Abstract
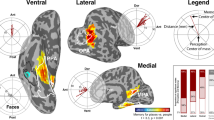
The neural systems that code for location and facing direction during spatial navigation have been investigated extensively; however, the mechanisms by which these quantities are referenced to external features of the world are not well understood. To address this issue, we examined behavioral priming and functional magnetic resonance imaging activity patterns while human subjects recalled spatial views from a recently learned virtual environment. Behavioral results indicated that imagined location and facing direction were represented during this task, and multivoxel pattern analyses indicated that the retrosplenial complex (RSC) was the anatomical locus of these spatial codes. Critically, in both cases, location and direction were defined on the basis of fixed elements of the local environment and generalized across geometrically similar local environments. These results suggest that RSC anchors internal spatial representations to local topographical features, thus allowing us to stay oriented while we navigate and retrieve from memory the experience of being in a particular place.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
12 January 2015
In the version of this article initially published, three citations present in an earlier draft were omitted. To correct this, the following sentence and phrase have been inserted in the first paragraph of the Results section "Whole-brain analyses," before the third sentence and at the end of the fourth, respectively: "This locus is just posterior to retrosplenial cortex proper (Brodmann area 29/30), which has been previously implicated in the coding of spatially fixed objects59." "..., an area that has been previously implicated in the coding of directional information60,61." In the sentence before these inserts, the corresponding description "and just posterior to the retrosplenial cortex proper" has been deleted. The references are as follows: 59. Auger, S.D. & Maguire, E.A. Assessing the mechanism of response in the retrosplenial cortex of good and poor navigators. Cortex 49, 2904–2913 (2013). 60. Schindler, A. & Bartels, A. Parietal cortex codes for egocentric space beyond the field of view. Curr. Biol. 23, 177–182 (2013). 61. Spiers, H.J. & Maguire, E.A. A navigational guidance system in the human brain. Hippocampus 17, 618–626 (2007). The error has been corrected in the HTML and PDF versions of the article.
References
O'Keefe, J. & Dostrovsky, J. The hippocampus as a spatial map. Preliminary evidence from unit activity in the freely-moving rat. Brain Res. 34, 171–175 (1971).
Hafting, T., Fyhn, M., Molden, S., Moser, M.-B. & Moser, E.I. Microstructure of a spatial map in the entorhinal cortex. Nature 436, 801–806 (2005).
Taube, J.S., Muller, R.U. & Ranck, J.B. Head-direction cells recorded from the postsubiculum in freely moving rats. I. Description and quantitative analysis. J. Neurosci. 10, 420–435 (1990).
Vass, L.K. & Epstein, R.A. Abstract representations of location and facing direction in the human brain. J. Neurosci. 33, 6133–6142 (2013).
Hassabis, D. et al. Decoding neuronal ensembles in the human hippocampus. Curr. Biol. 19, 546–554 (2009).
Doeller, C.F., Barry, C. & Burgess, N. Evidence for grid cells in a human memory network. Nature 463, 657–661 (2010).
Ekstrom, A.D. et al. Cellular networks underlying human spatial navigation. Nature 425, 184–188 (2003).
Jacobs, J. et al. Direct recordings of grid-like neuronal activity in human spatial navigation. Nat. Neurosci. 16, 1188–1190 (2013).
Knierim, J.J., Kudrimoti, H.S. & McNaughton, B.L. Place cells, head direction cells, and the learning of landmark stability. J. Neurosci. 15, 1648–1659 (1995).
Valerio, S. & Taube, J.S. Path integration: how the head direction signal maintains and corrects spatial orientation. Nat. Neurosci. 15, 1445–1453 (2012).
Yoder, R.M. et al. Both visual and idiothetic cues contribute to head direction cell stability during navigation along complex routes. J. Neurophysiol. 105, 2989–3001 (2011).
