Abstract
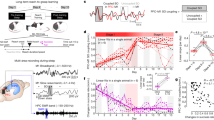
Brain-machine interfaces can allow neural control over assistive devices. They also provide an important platform for studying neural plasticity. Recent studies have suggested that optimal engagement of learning is essential for robust neuroprosthetic control. However, little is known about the neural processes that may consolidate a neuroprosthetic skill. On the basis of the growing body of evidence linking slow-wave activity (SWA) during sleep to consolidation, we examined whether there is 'offline' processing after neuroprosthetic learning. Using a rodent model, we found that, after successful learning, task-related units specifically experienced increased locking and coherency to SWA during sleep. Moreover, spike-spike coherence among these units was substantially enhanced. These changes were not present with poor skill acquisition or after control awake periods, demonstrating the specificity of our observations to learning. Notably, the time spent in SWA predicted the performance gains. Thus, SWA appears to be involved in offline processing after neuroprosthetic learning.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Chapin, J.K., Moxon, K.A., Markowitz, R.S. & Nicolelis, M.A. Real-time control of a robot arm using simultaneously recorded neurons in the motor cortex. Nat. Neurosci. 2, 664–670 (1999).
Koralek, A.C., Jin, X., Long, J.D. II, Costa, R.M. & Carmena, J.M. Corticostriatal plasticity is necessary for learning intentional neuroprosthetic skills. Nature 483, 331–335 (2012).
Arduin, P.J., Fregnac, Y., Shulz, D.E. & Ego–Stengel, V. “Master” neurons induced by operant conditioning in rat motor cortex during a brain-machine interface task. J. Neurosci. 33, 8308–8320 (2013).
Carmena, J.M. et al. Learning to control a brain-machine interface for reaching and grasping by primates. PLoS Biol. 1, E42 (2003).
Serruya, M.D., Hatsopoulos, N.G., Paninski, L., Fellows, M.R. & Donoghue, J.P. Instant neural control of a movement signal. Nature 416, 141–142 (2002).
Taylor, D.M., Tillery, S.I. & Schwartz, A.B. Direct cortical control of 3D neuroprosthetic devices. Science 296, 1829–1832 (2002).
Santhanam, G., Ryu, S.I., Yu, B.M., Afshar, A. & Shenoy, K.V. A high-performance brain-computer interface. Nature 442, 195–198 (2006).
Moritz, C.T., Perlmutter, S.I. & Fetz, E.E. Direct control of paralyzed muscles by cortical neurons. Nature 456, 639–642 (2008).
Jarosiewicz, B. et al. Functional network reorganization during learning in a brain-computer interface paradigm. Proc. Natl. Acad. Sci. USA 105, 19486–19491 (2008).
Musallam, S., Corneil, B.D., Greger, B., Scherberger, H. & Andersen, R.A. Cognitive control signals for neural prosthetics. Science 305, 258–262 (2004).
Hochberg, L.R. et al. Neuronal ensemble control of prosthetic devices by a human with tetraplegia. Nature 442, 164–171 (2006).
Hochberg, L.R. et al. Reach and grasp by people with tetraplegia using a neurally controlled robotic arm. Nature 485, 372–375 (2012).
Collinger, J.L. et al. High-performance neuroprosthetic control by an individual with tetraplegia. Lancet 381, 557–564 (2013).
Ganguly, K. & Carmena, J.M. Emergence of a stable cortical map for neuroprosthetic control. PLoS Biol. 7, e1000153 (2009).
Gilja, V. et al. A high-performance neural prosthesis enabled by control algorithm design. Nat. Neurosci. 15, 1752–1757 (2012).
Stickgold, R. Sleep-dependent memory consolidation. Nature 437, 1272–1278 (2005).
Diekelmann, S. & Born, J. The memory function of sleep. Nat. Rev. Neurosci. 11, 114–126 (2010).
