Abstract
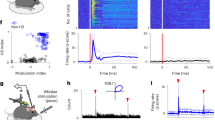
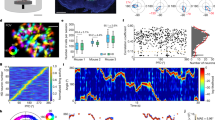
The head-direction (HD) system functions as a compass, with member neurons robustly increasing their firing rates when the animal's head points in a specific direction. HD neurons may be driven by peripheral sensors or, as computational models postulate, internally generated (attractor) mechanisms. We addressed the contributions of stimulus-driven and internally generated activity by recording ensembles of HD neurons in the antero-dorsal thalamic nucleus and the post-subiculum of mice by comparing their activity in various brain states. The temporal correlation structure of HD neurons was preserved during sleep, characterized by a 60°-wide correlated neuronal firing (activity packet), both within and across these two brain structures. During rapid eye movement sleep, the spontaneous drift of the activity packet was similar to that observed during waking and accelerated tenfold during slow-wave sleep. These findings demonstrate that peripheral inputs impinge on an internally organized network, which provides amplification and enhanced precision of the HD signal.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
10 March 2015
In the version of this article initially published online, the Accession Codes section was missing and the x-axis units in Figures 3b and 4d were mislabeled as ms. The correct units are s. The errors have been corrected for the print, PDF and HTML versions of this article.
References
Varela, F., Lachaux, J.P., Rodriguez, E. & Martinerie, J. The brainweb: phase synchronization and large-scale integration. Nat. Rev. Neurosci. 2, 229–239 (2001).
Engel, A.K. & Singer, W. Temporal binding and the neural correlates of sensory awareness. Trends Cogn. Sci. 5, 16–25 (2001).
Buzsáki, G. Rhythms of the Brain (Oxford University Press, 2006).
Rao, R.P. & Ballard, D.H. Predictive coding in the visual cortex: a functional interpretation of some extra-classical receptive-field effects. Nat. Neurosci. 2, 79–87 (1999).
Ranck, J.B. Head direction cells in the deep cell layer of dorsal presubiculum in freely moving rats. in Electrical Activity of Archicortex (eds. Buzsáki, G. & Vanderwolf, C.H.) 217–220 (Akademiai Kiado, 1985).
Taube, J.S., Muller, R.U. & Ranck, J.B. Jr. Head-direction cells recorded from the postsubiculum in freely moving rats. II. Effects of environmental manipulations. J. Neurosci. 10, 436–447 (1990).
Taube, J.S. The head direction signal: origins and sensory-motor integration. Annu. Rev. Neurosci. 30, 181–207 (2007).
Sharp, P.E., Blair, H.T. & Cho, J. The anatomical and computational basis of the rat head-direction cell signal. Trends Neurosci. 24, 289–294 (2001).
Taube, J.S. Head direction cells recorded in the anterior thalamic nuclei of freely moving rats. J. Neurosci. 15, 70–86 (1995).
Zugaro, M.B., Arleo, A., Berthoz, A. & Wiener, S.I. Rapid spatial reorientation and head direction cells. J. Neurosci. 23, 3478–3482 (2003).
Knierim, J.J., Kudrimoti, H.S. & McNaughton, B.L. Place cells, head direction cells and the learning of landmark stability. J. Neurosci. 15, 1648–1659 (1995).
McNaughton, B.L., Battaglia, F.P., Jensen, O., Moser, E.I. & Moser, M.-B. Path integration and the neural basis of the 'cognitive map'. Nat. Rev. Neurosci. 7, 663–678 (2006).
Sargolini, F. et al. Conjunctive representation of position, direction, and velocity in entorhinal cortex. Science 312, 758–762 (2006).
Goodridge, J.P. & Taube, J.S. Interaction between the postsubiculum and anterior thalamus in the generation of head direction cell activity. J. Neurosci. 17, 9315–9330 (1997).
Brandon, M.P. et al. Reduction of theta rhythm dissociates grid cell spatial periodicity from directional tuning. Science 332, 595–599 (2011).
Langston, R.F. et al. Development of the spatial representation system in the rat. Science 328, 1576–1580 (2010).