Yoder, R.M., Clark, B.J. & Taube, J.S. Origins of landmark encoding in the brain. Trends Neurosci. 34, 561–571 (2011).
Epstein, R.A. Parahippocampal and retrosplenial contributions to human spatial navigation. Trends Cogn. Sci. 12, 388–396 (2008).
Vann, S.D., Aggleton, J.P. & Maguire, E.A. What does the retrosplenial cortex do? Nat. Rev. Neurosci. 10, 792–802 (2009).
Aguirre, G.K. & D'Esposito, M. Topographical disorientation: a synthesis and taxonomy. Brain 122, 1613–1628 (1999).
Baumann, O. & Mattingley, J.B. Medial parietal cortex encodes perceived heading direction in humans. J. Neurosci. 30, 12897–12901 (2010).
Byrne, P., Becker, S. & Burgess, N. Remembering the past and imagining the future: a neural model of spatial memory and imagery. Psychol. Rev. 114, 340–375 (2007).
McNaughton, B.L., Knierim, J.J. & Wilson, M.A. Vector encoding and the vestibular foundations of spatial cognition: neurophysiological and computational mechanisms. in The Cognitive Neurosciences (ed. Gazzaniga, M.S.) 585–595 (The MIT Press, Cambridge, MA, 1995).
Shelton, A.L. & McNamara, T.P. Systems of spatial reference in human memory. Cognit. Psychol. 43, 274–310 (2001).
Mou, W. & McNamara, T.P. Intrinsic frames of reference in spatial memory. J. Exp. Psychol. Learn. Mem. Cogn. 28, 162–170 (2002).
Kriegeskorte, N., Goebel, R. & Bandettini, P. Information-based functional brain mapping. Proc. Natl. Acad. Sci. USA 103, 3863–3868 (2006).
Maguire, E.A. The retrosplenial contribution to human navigation: a review of lesion and neuroimaging findings. Scand. J. Psychol. 42, 225–238 (2001).
Burgess, N. Spatial cognition and the brain. Ann. NY Acad. Sci. 1124, 77–97 (2008).
Epstein, R.A., Higgins, J.S., Jablonski, K. & Feiler, A.M. Visual scene processing in familiar and unfamiliar environments. J. Neurophysiol. 97, 3670–3683 (2007).
Epstein, R.A., Parker, W.E. & Feiler, A.M. Where am I now? Distinct roles for parahippocampal and retrosplenial cortices in place recognition. J. Neurosci. 27, 6141–6149 (2007).
Rosenbaum, R.S., Ziegler, M., Winocur, G., Grady, C.L. & Moscovitch, M. “I have often walked down this street before”: fMRI studies on the hippocampus and other structures during mental navigation of an old environment. Hippocampus 14, 826–835 (2004).
Zhang, H., Copara, M. & Ekstrom, A.D. Differential recruitment of brain networks following route and cartographic map learning of spatial environments. PLoS ONE 7, e44886 (2012).
Wolbers, T. & Büchel, C. Dissociable retrosplenial and hippocampal contributions to successful formation of survey representations. J. Neurosci. 25, 3333–3340 (2005).
Burgess, N., Spiers, H.J. & Paleologou, E. Orientational manoeuvres in the dark: dissociating allocentric and egocentric influences on spatial memory. Cognition 94, 149–166 (2004).
Cheng, K. A purely geometric module in the rat's spatial representation. Cognition 23, 149–178 (1986).
Gallistel, C.R. The Organization of Learning (The MIT Press, Cambridge, MA, 1990).
Knight, R., Hayman, R., Lin Ginzberg, L. & Jeffery, K. Geometric cues influence head direction cells only weakly in nondisoriented rats. J. Neurosci. 31, 15681–15692 (2011).
Cho, J. & Sharp, P.E. Head direction, place, and movement correlates for cells in the rat retrosplenial cortex. Behav. Neurosci. 115, 3–25 (2001).