Peigneux, P., Laureys, S., Delbeuck, X. & Maquet, P. Sleeping brain, learning brain. The role of sleep for memory systems. Neuroreport 12, A111–A124 (2001).
Lee, A.K. & Wilson, M.A. Memory of sequential experience in the hippocampus during slow wave sleep. Neuron 36, 1183–1194 (2002).
Walker, M.P., Brakefield, T., Morgan, A., Hobson, J.A. & Stickgold, R. Practice with sleep makes perfect: sleep-dependent motor skill learning. Neuron 35, 205–211 (2002).
Fischer, S., Nitschke, M.F., Melchert, U.H., Erdmann, C. & Born, J. Motor memory consolidation in sleep shapes more effective neuronal representations. J. Neurosci. 25, 11248–11255 (2005).
Cohen, D.A., Pascual–Leone, A., Press, D.Z. & Robertson, E.M. Off-line learning of motor skill memory: a double dissociation of goal and movement. Proc. Natl. Acad. Sci. USA 102, 18237–18241 (2005).
Fischer, S., Hallschmid, M., Elsner, A.L. & Born, J. Sleep forms memory for finger skills. Proc. Natl. Acad. Sci. USA 99, 11987–11991 (2002).
Korman, M. et al. Daytime sleep condenses the time course of motor memory consolidation. Nat. Neurosci. 10, 1206–1213 (2007).
Walker, M.P., Brakefield, T., Hobson, J.A. & Stickgold, R. Dissociable stages of human memory consolidation and reconsolidation. Nature 425, 616–620 (2003).
Huber, R., Ghilardi, M.F., Massimini, M. & Tononi, G. Local sleep and learning. Nature 430, 78–81 (2004).
Tamaki, M. et al. Enhanced spontaneous oscillations in the supplementary motor area are associated with sleep-dependent offline learning of finger-tapping motor-sequence task. J. Neurosci. 33, 13894–13902 (2013).
Huber, R. et al. Arm immobilization causes cortical plastic changes and locally decreases sleep slow wave activity. Nat. Neurosci. 9, 1169–1176 (2006).
Hanlon, E.C., Faraguna, U., Vyazovskiy, V.V., Tononi, G. & Cirelli, C. Effects of skilled training on sleep slow wave activity and cortical gene expression in the rat. Sleep 32, 719–729 (2009).
Moroni, F. et al. Procedural learning and sleep hippocampal low frequencies in humans. Neuroimage 42, 911–918 (2008).
Inostroza, M. & Born, J. Sleep for preserving and transforming episodic memory. Annu. Rev. Neurosci. 36, 79–102 (2013).
Ganguly, K., Dimitrov, D.F., Wallis, J.D. & Carmena, J.M. Reversible large-scale modification of cortical networks during neuroprosthetic control. Nat. Neurosci. 14, 662–667 (2011).
Fetz, E.E. Volitional control of neural activity: implications for brain-computer interfaces. J. Physiol. (Lond.) 579, 571–579 (2007).
Koralek, A.C., Costa, R.M. & Carmena, J.M. Temporally precise cell-specific coherence develops in corticostriatal networks during learning. Neuron 79, 865–872 (2013).
Peyrache, A., Khamassi, M., Benchenane, K., Wiener, S.I. & Battaglia, F.P. Replay of rule-learning related neural patterns in the prefrontal cortex during sleep. Nat. Neurosci. 12, 919–926 (2009).
Peyrache, A., Benchenane, K., Khamassi, M., Wiener, S.I. & Battaglia, F.P. Principal component analysis of ensemble recordings reveals cell assemblies at high temporal resolution. J. Comput. Neurosci. 29, 309–325 (2010).
Lopes-dos-Santos, V., Ribeiro, S. & Tort, A.B. Detecting cell assemblies in large neuronal populations. J. Neurosci. Methods 220, 149–166 (2013).