Wills, T.J., Cacucci, F., Burgess, N. & O'Keefe, J. Development of the hippocampal cognitive map in preweanling rats. Science 328, 1573–1576 (2010).
Skaggs, W.E., Knierim, J.J., Kudrimoti, H.S. & McNaughton, B.L. A model of the neural basis of the rat's sense of direction. Adv. Neural Inf. Process. Syst. 7, 173–180 (1995).
Zhang, K. Representation of spatial orientation by the intrinsic dynamics of the head-direction cell ensemble: a theory. J. Neurosci. 16, 2112–2126 (1996).
Redish, A.D., Elga, A.N. & Touretzky, D.S. A coupled attractor model of the rodent head direction system. Netw. Comput. Neural Syst. 7, 671–685 (1996).
Burak, Y. & Fiete, I.R. Fundamental limits on persistent activity in networks of noisy neurons. Proc. Natl. Acad. Sci. USA 109, 17645–17650 (2012).
Knierim, J.J. & Zhang, K. Attractor dynamics of spatially correlated neural activity in the limbic system. Annu. Rev. Neurosci. 35, 267–285 (2012).
Blair, H.T. Simulation of a thalamocortical circuit for computing directional heading in the rat. in Advances in Neural Information Processing Systems 8 (eds. Touretzky, D.S., Mozer, M.C. & Hasselmo, M.E.) 152–158 (MIT Press, 1996).
Amari, S. Dynamics of pattern formation in lateral-inhibition type neural fields. Biol. Cybern. 27, 77–87 (1977).
Song, P. & Wang, W.J. Angular path integration by moving “hill of activity”: a spiking neuron model without recurrent excitation of the head-direction system. J. Neurosci. 25, 1002–1014 (2005).
Ben-Yishai, R., Bar-Or, R.L. & Sompolinsky, H. Theory of orientation tuning in visual cortex. Proc. Natl. Acad. Sci. USA 92, 3844–3848 (1995).
Buzsáki, G. Two-stage model of memory trace formation: a role for 'noisy' brain states. Neuroscience 31, 551–570 (1989).
Brandon, M.P., Bogaard, A.R., Andrews, C.M. & Hasselmo, M.E. Head direction cells in the postsubiculum do not show replay of prior waking sequences during sleep. Hippocampus 22, 604–618 (2012).
Skaggs, W.E. & McNaughton, B.L. Replay of neuronal firing sequences in rat hippocampus during sleep following spatial experience. Science 271, 1870–1873 (1996).
Nádasdy, Z., Hirase, H., Czurkó, A., Csicsvari, J. & Buzsáki, G. Replay and time compression of recurring spike sequences in the hippocampus. J. Neurosci. 19, 9497–9507 (1999).
Lee, A.K. & Wilson, M.A. Memory of sequential experience in the hippocampus during slow wave sleep. Neuron 36, 1183–1194 (2002).
Euston, D.R., Tatsuno, M. & McNaughton, B.L. Fast-forward playback of recent memory sequences in prefrontal cortex during sleep. Science 318, 1147–1150 (2007).
Peyrache, A., Khamassi, M., Benchenane, K., Wiener, S.I. & Battaglia, F.P. Replay of rule-learning related neural patterns in the prefrontal cortex during sleep. Nat. Neurosci. 12, 919–926 (2009).
Johnson, A., Seeland, K. & Redish, A.D. Reconstruction of the postsubiculum head direction signal from neural ensembles. Hippocampus 15, 86–96 (2005).
Averbeck, B.B., Latham, P.E. & Pouget, A. Neural correlations, population coding and computation. Nat. Rev. Neurosci. 7, 358–366 (2006).
Harris, K.D., Csicsvari, J., Hirase, H., Dragoi, G. & Buzsaki, G. Organization of cell assemblies in the hippocampus. Nature 424, 552–556 (2003).
Mehta, M.R., Barnes, C.A. & McNaughton, B.L. Experience-dependent, asymmetric expansion of hippocampal place fields. Proc. Natl. Acad. Sci. USA 94, 8918 (1997).
Muir, G.M. et al. Disruption of the head direction cell signal after occlusion of the semicircular canals in the freely moving chinchilla. J. Neurosci. 29, 14521–14533 (2009).
