Abstract
The basolateral amygdala (BLA) is a site of convergence of negative and positive stimuli and is critical for emotional behaviors and associations. However, the neural substrate for negative and positive behaviors and relationship between negative and positive representations in the basolateral amygdala are unknown. Here we identify two genetically distinct, spatially segregated populations of excitatory neurons in the mouse BLA that participate in valence-specific behaviors and are connected through mutual inhibition. These results identify a genetically defined neural circuit for the antagonistic control of emotional behaviors and memories.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Accession codes
References
Pitkänen, A., Savander, V. & LeDoux, J.E. Organization of intra-amygdaloid circuitries in the rat: an emerging framework for understanding functions of the amygdala. Trends Neurosci. 20, 517–523 (1997).
McDonald, A.J. Neuronal organization of the lateral and basolateral amygdaloid nuclei in the rat. J. Comp. Neurol. 222, 589–606 (1984).
Swanson, L.W. & Petrovich, G.D. What is the amygdala? Trends Neurosci. 21, 323–331 (1998).
Hall, E. The amygdala of the cat: a Golgi study. Z. Zellforsch. Mikrosk. Anat. 134, 439–458 (1972).
McDonald, A.J. Neurons of the lateral and basolateral amygdaloid nuclei: a Golgi study in the rat. J. Comp. Neurol. 212, 293–312 (1982).
Carlsen, J. & Heimer, L. The basolateral amygdaloid complex as a cortical-like structure. Brain Res. 441, 377–380 (1988).
Mascagni, F. & McDonald, A.J. A novel subpopulation of 5-HT type 3A receptor subunit immunoreactive interneurons in the rat basolateral amygdala. Neuroscience 144, 1015–1024 (2007).
McDonald, A.J., Muller, J.F. & Mascagni, F. GABAergic innervation of alpha type II calcium/calmodulin-dependent protein kinase immunoreactive pyramidal neurons in the rat basolateral amygdala. J. Comp. Neurol. 446, 199–218 (2002).
Redondo, R.L. et al. Bidirectional switch of the valence associated with a hippocampal contextual memory engram. Nature 513, 426–430 (2014).
Stuber, G.D. et al. Excitatory transmission from the amygdala to nucleus accumbens facilitates reward seeking. Nature 475, 377–380 (2011).
Ambroggi, F., Ishikawa, A., Fields, H.L. & Nicola, S.M. Basolateral amygdala neurons facilitate reward-seeking behavior by exciting nucleus accumbens neurons. Neuron 59, 648–661 (2008).
Goosens, K.A. & Maren, S. Contextual and auditory fear conditioning are mediated by the lateral, basal, and central amygdaloid nuclei in rats. Learn. Mem. 8, 148–155 (2001).
Cador, M., Robbins, T.W. & Everitt, B.J. Involvement of the amygdala in stimulus-reward associations: interaction with the ventral striatum. Neuroscience 30, 77–86 (1989).
Killcross, S., Robbins, T.W. & Everitt, B.J. Different types of fear-conditioned behaviour mediated by separate nuclei within amygdala. Nature 388, 377–380 (1997).
Gore, F. et al. Neural representations of unconditioned stimuli in basolateral amygdala mediate innate and learned responses. Cell 162, 134–145 (2015).
Paton, J.J., Belova, M.A., Morrison, S.E. & Salzman, C.D. The primate amygdala represents the positive and negative value of visual stimuli during learning. Nature 439, 865–870 (2006).
Zirlinger, M., Kreiman, G. & Anderson, D.J. Amygdala-enriched genes identified by microarray technology are restricted to specific amygdaloid subnuclei. Proc. Natl. Acad. Sci. USA 98, 5270–5275 (2001).
Namburi, P. et al. A circuit mechanism for differentiating positive and negative associations. Nature 520, 675–678 (2015).
Heiman, M. et al. A translational profiling approach for the molecular characterization of CNS cell types. Cell 135, 738–748 (2008).
Knight, Z.A. et al. Molecular profiling of activated neurons by phosphorylated ribosome capture. Cell 151, 1126–1137 (2012).
Sanz, E. et al. Cell-type-specific isolation of ribosome-associated mRNA from complex tissues. Proc. Natl. Acad. Sci. USA 106, 13939–13944 (2009).
Gay, L. et al. Mouse TU tagging: a chemical/genetic intersectional method for purifying cell type-specific nascent RNA. Genes Dev. 27, 98–115 (2013).