Tononi, G. & Cirelli, C. Sleep and the price of plasticity: from synaptic and cellular homeostasis to memory consolidation and integration. Neuron 81, 12–34 (2014).
Marshall, L. & Born, J. The contribution of sleep to hippocampus-dependent memory consolidation. Trends Cogn. Sci. 11, 442–450 (2007).
Pavlides, C. & Winson, J. Influences of hippocampal place cell firing in the awake state on the activity of these cells during subsequent sleep episodes. J. Neurosci. 9, 2907–2918 (1989).
Wilson, M.A. & McNaughton, B.L. Reactivation of hippocampal ensemble memories during sleep. Science 265, 676–679 (1994).
Buzsaki, G. Two-stage model of memory trace formation: a role for 'noisy' brain states. Neuroscience 31, 551–570 (1989).
Rasch, B., Buchel, C., Gais, S. & Born, J. Odor cues during slow-wave sleep prompt declarative memory consolidation. Science 315, 1426–1429 (2007).
Jadhav, S.P., Kemere, C., German, P.W. & Frank, L.M. Awake hippocampal sharp-wave ripples support spatial memory. Science 336, 1454–1458 (2012).
Vyazovskiy, V., Borbely, A.A. & Tobler, I. Unilateral vibrissae stimulation during waking induces interhemispheric EEG asymmetry during subsequent sleep in the rat. J. Sleep Res. 9, 367–371 (2000).
Hill, D.N., Mehta, S.B. & Kleinfeld, D. Quality metrics to accompany spike sorting of extracellular signals. J. Neurosci. 31, 8699–8705 (2011).
Fee, M.S., Mitra, P.P. & Kleinfeld, D. Automatic sorting of multiple unit neuronal signals in the presence of anisotropic and non–Gaussian variability. J. Neurosci. Methods 69, 175–188 (1996).
Bjorvatn, B., Fagerland, S. & Ursin, R. EEG power densities (0.5–20 Hz) in different sleep-wake stages in rats. Physiol. Behav. 63, 413–417 (1998).
Phillips, K.G. et al. Decoupling of sleep-dependent cortical and hippocampal interactions in a neurodevelopmental model of schizophrenia. Neuron 76, 526–533 (2012).
Pack, A.I. et al. Novel method for high-throughput phenotyping of sleep in mice. Physiol. Genomics 28, 232–238 (2007).
Mitra, P. & Bokil, H. Observed Brain Dynamics (Oxford University Press, 2008).
Acknowledgements
This work was supported by the Department of Veterans Affairs (B6674 to K.G.), the Burroughs Wellcome Fund (1009855 to K.G.), the American Heart/Stroke Association (0875016N to K.G.) and a VA Psychiatric Research Advanced Fellowship (to D.S.R.).
Author information
Authors and Affiliations
Contributions
C.C.W., T.G. and K.G. conducted the experiments. T.G. and D.S.R. analyzed the data. K.G. supervised the study. All of the authors contributed to the writing and editing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Single-unit waveforms and interspike interval (ISI) from microwire arrays and tetrodes and characteristics of recorded ensembles.
a, Left panel shows 100 waveforms of the task related (tri and trd) and task unrelated (tu) units shown in Fig. 1d (conventions are same). Right panel shows ISI histograms for the corresponding units in the left panel. Scales are same for all waveforms. b, Spike–width calculation for identifying putative interneurons and pyramidal cell types. W1, W2 and W3 are widths of three units between the peaks marked by shaded grey lines. c, Histogram showing counts of spike widths for each of the three category of units (bin size = 10 µs). d, Spike widths for the three categories of units (mean ± s.e.m., one–way ANOVA, F2,171 = 0.55, P > 0.05). e, Relationship between the SWSpre and SWSpost firing rates for each of the three category of units. Also plotted is the linear regression fit for trd units (slope, m= 0.89,R2 =0.89, P < 1 × 10−6). f, Firing rates of the three categories of units in SWS prior to learning the task (mean ± s.e.m., one–way ANOVA, F2,171 = 23, P > 0.05). g, Firing rates of three categories of units in SWS after learning (mean ± s.e.m., one–way ANOVA, F2,171 = 2, P > 0.05). h, Example of clusters based on amplitude feature from two channels. i, Example of one tetrode with 3 units, each of which had no RPVs (Refractory Period Violations). Shown are the waveforms, autocorrelation, cross–correlation, and linear discriminant analysis (LDA). There was no evidence of a relationship in cross–correlations and clear separation of units according to Fisher’s LDA, suggesting over sorting had not occurred. j, Respective STA averages from experiments conducted with either tetrode or microelectrode arrays. The distributions were not significantly different (mean ± s.e.m., t–test, t25 = –0.05, P > 0.05).