Chen, L.L., Lin, L.-H., Green, E.J., Barnes, C.A. & McNaughton, B.L. Head-direction cells in the rat posterior cortex. Exp. Brain Res. 101, 8–23 (1994).
Taube, J.S. & Burton, H.L. Head direction cell activity monitored in a novel environment and during a cue conflict situation. J. Neurophysiol. 74, 1953–1971 (1995).
Dudchenko, P.A. & Zinyuk, L.E. The formation of cognitive maps of adjacent environments: evidence from the head direction cell system. Behav. Neurosci. 119, 1511–1523 (2005).
McNaughton, B.L., Battaglia, F.P., Jensen, O., Moser, E.I. & Moser, M.-B. Path integration and the neural basis of the 'cognitive map'. Nat. Rev. Neurosci. 7, 663–678 (2006).
Sato, N., Sakata, H., Tanaka, Y.L. & Taira, M. Navigation-associated medial parietal neurons in monkeys. Proc. Natl. Acad. Sci. USA 103, 17001–17006 (2006).
Lever, C., Burton, S., Jeewajee, A., O'Keefe, J. & Burgess, N. Boundary vector cells in the subiculum of the hippocampal formation. J. Neurosci. 29, 9771–9777 (2009).
Solstad, T., Boccara, C.N., Kropff, E., Moser, M.-B. & Moser, E.I. Representation of geometric borders in the entorhinal cortex. Science 322, 1865–1868 (2008).
Sharp, P.E. Subicular place cells generate the same “map” for different environments: comparison with hippocampal cells. Behav. Brain Res. 174, 206–214 (2006).
Hung, C.P., Kreiman, G., Poggio, T. & DiCarlo, J.J. Fast readout of object identity from macaque inferior temporal cortex. Science 310, 863–866 (2005).
Marchette, S.A., Yerramsetti, A., Burns, T.J. & Shelton, A.L. Spatial memory in the real world: long-term representations of everyday environments. Mem. Cognit. 39, 1401–1408 (2011).
Tse, D. et al. Schemas and memory consolidation. Science 316, 76–82 (2007).
Spiers, H.J., Hayman, R.M.A., Jovalekic, A., Marozzi, E. & Jeffery, K.J. Place field repetition and purely local remapping in a multicompartment environment. Cereb. Cortex 10.1093/cercor/bht198 (13 August 2013).
Lever, C., Wills, T., Cacucci, F., Burgess, N. & O'Keefe, J. Long-term plasticity in hippocampal place-cell representation of environmental geometry. Nature 416, 90–94 (2002).
Ranganath, C. & Ritchey, M. Two cortical systems for memory-guided behaviour. Nat. Rev. Neurosci. 13, 713–726 (2012).
Hassabis, D., Kumaran, D. & Maguire, E.A. Using imagination to understand the neural basis of episodic memory. J. Neurosci. 27, 14365–14374 (2007).
Schacter, D.L. et al. The future of memory: remembering, imagining, and the brain. Neuron 76, 677–694 (2012).
Lambrey, S., Doeller, C., Berthoz, A. & Burgess, N. Imagining being somewhere else: neural basis of changing perspective in space. Cereb. Cortex 22, 166–174 (2012).
Aguirre, G.K. Continuous carry-over designs for fMRI. Neuroimage 35, 1480–1494 (2007).
Jenkinson, M., Bannister, P., Brady, M. & Smith, S. Improved optimization for the robust and accurate linear registration and motion correction of brain images. Neuroimage 17, 825–841 (2002).
Julian, J.B., Fedorenko, E., Webster, J. & Kanwisher, N. An algorithmic method for functionally defining regions of interest in the ventral visual pathway. Neuroimage 60, 2357–2364 (2012).
Haxby, J.V. et al. Distributed and overlapping representations of faces and objects in ventral temporal cortex. Science 293, 2425–2430 (2001).
