type and raises the possibility of their use in personalized medicine applications.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Nimmerjahn, A., Kirchhoff, F. & Helmchen, F. Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 308, 1314–1318 (2005).
Davalos, D. et al. ATP mediates rapid microglial response to local brain injury in vivo. Nat. Neurosci. 8, 752–758 (2005).
Cardona, A.E. et al. Control of microglial neurotoxicity by the fractalkine receptor. Nat. Neurosci. 9, 917–924 (2006).
Aguzzi, A., Barres, B.A. & Bennett, M.L. Microglia: scapegoat, saboteur, or something else? Science 339, 156–161 (2013).
Nayak, D., Roth, T.L. & McGavern, D.B. Microglia development and function. Annu. Rev. Immunol. 32, 367–402 (2014).
Hwang, S.Y. et al. Induction of glioma apoptosis by microglia-secreted molecules: the role of nitric oxide and cathepsin B. Biochim. Biophys. Acta 1793, 1656–1668 (2009).
Mora, R. et al. TNF-alpha- and TRAIL-resistant glioma cells undergo autophagy-dependent cell death induced by activated microglia. Glia 57, 561–581 (2009).
Brantley, E.C. et al. Nitric oxide-mediated tumoricidal activity of murine microglial cells. Transl. Oncol. 3, 380–388 (2010).
Chicoine, M.R. et al. The in vivo antitumoral effects of lipopolysaccharide against glioblastoma multiforme are mediated in part by Toll-like receptor 4. Neurosurgery 60, 372–380, discussion 381 (2007).
Sarkar, S. et al. Therapeutic activation of macrophages and microglia to suppress brain tumor-initiating cells. Nat. Neurosci. 17, 46–55 (2014).
Saijo, K. & Glass, C.K. Microglial cell origin and phenotypes in health and disease. Nat. Rev. Immunol. 11, 775–787 (2011).
Gandy, S. & Heppner, F.L. Microglia as dynamic and essential components of the amyloid hypothesis. Neuron 78, 575–577 (2013).
Prinz, M., Priller, J., Sisodia, S.S. & Ransohoff, R.M. Heterogeneity of CNS myeloid cells and their roles in neurodegeneration. Nat. Neurosci. 14, 1227–1235 (2011).
Capotondo, A. et al. Brain conditioning is instrumental for successful microglia reconstitution following hematopoietic stem cell transplantation. Proc. Natl. Acad. Sci. USA 109, 15018–15023 (2012).
Chen, S.K. et al. Hematopoietic origin of pathological grooming in Hoxb8 mutant mice. Cell 141, 775–785 (2010).
Biju, K. et al. Macrophage-mediated GDNF delivery protects against dopaminergic neurodegeneration: a therapeutic strategy for Parkinson's disease. Mol. Ther. 18, 1536–1544 (2010).
Biju, K.C. et al. Bone marrow-derived microglia-based neurturin delivery protects against dopaminergic neurodegeneration in a mouse model of Parkinson's disease. Neurosci. Lett. 535, 24–29 (2013).
Butovsky, O. et al. Modulating inflammatory monocytes with a unique microRNA gene signature ameliorates murine ALS. J. Clin. Invest. 122, 3063–3087 (2012).
Sedgwick, J.D., Ford, A.L., Foulcher, E. & Airriess, R. Central nervous system microglial cell activation and proliferation follows direct interaction with tissue-infiltrating T cell blasts. J. Immunol. 160, 5320–5330 (1998).
Vodyanik, M.A., Thomson, J.A. & Slukvin, I.I. Leukosialin (CD43) defines hematopoietic progenitors in human embryonic stem cell differentiation cultures. Blood 108, 2095–2105 (2006).
Butovsky, O. et al. Identification of a unique TGF-β-dependent molecular and functional signature in microglia. Nat. Neurosci. 17, 131–143 (2014).
Jung, S. et al. Analysis of fractalkine receptor CX(3)CR1 function by targeted deletion and green fluorescent protein reporter gene insertion. Mol. Cell. Biol. 20, 4106–4114 (2000).
Napoli, I., Kierdorf, K. & Neumann, H. Microglial precursors derived from mouse embryonic stem cells. Glia 57, 1660–1671 (2009).
Hambardzumyan, D., Gutmann, D.H. & Kettenmann, H. The role of microglia and macrophages in glioma maintenance and progression. Nat. Neurosci. 19, 20–27 (2016).
Yang, I., Han, S.J., Kaur, G., Crane, C. & Parsa, A.T. The role of microglia in central nervous system immunity and glioma immunology. J. Clin. Neurosci. 17, 6–10 (2010).
Hussain, S.F. et al. The role of human glioma-infiltrating microglia/macrophages in mediating antitumor immune responses. Neuro-oncol. 8, 261–279 (2006).
Van Gool, S. et al. Dendritic cell therapy of high-grade gliomas. Brain Pathol. 19, 694–712 (2009).
Beutner, C. et al. Engineered stem cell-derived microglia as therapeutic vehicle for experimental autoimmune encephalomyelitis. Gene Ther. 20, 797–806 (2013).
Beutner, C., Roy, K., Linnartz, B., Napoli, I. & Neumann, H. Generation of microglial cells from mouse embryonic stem cells. Nat. Protoc. 5, 1481–1494 (2010).