Supplementary Figure 2 Comparison of the modulation depth and post-learning effects for negative and positive weight units and changes in STA during task performance.
a, Comparison of the post–learning firing rates of a single positive–weight unit (trd+) and a single negative–weight unit (trd–). b, Average depth of modulation for trd+ and trd– units (mean ± s.e.m., t–test, t25 = 5.17, P <1 × 10−4). c, Distribution of the modulation depths for all trd+ and trd– units (bin size = 5%). d, Mean STA amplitude change for the respective population of trd+ and trd– units (mean ± s.e.m., t–test, t25 = 3.37, P < 0.005). e, Top panel compares the STA of a task related (trd) unit during early (blue) and late (red) trials during task performance/learning in a single session. This STA is based on ‘Beta’ band filtered LFP (i.e. 12–40 Hz). In contrast, we did not observe significant changes in other bands (e.g. SWA band, theta, alpha, higher frequencies) as shown in the unfiltered STA beneath it. f, This shows the population averages (mean ± s.e.m.) of the change in STA amplitude when comparing early and late trials from robust learning sessions. This indicates a significant drop in locking of spiking to the Beta band for task related units. Reduced locking of units to this frequency is consistent with the broader notion of ‘movement–related desynchronization’ that is commonly observed with movements (Murthy, V.N. & Fetz, E.E, 1992. Coherent 25– to 35–Hz oscillations in the sensorimotor cortex of awake behaving monkeys. Proc Natl Acad Sci USA 89, 5670–4) or neuroprosthetic control (McFarland, D.J., Krusienski, D.J. & Wolpaw, J.R, 2006. Brain–computer interface signal processing at the Wadsworth Center: mu and sensorimotor beta rhythms. Prog Brain Res 159, 411–9). Interestingly, tu units did not display this relationship (one–way ANOVA, F2,171 = 36, P < 1 × 10−13with significant post hoc t–tests marked by ‘*’, P < 0.05).
Supplementary Figure 3 Hypnogram.
a, Top panel depicts sleep and awake transitions. Bottom panelsshow the respective LFP activity/EMG activity. b, Comparison of SWS duration for all of the pre– and post– robust learning sessions (mean ± s.e.m., t–test, t14 = –0.02, P = 0.98).
Supplementary Figure 4 Changes in locking to spindle and ripple frequency bands.
a, Left panel shows the STA of a trd unit in the ripple band (100–300Hz) in SWSpre and SWSpost. Right panel illustrates the mean ± s.e.m. change in STA for the three classes of units (one–way ANOVA, F2,171 = 0.27, P = 0.75). b, Left panel shows the STAs in the spindle band (8–20Hz). Right panel shows the mean ± s.e.m. changes in STAs for the three unit classes (one–way ANOVA, F2,171 = 0.35, P = 0.69).
Supplementary Figure 5 Firing characteristics prior to and after learning.
a, Interspike–interval histograms of a task–related unit during SWSpre and SWSpost. These were based on spiking that occurred during the SWA negativity. The two distributions are not significantly different from each other (KS test, P> 0.05), indicating no significant change in bursting. b, Comparison of the ISI distributions for SWSpre and SWSpost. Each bar shows the percentage of units that did not experience a significant change in the ISI distribution. c, Comparison of the delta wave density in SWSpre and SWSpost for robust learning sessions (mean ± s.e.m., t–test, t14 = –1.48, P = 0.16).