Jenkinson, M., Beckmann, C.F., Behrens, T.E.J., Woolrich, M.W. & Smith, S.M. FSL. Neuroimage 62, 782–790 (2012).
Kruskal, J.B. & Wish, M. Multidimensional Scaling (Sage, Beverly Hills, 1978).
Gower, J.C. & Dijksterhuis, G.B. Procrustes Problems (Oxford University Press Oxford, 2004).
Nichols, T.E. & Holmes, A.P. Nonparametric permutation tests for functional neuroimaging: a primer with examples. Hum. Brain Mapp. 15, 1–25 (2002).
Auger, S.D. & Maguire, E.A. Assessing the mechanism of response in the retrosplenial cortex of good and poor navigators. Cortex 49, 2904–2913 (2013).
Schindler, A. & Bartels, A. Parietal cortex codes for egocentric space beyond the field of view. Curr. Biol. 23, 177–182 (2013).
Spiers, H.J. & Maguire, E.A. A navigational guidance system in the human brain. Hippocampus 17, 618–626 (2007).
Acknowledgements
This research was supported by the US National Institutes of Health (EY022350) and the National Science Foundation (SBE-0541957 and SBE-1041707). We thank A. Stigliani for assistance with data collection and three anonymous referees for useful comments on an earlier version of the manuscript.
Author information
Authors and Affiliations
Contributions
S.A.M., L.K.V. and R.A.E. designed the experiments. S.A.M. and J.R. collected the data. S.A.M. analyzed data with input from L.K.V. and R.A.E. S.A.M. and R.A.E. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Reaction time as a function of local direction in experiments 1 and 2.
A. Reaction times in Experiment 1 were faster for imagined views facing the back wall of the museums (local North) than for imagined views facing other directions. Repeated-measures ANOVA revealed a main effect of local direction of the imagined heading, F(3,63) = 8.221, p = 0.0001, and a post-hoc contrast [−3 for the back wall, 1 1 1 for the other directions] confirmed that participants were reliably faster when imagining facing the back wall of the museum compared to the other directions, F(1, 21) = 23.196, p = 0.000008. B. Reaction times in Experiment 2 were faster for imagined views facing the back wall of the museums (local North) than for imagined views facing other directions. Repeated-measures ANOVA revealed a main effect of local direction of the imagined heading, F(3,69) = 11.865, p = 0.000002, and a post-hoc contrast [–3 for the back wall, 1 1 1 for the other directions] confirmed that participants were reliably faster when imagining facing the back wall of the museum compared to the other directions, F(1,23) = 14.077, p = 0.0004. Error bars in both panels indicate standard error of the mean.
Supplementary Figure 2 Decoding of facing direction and location from fMRI activity patterns in the left superior parietal lobe (SPL).
A. Decoding of local direction in left SPL. Left panel. Pattern similarity between views that face the same direction in local space (black bars) was greater than pattern similarity between views that face different local direction (gray bars), (F(1,23) = 27.881, p = 0.00002). There was no main effect of museum, F(1,23) = 1.114, p = 0.302, and local direction did not interact with museum, F(1,23) = 2.359, p = 0.135. Right panel. Residual coding of global direction in left SPL. Pattern similarity was greater for views in different museums that faced the same global but different local directions than for views in different museums that faced different directions in both reference frames (t(23) = 2.349, p = 0.028). B. Decoding of location defined in the local reference frame in SPL. Left panel. Average pattern similarity between views in the same or geometrically-equivalent corners (black bars) was greater than average pattern similarity between views in different corners (gray bars) (F(1,23) = 8.429, p = 0.008). There was no main effect of museum, F(1,23) = 1.524, p = 0.230, and local location did not interact with museum, F(1,23) = 1.840, p = 0.188. Right panel. Absence of residual coding of location in the global reference frame in left SPL. Pattern similarity in SPL was no greater for views in different museums that were in equivalent locations as defined by the global reference frame than for views that did not share location in either the global or local reference frames (t(23) = 1.191, p = 0.245). Error bars in both panels indicate standard error of the mean.