Yang, Z., Edenberg, H.J. & Davis, R.L. Isolation of mRNA from specific tissues of Drosophila by mRNA tagging. Nucleic Acids Res. 33, e148 (2005).
Lein, E.S. et al. Genome-wide atlas of gene expression in the adult mouse brain. Nature 445, 168–176 (2007).
Hemmings, H.C. Jr., Greengard, P., Tung, H.Y. & Cohen, P. DARPP-32, a dopamine-regulated neuronal phosphoprotein, is a potent inhibitor of protein phosphatase-1. Nature 310, 503–505 (1984).
Savander, V., Go, C.G., LeDoux, J.E. & Pitkänen, A. Intrinsic connections of the rat amygdaloid complex: projections originating in the basal nucleus. J. Comp. Neurol. 361, 345–368 (1995).
Kantak, K.M., Black, Y., Valencia, E., Green-Jordan, K. & Eichenbaum, H.B. Dissociable effects of lidocaine inactivation of the rostral and caudal basolateral amygdala on the maintenance and reinstatement of cocaine-seeking behavior in rats. J. Neurosci. 22, 1126–1136 (2002).
Choi, G.B. et al. Lhx6 delineates a pathway mediating innate reproductive behaviors from the amygdala to the hypothalamus. Neuron 46, 647–660 (2005).
Root, C.M., Denny, C.A., Hen, R. & Axel, R. The participation of cortical amygdala in innate, odour-driven behaviour. Nature 515, 269–273 (2014).
Peng, Y. et al. Sweet and bitter taste in the brain of awake behaving animals. Nature 527, 512–515 (2015).
Aso, Y. et al. Mushroom body output neurons encode valence and guide memory-based action selection in Drosophila. Elife 3, e04580 (2014).
Cohn, R., Morantte, I. & Ruta, V. Coordinated and Compartmentalized Neuromodulation Shapes Sensory Processing in Drosophila. Cell 163, 1742–1755 (2015).
Duvarci, S. & Paré, D. Glucocorticoids enhance the excitability of principal basolateral amygdala neurons. J. Neurosci. 27, 4482–4491 (2007).
Davis, M., Rainnie, D. & Cassell, M. Neurotransmission in the rat amygdala related to fear and anxiety. Trends Neurosci. 17, 208–214 (1994).
Herry, C. & Johansen, J.P. Encoding of fear learning and memory in distributed neuronal circuits. Nat. Neurosci. 17, 1644–1654 (2014).
Duvarci, S. & Pare, D. Amygdala microcircuits controlling learned fear. Neuron 82, 966–980 (2014).
Tovote, P., Fadok, J.P. & Lüthi, A. Neuronal circuits for fear and anxiety. Nat. Rev. Neurosci. 16, 317–331 (2015).
Janak, P.H. & Tye, K.M. From circuits to behaviour in the amygdala. Nature 517, 284–292 (2015).
Parkinson, J.A., Robbins, T.W. & Everitt, B.J. Dissociable roles of the central and basolateral amygdala in appetitive emotional learning. Eur. J. Neurosci. 12, 405–413 (2000).
Cai, H., Haubensak, W., Anthony, T.E. & Anderson, D.J. Central amygdala PKC-δ(+) neurons mediate the influence of multiple anorexigenic signals. Nat. Neurosci. 17, 1240–1248 (2014).
Gallagher, M., Graham, P.W. & Holland, P.C. The amygdala central nucleus and appetitive Pavlovian conditioning: lesions impair one class of conditioned behavior. J. Neurosci. 10, 1906–1911 (1990).
Knapska, E. et al. Differential involvement of the central amygdala in appetitive versus aversive learning. Learn. Mem. 13, 192–200 (2006).
Knapska, E., Radwanska, K., Werka, T. & Kaczmarek, L. Functional internal complexity of amygdala: focus on gene activity mapping after behavioral training and drugs of abuse. Physiol. Rev. 87, 1113–1173 (2007).
Han, S., Soleiman, M.T., Soden, M.E., Zweifel, L.S. & Palmiter, R.D. Elucidating an affective pain circuit that creates a threat memory. Cell 162, 363–374 (2015).
Acknowledgements
We acknowledge A. Wagatsuma and R.L. Redondo for help designing behavioral apparatuses, the MIT BioMicroCenter for support collecting the RNA array data, X. Liu for cloning the Fos-tTA plasmid, T.J. Ryan and D.S. Roy for comments on the manuscript, and all the members of the Tonegawa laboratory for their support. This work is supported in part by NIH Pre-Doctoral Training Grant T32GM007287 (to J.K.) and by the RIKEN Brain Science Institute, the Howard Hughes Medical Institute and the JPB Foundation (to S.T.).