Wilson, M.A. & McNaughton, B.L. Reactivation of hippocampal ensemble memories during sleep. Science 265, 676 (1994).
Reid, R.C. & Alonso, J.-M. Specificity of monosynaptic connections from thalamus to visual cortex. Nature 378, 281–284 (1995).
Bruno, R.M. & Sakmann, B. Cortex is driven by weak but synchronously active thalamocortical synapses. Science 312, 1622–1627 (2006).
Neuenschwander, S. & Varela, F.J. Visually Triggered neuronal oscillations in the pigeon: an autocorrelation study of tectal activity. Eur. J. Neurosci. 5, 870–881 (1993).
Craig, A.D. (Bud). Pain mechanisms: labeled lines versus convergence in central processing. Annu. Rev. Neurosci. 26, 1–30 (2003).
Jones, E.G. The Thalamus (Cambridge University Press, 2007).
Shibata, H. Topographic organization of subcortical projections to the anterior thalamic nuclei in the rat. J. Comp. Neurol. 323, 117–127 (1992).
Gonzalo-Ruiz, A. & Lieberman, A.R. Topographic organization of projections from the thalamic reticular nucleus to the anterior thalamic nuclei in the rat. Brain Res. Bull. 37, 17–35 (1995).
Couey, J.J. et al. Recurrent inhibitory circuitry as a mechanism for grid formation. Nat. Neurosci. 16, 318–324 (2013).
Moser, E.I., Moser, M.-B. & Roudi, Y. Network mechanisms of grid cells. Philos. Trans. R. Soc. B Biol. Sci. 369, 20120511 (2014).
Burgess, N., Barry, C. & O'Keefe, J. An oscillatory interference model of grid cell firing. Hippocampus 17, 801–812 (2007).
Moser, E.I., Kropff, E. & Moser, M.-B. Place cells, grid cells, and the brain's spatial representation system. Annu. Rev. Neurosci. 31, 69–89 (2008).
Stark, E., Koos, T. & Buzsáki, G. Diode probes for spatiotemporal optical control of multiple neurons in freely moving animals. J. Neurophysiol. 108, 349–363 (2012).
Berényi, A. et al. Large-scale, high-density (up to 512 channels) recording of local circuits in behaving animals. J. Neurophysiol. 111, 1132–1149 (2014).
Harris, K.D., Henze, D.A., Csicsvari, J., Hirase, H. & Buzsáki, G. Accuracy of tetrode spike separation as determined by simultaneous intracellular and extracellular measurements. J. Neurophysiol. 84, 401–414 (2000).
Hazan, L., Zugaro, M. & Buzsáki, G. Klusters, NeuroScope, NDManager: a free software suite for neurophysiological data processing and visualization. J. Neurosci. Methods 155, 207–216 (2006).
Grosmark, A.D., Mizuseki, K., Pastalkova, E., Diba, K. & Buzsáki, G. REM sleep reorganizes hippocampal excitability. Neuron 75, 1001–1007 (2012).
Zhang, K., Ginzburg, I., McNaughton, B.L. & Sejnowski, T.J. Interpreting neuronal population activity by reconstruction: unified framework with application to hippocampal place cells. J. Neurophysiol. 79, 1017–1044 (1998).
Brown, N.E., Barbieri, R., Eden, U.T. & Frank, L.M. Likelihood methods for neural spike train data analysis. in Computational Neuroscience: a Comprehensive Approach (ed. Feng, J.) 252–283 (Chapman and Hall/CRC, 2003).
Stark, E. & Abeles, M. Unbiased estimation of precise temporal correlations between spike trains. J. Neurosci. Methods 179, 90–100 (2009).
Fujisawa, S., Amarasingham, A., Harrison, M.T. & Buzsáki, G. Behavior-dependent short-term assembly dynamics in the medial prefrontal cortex. Nat. Neurosci. 11, 823–833 (2008).
McCormick, D.A., Connors, B.W., Lighthall, J.W. & Prince, D.A. Comparative electrophysiology of pyramidal and sparsely spiny stellate neurons of the neocortex. J. Neurophysiol. 54, 782–806 (1985).