Supplementary Figure 3 Decoding of facing direction from fMRI activity patterns in other functionally and anatomically defined regions of interest.
Decoding of local direction within parahippocampal place area (PPA), occipital place area (OPA/TOS), early visual cortex (EVC), hippocampus, and pre-subiculum. Pattern similarity between views that face the same direction in local space was only marginally greater than pattern similarity between views that face different local directions in the PPA and OPA/TOS (PPA: F(1,23) = 4.247, p = 0.051; OPA/TOS: F(1,23) = 3.463, p = 0.076), while EVC, presubiculum and hippocampus showed no evidence of direction coding (all ps > 0.38). In addition, patterns of activity in OPA/TOS and EVC contained sufficient information to decode the identity of the museum (OPA/TOS: F(1,23) = 5.175, p = 0.033; EVC: F(1,23) = 6.481, p = 0.018), while patterns of activity in PPA, presubiculum, and hippocampus did not (all ps > 0.16). We did not observe interactions between museum and direction coding in any of these regions (all ps > 0.13). In a previous experiment, we observed robust evidence of direction coding within the presubiculum when participants viewed familiar scenes from their college campus4. There are at least four differences between the two studies that could potentially explain this discrepancy. First, differences in task: in the previous experiment participants viewed scenes and reported which (canonical) direction they faced in the world, whereas in our current experiment participants viewed words and never explicitly reported their own heading. Second, differences in environmental familiarity: it may be that more extensive training, or training with both idiothetic and allothetic inputs, is necessary for the presubiculum to engage during memory recall. Third, the number of distinct problems that subjects had to solve was smaller in the earlier experiment (16 problems repeated 17 times each) than in the current experiment (96 problems repeated 5 or 6 times); as such, subjects in the earlier experiment may have relied more heavily on episodic memory to re-create the spatial codes retrieved on earlier trials leading to greater presubicular involvement. Finally, the voxel size was larger in the current experiment (3×3×3 mm) compared to the earlier experiment (3×3×2 mm) which might have made presubicular activity patterns more difficult to discern in the current case.
Supplementary Figure 4 Decoding of location from fMRI activity patterns in other functionally and anatomically defined regions of interest.
Decoding of location within parahippocampal place area (PPA), occipital place area (OPA/TOS), early visual cortex (EVC), hippocampus, and pre-subiculum. Pattern similarity between views in the same or geometrically-equivalent corners was not reliably greater than pattern similarity between views in different corners in any of these ROIs (all ps > 0.22). Main effects of museum were observed in OPA/TOS (F(1,23) = 5.151, p = 0.033) and EVC (F(1,23) = 7.311, p = 0.013) but not in PPA, presubiculum, or hippocampus (all ps > 0.70). We observed no interactions between location and museum in any of these regions (all ps > 0.19). Error bars in both panels indicate standard error of the mean.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4 (PDF 544 kb)
Supplementary Methods Checklist
(PDF 518 kb)
Rights and permissions
About this article
Cite this article
Marchette, S., Vass, L., Ryan, J. et al. Anchoring the neural compass: coding of local spatial reference frames in human medial parietal lobe. Nat Neurosci 17, 1598–1606 (2014). https://doi.org/10.1038/nn.3834
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3834
This article is cited by
-
Perceived spatial presence and body orientation affect the recall of out-of-sight places in an immersive sketching experiment
Psychological Research (2024)
-
From cognitive maps to spatial schemas
Nature Reviews Neuroscience (2023)
-
A proposed attention-based model for spatial memory formation and retrieval
Cognitive Processing (2023)
-
Combining egoformative and alloformative cues in a novel tabletop navigation task
Psychological Research (2023)
-
Sense of self impacts spatial navigation and hexadirectional coding in human entorhinal cortex
Communications Biology (2022)