Author information
Authors and Affiliations
Contributions
J.K. and S.T. conceived the study. J.K. identified gene markers. S.I. generated the transgenic Rspo2-Cre mouse. J.K. collected and analyzed histological data. J.K. and S.X., collected and analyzed behavioral data. M.P. collected and analyzed electrophysiological data. J.K. and S.T. wrote the manuscript. J.K., M.P., S.X., S.I. and S.T. discussed and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 RNA analysis of activity-dependent transcriptional profiles from BLA neurons.
a, Example bioanalyzer traces of RNA samples collected from footshock (green) (n = 3), female (red) (n = 3), on dox (black) group (n = 1). Bioanalyzer traces was used to test the quality of RNA sample for RNA microarray, the graph shows the fluorescence levels, which corresponds to RNA levels, of different RNA species of different size (nt). Bioanalyzer traces showed that footshock and female samples yielded RNA samples with RNA quality number (RQN) >6 (n = 6), while the on dox RNA sample RQN <4 (n = 1). Peaks at.02kb, 1.9kb and 4.7kb correspond to the marker, 18S rRNAs, and 28S rRNAs, respectively. b, Analysis of MAS5 normalized data of arrays from the footshock (n = 3) and female (n = 3) group.
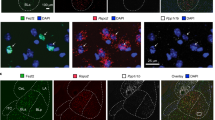
Supplementary Figure 2 In situ hybridization of candidate genetic markers of BLA neurons.
Gene expression of candidate genetic markers in the BLA using in situ hybridization. a-g, Genes that were enriched in the array of the footshock group (green). h-p, Genes that were enriched in the array of the female group (red). q-t, Positive control for interneurons (yellow). u,v, Positive control for excitatory neurons (yellow). Micrographs represent FISH with the exception of Ppp1r1b (smFISH). a-v, nuclear marker, DAPI (blue). Scale bar 100μm.
Supplementary Figure 3 Rspo2+ and Ppp1r1b+ BLA neurons collectively constitute all BLA pyramidal neurons.
smFISH of Rpso2/Camk2a (a), Rspo2/Gad1 (b), Ppp1r1b/Camk2a (c), Ppp1r1b/Gad1 (d), coronal BLA, scale bar 200 μm. e, smFISH of Rpso2+Ppp1r1b/Camk2a, sagittal BLA, scale bar 250 μm. f, higher magnification expression of Rpso2+Ppp1r1b/Camk2a, scale bar 50μm.
Supplementary Figure 4 Spatial distribution of c-Fos expression in the BLA in response to valence-specific stimuli.
c-Fos protein was visualized using IHC by an Alexa Fluor 555 secondary antibody. For improved graphical representation, images were inverted and saturation removed. a, c-Fos expression across the AP-axis of the BLA in response to shock, context, female. b, c-Fos expression across the AP-axis of the BLA in response to olfactory stimuli. c, c-Fos expression across the AP-axis of the BLA in response to gustatory stimuli. Scale bar 250μm.
Supplementary Figure 5 Spatial distribution of Fos expression in Rpso2+ or Ppp1r1b+ BLA neurons in response to valence-specific stimuli.
Analysis from data found in Fig. 3g-j. c-Fos expression in Rpso2+ (a) or Ppp1r1b+ (b) BLA neurons across the AP-axis (mm distance from bregma) in response to shock or no shock. c-Fos expression in Rpso2+ (c) or Ppp1r1b+ (d) BLA neurons across the AP-axis in response to water or no water. Results show mean ± s.e.m (a-d).
Supplementary Figure 6 Validation of Cre-driver mouse lines for targeting Rspo2+ and Ppp1r1b+ BLA neurons.
Rpso2-Cre and Cartpt-Cre mice were injected with a Cre-dependent eYFP virus into the BLA and smFISH was performed against Rspo2 and Ppp1r1b, respectively. a, Quantification of the percentage of Rpso2+ BLA neurons that express eYFP (eYFP/Rspo2) and the percentage of eYFP+ BLA neurons that express Rspo2 (Rpso2/eYFP) (n = 4). b, eYFP (green) and Rspo2 (red) expression in the BLA of virus injected Rspo2-Cre mice. c, Quantification of the percentage of Ppp1r1b+ BLA neurons that express eYFP (eYFP/Ppp1r1b) and the percentage of eYFP+ BLA neurons that express Ppp1r1b (Ppp1r1b/eYFP) (n = 4). d, eYFP (green) and Ppp1r1b (red) expression in the BLA of virus injected Cartpt-Cre mice. Though Ppp1r1b is endogenously expressed outside of the BLA, such as in the intercalated cell mass, choroid plexus, and striatum (Fig. 2c), Cre-dependent virus targeted in the Cartpt-Cre mice does not express in these off targeted cells. Fuzziness of the eYFP signal reflect the effects of the protease digestion step of the smFISH. Scale bar 250μm.