Barthó, P. Characterization of neocortical principal cells and interneurons by network interactions and extracellular features. J. Neurophysiol. 92, 600–608 (2004).
Acknowledgements
This work was supported by US National Institute of Health grants NS34994, MH54671 and NS074015, the Human Frontier Science Program and the J.D. McDonnell Foundation. A.P. was supported by EMBO Fellowship ALTF 1345-2010, Human Frontier Science Program Fellowship LT000160/2011-l and National Institute of Health Award K99 NS086915-01.
Author information
Authors and Affiliations
Contributions
A.P. and G.B. designed the experiments. A.P., M.M.L. and P.C.P. conducted the experiments. A.P. designed and performed the analyses. A.P. and G.B. wrote the paper with input from the other authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Extension of Fig. 1. Anatomical location of HD cells.
a-c: Histology. a: 4',6-Diamidino-2-Phenylindole (Dapi) staining of a thalamic slice. Three electrode tracks are visible in this section. b: Fluorescent photograph of PV-YFP showing high-density parvalbumin-immunoreactivity signal in the reticular nucleus of the thalamus. c: Merged picture with labeled nuclei. AD, anterodorsal; LD, laterodorsal; AV, anteroventral, AM, anteromedial, MD, mediodorsal; Re, reuniens, RT reticular nucleus. d-f: Localization of HD cells. d: fraction of HD cells recorded by two adjacent shanks. Session #1 (top squares) corresponds to the first session which detected units in the thalamus. The probe was lowered by 70–140 mm at the end of each session. e: Interpolated density of HD cells, based on known inter-shank spacing and the amount of incremental movement of the recording sites between each session. Brightness codes for total number of clustered HD units. f: Putative anatomical density of HD cells superimposed on mouse brain atlas (Allen Mouse Brain Atlas. Available from: http://mouse.brain-map.org). g: Same as a for a slice including the post-subiculum. Arrowheads show four out of the six electrode tracks. PoS: post-subiculum; PRE: pre-subiculum; SUB: subiculum; RSP: retrosplenial cotex; V1: primary visual cortex; SC: superior colliculus. h: same as f for the animal shown in g.
Supplementary Figure 2 Extension of Fig. 1.
a: Superimposed tuning curves of ADn (left, n = 242) and PoS (right, n = 111) HD cells, as well as mean tuning curves (colored solid lines). Gray lines, individual HD neurons. 12% of ADn HD cells and 32% of PoS HD cells were not unimodal: their tuning curve showed at least one other peak that was 50% or more than the maximal peak firing rate. b: Distribution of peak firing rate for ADn HD cells (red) and PoS HD cells (blue; p < 10−10, Mann-Whitney U-test, n = 242 ADn and n = 111 PoS HD cells). c: Distribution of cross-validated HD information content for the two populations of cells (same colors as in b; p < 10−6, Mann-Whitney U-test).
Supplementary Figure 3 Extension of Fig. 1. Two additional examples of thalamic HD cell assembly dynamics across brain states.
a: Top, spectrogram of local field potential recorded from the hippocampal CA1 pyramidal layer. Middle, Bayesian-based decoding (see Methods) of HD signal from the population of ADn HD cells (dark curve) and the animal’s actual head orientation (red curve). Bottom, raster plot of 19 simultaneously recorded HD cells sorted by preferred head direction during waking. b: Close-up of HD cell population activity during SWS (left) and REM (right). Colors code for preferred head direction during waking. Note orderly changes of unit firing of HD cells in all brain states. Data are from session m12-120806. a’–b’: same as above for 23 simultaneously recorded HD cells, also in the ADn. Data are from session m32-140822.
Supplementary Figure 4 Illustration of the algorithm used to estimate the head angular velocity from pairwise temporal cross-correlograms of HD cell spike trains.
The angular correlation function is directly inferred from the two tuning curves as the correlation of the firing rates as a function of the angular offset. It is normalized so that 1 represents chance level (as are all other correlations throughout the paper).
Supplementary Figure 5 Pairwise neuronal correlations were stronger in the ADn than in the PoS, independent of firing rate differences.