Tsuchiya, T. et al. Characterization of microglia induced from mouse embryonic stem cells and their migration into the brain parenchyma. J. Neuroimmunol. 160, 210–218 (2005).
Eglitis, M.A. & Mezey, E. Hematopoietic cells differentiate into both microglia and macroglia in the brains of adult mice. Proc. Natl. Acad. Sci. USA 94, 4080–4085 (1997).
Ginhoux, F. et al. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330, 841–845 (2010).
Kierdorf, K. et al. Microglia emerge from erythromyeloid precursors via Pu.1- and Irf8-dependent pathways. Nat. Neurosci. 16, 273–280 (2013).
Muffat, J. et al. Efficient derivation of microglia-like cells from human pluripotent stem cells. Nat. Med. 22, 1358–1367 (2016).
Bellin, M., Marchetto, M.C., Gage, F.H. & Mummery, C.L. Induced pluripotent stem cells: the new patient? Nat. Rev. Mol. Cell Biol. 13, 713–726 (2012).
Clement, A.M. et al. Wild-type nonneuronal cells extend survival of SOD1 mutant motor neurons in ALS mice. Science 302, 113–117 (2003).
Merling, R.K. et al. Transgene-free iPSCs generated from small volume peripheral blood nonmobilized CD34+ cells. Blood 121, e98–e107 (2013).
Pripuzova, N.S. et al. Development of a protein marker panel for characterization of human induced pluripotent stem cells (hiPSCs) using global quantitative proteome analysis. Stem Cell Res 14, 323–338 (2015).
Chen, G. et al. Chemically defined conditions for human iPSC derivation and culture. Nat. Methods 8, 424–429 (2011).
Senju, S. et al. Characterization of dendritic cells and macrophages generated by directed differentiation from mouse induced pluripotent stem cells. Stem Cells 27, 1021–1031 (2009).
Acknowledgements
This work was supported by the Intramural Research Programs of the National Institute of Neurological Disorders and Stroke, the National Human Genome Research Institute and the National Institute of Allergy and Infectious Diseases, National Institutes of Health.
Author information
Authors and Affiliations
Contributions
J.K.P. conceived the project; H.P., M.J.S., D.M.I., Y.C., D.B.M. and J.K.P. designed the experiments; H.P., M.J.S., D.M.I., Y.C., A.B.S., G.K. and M.A.B. performed the experiments; H.P., M.J.S., D.B.M. and J.K.P. analyzed the data; K.R.J. performed microarray data analysis; A.G.E., D.M., C.L.S. and S.G. provided technical support; H.L.M. provided conceptual advice; and H.P., M.J.S., D.B.M. and J.K.P. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 The stage 2 co-culture of human iPS-MG and the astrocyte feeder layers were analyzed by staining with ENTPD1 (CD39).
Co-cultures of human iPS-MG and the astrocyte feeder layers were analyzed by staining with ENTPD1 (CD39), a cell surface protein specific for microglia (right). Isotype antibody (left) was used as a control. Data is representative of two different differentiation experiments.
Supplementary Figure 2 Gene-expression data extracted from the human microarray for the six microglia-specific genes.
Our iMG show high expression of all the six microglia genes whereas the macrophages (Mac) and dendritic cells (DC) exhibit high expression of only selected genes. Data is representative of three different differentiation experiments.
Supplementary Figure 3 The stage 2 co-culture of murine iPS-MG and the astrocyte feeder layers were analyzed by GFP+ cell sorting.
The iPS-MG were differentiated from CX3CR1-GFP+ iPSC cells. The CX3CR1 gene is specifically expressed in microglia, and hence the iPS-MG express GFP and can be conveniently separated from the co-culture through FACs sorting. Data is representative of three different differentiation experiments.
Supplementary Figure 4 miPSC cells generated from Cx3cr1Gfp/+ knock-in mouse embryonic fibroblasts demonstrate properties of identity, authenticity and pluripotency.
Cx3cr1gfp/+ iPSCs stain for alkaline phosphatase (a), express the stem cell markers Oct4 (b) and Sox2 (c), form teratomas (d), can be used to generate chimeric mice (e), and display a normal karyotype (f). Data is representative of three different cell-culture for the alkaline phosphatase and immunocytochemistry experiments and 4 mice for the chimera experiments.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4 and Supplementary Table 1 (PDF 6827 kb)
Rights and permissions
About this article
Cite this article
Pandya, H., Shen, M., Ichikawa, D. et al. Differentiation of human and murine induced pluripotent stem cells to microglia-like cells. Nat Neurosci 20, 753–759 (2017). https://doi.org/10.1038/nn.4534
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.4534
This article is cited by
-
An integrated toolkit for human microglia functional genomics
Stem Cell Research & Therapy (2024)
-
Insights for disease modeling from single-cell transcriptomics of iPSC-derived Ngn2-induced neurons and astrocytes across differentiation time and co-culture
BMC Biology (2024)
-
Selection of suitable reference genes for gene expression studies in HMC3 cell line by quantitative real-time RT-PCR
Scientific Reports (2024)
-
Transcriptional characterization of iPSC-derived microglia as a model for therapeutic development in neurodegeneration
Scientific Reports (2024)
-
Recent advances and current challenges of new approach methodologies in developmental and adult neurotoxicity testing
Archives of Toxicology (2024)