Supplementary Figure 7 Fiber placement for targeting Rspo2+ and Ppp1r1b+ BLA neurons.
Example of optic fiber placement in Rspo2-Arch (a) and Ppp1r1b-Arch (b) mice. Scale bar, 500μm.
Supplementary Figure 8 Retrograde tracing from putative projection targets of Rspo2+ and Ppp1r1b+ BLA neurons.
smFISH of Rspo2 and Ppp1r1b in CTB injected brains. Rspo2 (a) and Ppp1r1b (b) expression in the BLA of CeC-CTB mice. Rspo2 (c) and Ppp1r1b (d) expression in the BLA of CeL/M-CTB mice. Rspo2 (e) and Ppp1r1b (f) expression in the BLA of NAc-CTB mice. Scale bar 250μm. (a) Day 1 t(14) = 0.4779, day 2 t(14) = 1.338. (b) Day 1 t(16) = 1.190, day 2 t(16) = 1.240. (c) t(18) = 0.5887.
Supplementary Figure 9 Activation of NAc fibers of Rspo2+ BLA neurons elicits negative behaviors.
Optic fiber was unilaterally implanted above the NAc of Rspo-ChR2 mice (NAc Rpso2-ChR2). NAc Rspo2-ChR2 underwent behavioral assays. a, Optogenetic freezing test (n = 9). b, Optogenetic self-stimulation test (n = 11). c, Optogenetic place preference test (n = 9). Behavioral performance was compared against Rspo2-ChR2 (Fig. 4) using an unpaired t-test. No significant difference was observed across all assays. d, Optic fiber placement in the NAc of Rspo2-ChR2 mice. Significance for unpaired t-test, all comparison between ChR2 and eYFP groups not significant (N.S).
Supplementary Figure 10 Circuit model of the BLA.
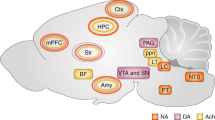
a, Anatomical connections of genetically identifiable populations of amygdala neurons. Projections identified, but cell-type unknown*, hypothetical**. b, The negative circuit of the amygdala (colored). CeC and PL projections are key distinguishing features of Rspo2+ BLA neurons to Ppp1r1b+ BLA neurons. Rspo2+ BLA neurons project the CeC, but the genetic identity of the neurons that are innervated has yet to be identified; one possibility is CeL Calcrl+ neurons. Nevertheless, if Rspo2+ BLA neurons ultimately activate the effector neurons of freezing in the CeM, then an indirect route must be taken through the CeC and/or possibility the intercalated cell (not depicted). c, The positive circuit of the amygdala (colored). CeL, CeM, and IL projections are distinguishing features of Ppp1r1b + BLA neurons to Rspo2+ BLA neurons. Ppp1r1b + BLA neurons send dense fibers to the CeL and CeM. Therefore, a population in the CeL and/or CeM may mediate appetitive behaviors.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–10 (PDF 2302 kb)
Rights and permissions
About this article
Cite this article
Kim, J., Pignatelli, M., Xu, S. et al. Antagonistic negative and positive neurons of the basolateral amygdala. Nat Neurosci 19, 1636–1646 (2016). https://doi.org/10.1038/nn.4414
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.4414
This article is cited by
-
Persistent enhancement of basolateral amygdala-dorsomedial striatum synapses causes compulsive-like behaviors in mice
Nature Communications (2024)
-
Activation patterns in male and female forebrain circuitries during food consumption under novelty
Brain Structure and Function (2024)
-
Closed-loop brain stimulation augments fear extinction in male rats
Nature Communications (2023)
-
Stress deficits in reward behaviour are associated with and replicated by dysregulated amygdala-nucleus accumbens pathway function in mice
Communications Biology (2023)
-
Different projection neurons of basolateral amygdala participate in the retrieval of morphine withdrawal memory with diverse molecular pathways
Molecular Psychiatry (2023)