Left, average correlations (color-coded) as a function of the difference of preferred directions for increasing value of peak firing rate (geometric mean) in the ADn. Middle, PoS neurons. Right, average (± s.e.m.) correlation for pairs showing firing rate mean <5 Hz (filled bars) or between 5 and 10 Hz (empty bars) in the ADn (left) and the PoS (right). The brain areas comparison effect was significant (p < 10−7) but the firing rate group effect was not (p > 0.05, two-way ANOVA).
Supplementary Figure 6 Synchronous spiking in the ADn and synaptic recruitment of PoS cells. Separation
a: Average wavelet transform of the ADn-ADn cross-correlograms across brain states. b: Histogram of significant cross-correlogram peak times indicates putative mono-synaptic connections between ADn and PoS interneurons (black), pyramidal cells (orange), or undetermined cell types (white). Positive and negative lags correspond to ADn-to-PoS and PoS-to-ADn monosynaptic connections, respectively. c: Left, color-coded cross-correlograms between monosynaptically connected ADn HD cell and putative interneurons in the PoS. Right, putative pyramidal cells. d: Average wavelet transform of the cross-correlograms shown in c. e: Two examples of putative monosynaptic connections between PoS HD cells and ADn HD cell targets. Peaks occur at ∼5 ms. Insets indicate the HD tuning curves of each neuron (red: ADn, blue: PoS).
Supplementary Figure 7 Separation between putative pyramidal and interneurons on the basis of waveform features in PoS.
a: Scatter plot of spike duration and trough-to-peak width (see Methods). b: Marginal distribution of trough-to-peak width for broad spikes (duration > 0.95ms). The subgroup with short trough-to-peak features was classified as ‘undetermined’ (black), whereas the rest were classified as putative pyramidal cells (red). c: Average waveforms for putative pyramidal cells (red) and interneurons (blue).
Supplementary Figure 8 Spike sorting and isolation quality of waveform clusters.
a–b: example of 13 ADn units recorded simultaneously (same data as Supplementary Fig. 3; cluster 1 starts at 2 by convention) by one shank of a silicon probe. a: Average (± s.d.) of unit waveforms. b: Auto-correlograms (in colors) and cross-correlograms (in grey) of all the units. Numbers (and horizontal lines in auto-correlograms) indicate average firing rate. c: Distribution of L-ratio measures for ADn (black) and PoS (red) units. d: Same as c for the Isolation Distance index.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Table 1 (PDF 1835 kb)
Illustration of the relationship between the 'activity packet' of head direction neurons in the AD thalamic nucleus and true head direction.
Left: Possible head directions are shown on a ring with the corresponding activity of neurons. True head direction corresponds to the axis of the mouse head. The activity packet is computed as the sum of the HD tuning curves weighted by the instantaneous activity of HD cells. The mouse was foraging on a large open field. Note that during slow wave sleep in the home cage, the rotation speed of the activity packet is considerably faster, while during REM sleep the speed of the activity packet is comparable to that of the waking state. The artifactual small movements (jitter) of the head during sleep are due to small detection errors of the centers of the LEDs. Right, spiking activity of individual neurons shown in 1 sec frames. Each line is a single neuron. Hot colors indicate increased frequency of discharge. Bottom trace: hippocampal local field potentials. Note theta oscillations during wake and REM. (MPG 40368 kb)
Rights and permissions
About this article
Cite this article
Peyrache, A., Lacroix, M., Petersen, P. et al. Internally organized mechanisms of the head direction sense. Nat Neurosci 18, 569–575 (2015). https://doi.org/10.1038/nn.3968
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3968
This article is cited by
-
Sensory and behavioral modulation of thalamic head-direction cells
Nature Neuroscience (2024)
-
Local origin of excitatory–inhibitory tuning equivalence in a cortical network
Nature Neuroscience (2024)
-
Coregistration of heading to visual cues in retrosplenial cortex
Nature Communications (2023)
-
Population dynamics of head-direction neurons during drift and reorientation
Nature (2023)
-
Toroidal topology of population activity in grid cells
Nature (2022)