Supplementary Figure 6 ON/OFF periods across LFP channels.
a, Grid shows task related and unrelated units at various sites on the microwire probe. Segments of the SWSpost LFP are shown for a variety of electrode recording sites. b, Bar plot showing temporal lags (i.e. temporal delay in milliseconds, mean ± s.e.m) between OFF periods for channels with trd and ‘distant’ tu units (i.e. at least two channels away from a trd), t–test, t13 = –0.30, P = 0.77. c, Bar plot showing density of OFF periods in the channels with trd and distant tu units (mean ± s.e.m., t–test, t13 = –0.30, P = 0.76).
Supplementary Figure 7 SFC magnitude and phase after learning.
a, Plot of the full SFC spectrum as a function of frequency before (blue) and after (red) skill acquisition for a trd and tu unit (0–20Hz band is shown in Fig 3). b, Distribution of phases of trd, tri and tu units are shown in pre and post–SWS in a polar plot. Mean phase of trd units is shown in bold blue (before) and red (after) vector. The phases were uniformly distributed and didn't change significantly for each of the comparisons for the three classes of units between SWSpre and SWSpost (watson–williams circular t-test, P > 0.05).
Supplementary Figure 8 Averaged cross-correlation histograms (CCH).
The mean of the peak–to–tail ratio for each of the three categories (trd–trd, trd–tri and trd–tu neuronal pairs) in their CCHs (one–way ANOVA, F2,156 = 27, P < 1 × 10−10 with significant post hoc t–tests marked by ‘*’, P < 0.05).
Supplementary Figure 9 Changes in reactivation strength during awake versus SWS.
a, Reactivation strength of the signal component during SWSpre, Awakepost (shown in gray in right panel) and SWSpost. b, Mean population difference histograms of pre– and post– reactivation strengths for the awake and SWS period. In each case the respective histogram of SWSpre was subtracted from the respective awake and SWSpost periods. Also shown is the mean population difference curve for poor learning sessions. There was a significantly greater reactivation during post–sleep (logrank test, ‘*’ indicates P < 0.05). c, Event triggered average of reactivation strength centered on the maximum delta wave negativity for SWSpost (red) and awake (purple) sessions. d, Comparison of the change in event triggered negativity for the Awakepost and SWSpost time periods. The relative reactivation compares the baseline (time less than 0) to the peak (mean ± s.e.m., t–test, t17 = –3.61, P < 0.005).
Supplementary Figure 10 Task improvements after sleep.
Average time to reward for the 30 best trials during the first BMI training session (i.e. Block1best) compared to the beginning of second session (Block2begin),overall t-test, t7 = 4.38, P < 0.005.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–10 and Supplementary Table 1 (PDF 1253 kb)
Rights and permissions
About this article
Cite this article
Gulati, T., Ramanathan, D., Wong, C. et al. Reactivation of emergent task-related ensembles during slow-wave sleep after neuroprosthetic learning. Nat Neurosci 17, 1107–1113 (2014). https://doi.org/10.1038/nn.3759
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3759
This article is cited by
-
Adaptive control of synaptic plasticity integrates micro- and macroscopic network function
Neuropsychopharmacology (2023)
-
Cortical–hippocampal coupling during manifold exploration in motor cortex
Nature (2023)
-
Electroencephalogram based brain-computer interface: Applications, challenges, and opportunities
Multimedia Tools and Applications (2023)
-
Characteristics and stability of sensorimotor activity driven by isolated-muscle group activation in a human with tetraplegia
Scientific Reports (2022)
-
Selective modulation of cortical population dynamics during neuroprosthetic skill learning
Scientific Reports (2022)