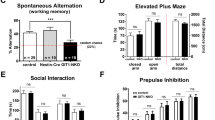
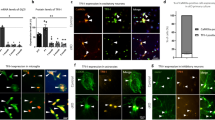
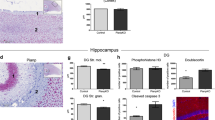
Abstract
Human chromosome 16p11.2 microdeletion is the most common gene copy number variation in autism, but the synaptic pathophysiology caused by this mutation is largely unknown. Using a mouse with the same genetic deficiency, we found that metabotropic glutamate receptor 5 (mGluR5)-dependent synaptic plasticity and protein synthesis was altered in the hippocampus and that hippocampus-dependent memory was impaired. Notably, chronic treatment with a negative allosteric modulator of mGluR5 reversed the cognitive deficit.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Auerbach, B.D., Osterweil, E.K. & Bear, M.F. Mutations causing syndromic autism define an axis of synaptic pathophysiology. Nature 480, 63–68 (2011).
Michalon, A. et al. Chronic pharmacological mGlu5 inhibition corrects fragile X in adult mice. Neuron 74, 49–56 (2012).
Levy, D. et al. Rare de novo and transmitted copy-number variation in autistic spectrum disorders. Neuron 70, 886–897 (2011).
Malhotra, D. & Sebat, J. CNVs: harbingers of a rare variant revolution in psychiatric genetics. Cell 148, 1223–1241 (2012).
Kumar, R.A. et al. Recurrent 16p11.2 microdeletions in autism. Hum. Mol. Genet. 17, 628–638 (2008).
Weiss, L.A. et al. Association between microdeletion and microduplication at 16p11.2 and autism. N. Engl. J. Med. 358, 667–675 (2008).
Zufferey, F. et al. A 600 kb deletion syndrome at 16p11.2 leads to energy imbalance and neuropsychiatric disorders. J. Med. Genet. 49, 660–668 (2012).
Horev, G. et al. Dosage-dependent phenotypes in models of 16p11.2 lesions found in autism. Proc. Natl. Acad. Sci. USA 108, 17076–17081 (2011).
Golzio, C. et al. KCTD13 is a major driver of mirrored neuroanatomical phenotypes of the 16p11.2 copy number variant. Nature 485, 363–367 (2012).
Portmann, T. et al. Behavioral abnormalities and circuit defects in the basal ganglia of a mouse model of 16p11.2 deletion syndrome. Cell Reports 7, 1077–1092 (2014).
Huber, K.M., Gallagher, S.M., Warren, S.T. & Bear, M.F. Altered synaptic plasticity in a mouse model of fragile X mental retardation. Proc. Natl. Acad. Sci. USA 99, 7746–7750 (2002).
Nosyreva, E.D. & Huber, K.M. Metabotropic receptor-dependent long-term depression persists in the absence of protein synthesis in the mouse model of fragile X syndrome. J. Neurophysiol. 95, 3291–3295 (2006).
Waung, M.W., Pfeiffer, B.E., Nosyreva, E.D., Ronesi, J.A. & Huber, K.M. Rapid translation of Arc/Arg3.1 selectively mediates mGluR-dependent LTD through persistent increases in AMPAR endocytosis rate. Neuron 59, 84–97 (2008).
Jakkamsetti, V. et al. Experience-induced Arc/Arg3.1 primes CA1 pyramidal neurons for metabotropic glutamate receptor-dependent long-term synaptic depression. Neuron 80, 72–79 (2013).
Wilkerson, J.R. et al. A role for dendritic mGluR5-mediated local translation of Arc/Arg3.1 in MEF2-dependent synapse elimination. Cell Reports 7, 1589–1600 (2014).
Osterweil, E.K., Krueger, D.D., Reinhold, K. & Bear, M.F. Hypersensitivity to mGluR5 and ERK1/2 leads to excessive protein synthesis in the hippocampus of a mouse model of fragile X syndrome. J. Neurosci. 30, 15616–15627 (2010).
Stiedl, O., Palve, M., Radulovic, J., Birkenfeld, K. & Spiess, J. Differential impairment of auditory and contextual fear conditioning by protein synthesis inhibition in C57BL/6N mice. Behav. Neurosci. 113, 496–506 (1999).
Dölen, G. et al. Correction of fragile X syndrome in mice. Neuron 56, 955–962 (2007).
Darnell, J.C. et al. FMRP stalls ribosomal translocation on mRNAs linked to synaptic function and autism. Cell 146, 247–261 (2011).
Kelleher, R.J. III & Bear, M.F. The autistic neuron: troubled translation? Cell 135, 401–406 (2008).
Acknowledgements
We thank B. Auerbach, E. Sklar and S. Meagher for technical and administrative assistance. This work was partly supported by grants and funding to M.B. from the National Institute of Mental Health (R21MH090452), NICHD (R01HD046943), Simons Foundation (SFARI #240559) and the Simons Center for the Social Brain at the Massachusetts Institute of Technology, and a physician-scientist career development award from the National Institute of Child Health and Human Development (5K08HD053824) to D.T. L.J.S. was supported by National Institute of Mental Health training grant (5T32MH074249).
Author information
Authors and Affiliations
Contributions
M.F.B. and D.T. conceived and designed the study. M.F.B. and A.J.H. supervised the study. D.T. performed hippocampal electrophysiology and contextual fear conditioning. D.T. and A.J.H. performed inhibitory avoidance test. L.J.S. performed hippocampal protein synthesis and immunoblot experiments. D.T., A.J.H., L.J.S. and M.F.B. wrote the manuscript. L.L. and G.J. provided CTEP. A.A.M. provided the 16p11.2 df/– mice before publication and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
M.F.B. holds patents on the use of mGluR5 inhibitors for treatment of fragile X and autism. M.F.B. and D.T. have patents pending on use of mGluR5 inhibitors for treatment of 16p11.2 microdeletion. L.L. and G.J. are employees of Roche Pharmaceuticals.
Integrated supplementary information
Supplementary Figure 1 Steady decline of transmission rate of the 16p11.2 df/+ allele.
The percentage of the heterozygous 16p11.2 df/+ mutant mice, including both male and female, gradually declines across multiple generations during backcrossing to C57BL/6J. The number of 16p11.2 df/+ and total mice for each generation are: N5: 21/53, N6: 26/118, N7: 41/250, N8: 57/434, N9: 16/202, and N10: 8/102.
Supplementary Figure 2 Basal synaptic transmission is normal in the 16p11.2 df/+ mice.
(a) Input-output functions, plotted as fEPSP slope versus stimulus intensity, are not different between the WT (n = 14 animals) and mutant mice (n = 14 animals). Repeated measures one-way ANOVA, p = 0.92.
(b) Paired-pulse facilitation is comparable between the WT (n = 16 animals) and mutant (n = 17 animals) mice across multiple stimulus intervals (10, 20, 50, 100, 200, 300, 500 ms). Repeated measures one-way ANOVA, p = 0.76. All data are plotted as mean ± SEM.
Supplementary Figure 3 Presynaptic LTD is independent of genotype and is not affected by cycloheximide treatment.
DHPG increases paired-pulse facilitation in both the WT and 16p11.2 df/+ mutant slices and this effect is not affected by cycloheximide. PPF is calculated at a 50 ms inter-stimulus interval: WT baseline: 1.44 ± 0.019, n = 17 animals, 37 slices, WT DHPG: 1.55 ± 0.035, n = 17 animals, 18 slices, WT DHPG+CHX: 1.55 ± 0.027, n = 17 animals, 19 slices. Mut baseline: 1.45 ± 0.023, n = 16 animals, 42 slices, Mut DHPG:1.56 ± 0.053, n = 16 animals, 21 slices, Mut DHPG+CHX: 1.53 ± 0.038, n = 16 animals, 21 slices. Statistical analyses are performed using unpaired t-tests. All data are plotted as mean ± SEM.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 (PDF 190 kb)
Rights and permissions
About this article
Cite this article
Tian, D., Stoppel, L., Heynen, A. et al. Contribution of mGluR5 to pathophysiology in a mouse model of human chromosome 16p11.2 microdeletion. Nat Neurosci 18, 182–184 (2015). https://doi.org/10.1038/nn.3911
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3911
This article is cited by
-
Identifying foetal forebrain interneurons as a target for monogenic autism risk factors and the polygenic 16p11.2 microdeletion
BMC Neuroscience (2023)
-
16p11.2 deletion mice exhibit compromised fronto-temporal connectivity, GABAergic dysfunction, and enhanced attentional ability
Communications Biology (2023)
-
Group I and group II metabotropic glutamate receptors are upregulated in the synapses of infant rats prenatally exposed to valproic acid
Psychopharmacology (2023)
-
TAOK2 rescues autism-linked developmental deficits in a 16p11.2 microdeletion mouse model
Molecular Psychiatry (2022)
-
From bench to bedside: The mGluR5 system in people with and without Autism Spectrum Disorder and animal model systems
Translational Psychiatry (2022)