Abstract
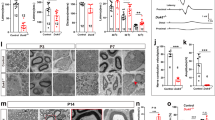
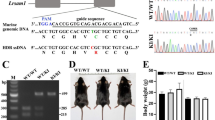
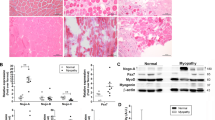
The autosomal dominant disorder neurofibromatosis type 2 (NF2) is a hereditary tumor syndrome caused by inactivation of the NF2 tumor suppressor gene, encoding merlin. Apart from tumors affecting the peripheral and central nervous systems, most NF2 patients develop peripheral neuropathies. This peripheral nerve disease can occur in the absence of nerve-damaging tumors, suggesting an etiology that is independent of gross tumor burden. We discovered that merlin isoform 2 (merlin-iso2) has a specific function in maintaining axonal integrity and propose that reduced axonal NF2 gene dosage leads to NF2-associated polyneuropathy. We identified a merlin-iso2–dependent complex that promotes activation of the GTPase RhoA, enabling downstream Rho-associated kinase to promote neurofilament heavy chain phosphorylation. Merlin-iso2–deficient mice exhibited impaired locomotor capacities, delayed sensory reactions and electrophysiological signs of axonal neuropathy. Sciatic nerves from these mice and sural nerve biopsies from NF2 patients revealed reduced phosphorylation of the neurofilament H subunit, decreased interfilament spacings and irregularly shaped axons.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Luo, L. Rho GTPases in neuronal morphogenesis. Nat. Rev. Neurosci. 1, 173–180 (2000).
Perrot, R., Berges, R., Bocquet, A. & Eyer, J. Review of the multiple aspects of neurofilament functions, and their possible contribution to neurodegeneration. Mol. Neurobiol. 38, 27–65 (2008).
Zhu, Q., Couillard–Despres, S. & Julien, J.P. Delayed maturation of regenerating myelinated axons in mice lacking neurofilaments. Exp. Neurol. 148, 299–316 (1997).
de Waegh, S.M., Lee, V.M. & Brady, S.T. Local modulation of neurofilament phosphorylation, axonal caliber and slow axonal transport by myelinating Schwann cells. Cell 68, 451–463 (1992).
Dubois, M., Strazielle, C., Julien, J.P. & Lalonde, R. Mice with the deleted neurofilament of low molecular weight (Nefl) gene. 2. Effects on motor functions and spatial orientation. J. Neurosci. Res. 80, 751–758 (2005).
England, J.D. & Asbury, A.K. Peripheral neuropathy. Lancet 363, 2151–2161 (2004).
Asthagiri, A.R. et al. Neurofibromatosis type 2. Lancet 373, 1974–1986 (2009).
Sperfeld, A.D., Hein, C., Schroder, J.M., Ludolph, A.C. & Hanemann, C.O. Occurrence and characterization of peripheral nerve involvement in neurofibromatosis type 2. Brain 125, 996–1004 (2002).
Schulz, A. et al. Merlin inhibits neurite outgrowth in the CNS. J. Neurosci. 30, 10177–10186 (2010).
Gianola, S. & Rossi, F. Evolution of the Purkinje cell response to injury and regenerative potential during postnatal development of the rat cerebellum. J. Comp. Neurol. 430, 101–117 (2001).
Jones–Villeneuve, E.M., McBurney, M.W., Rogers, K.A. & Kalnins, V.I. Retinoic acid induces embryonal carcinoma cells to differentiate into neurons and glial cells. J. Cell Biol. 94, 253–262 (1982).
Hashimoto, R. et al. Domain- and site-specific phosphorylation of bovine NF-L by Rho-associated kinase. Biochem. Biophys. Res. Commun. 245, 407–411 (1998).
Moorman, J.P., Luu, D., Wickham, J., Bobak, D.A. & Hahn, C.S. A balance of signaling by Rho family small GTPases RhoA, Rac1 and Cdc42 coordinates cytoskeletal morphology, but not cell survival. Oncogene 18, 47–57 (1999).
Maeda, M., Matsui, T., Imamura, M. & Tsukita, S. Expression level, subcellular distribution and rho-GDI binding affinity of merlin in comparison with Ezrin/Radixin/Moesin proteins. Oncogene 18, 4788–4797 (1999).
Yamashita, T. & Tohyama, M. The p75 receptor acts as a displacement factor that releases Rho from Rho-GDI. Nat. Neurosci. 6, 461–467 (2003).
Sherman, L. et al. Interdomain binding mediates tumor growth suppression by the NF2 gene product. Oncogene 15, 2505–2509 (1997).
Bremer, J. et al. Axonal prion protein is required for peripheral myelin maintenance. Nat. Neurosci. 13, 310–318 (2010).
Elder, G.A., Friedrich, V.L. Jr., Margita, A. & Lazzarini, R.A. Age-related atrophy of motor axons in mice deficient in the mid-sized neurofilament subunit. J. Cell Biol. 146, 181–192 (1999).
Giovannini, M. et al. Conditional biallelic Nf2 mutation in the mouse promotes manifestations of human neurofibromatosis type 2. Genes Dev. 14, 1617–1630 (2000).
Feltri, M.L. et al. Conditional disruption of beta 1 integrin in Schwann cells impedes interactions with axons. J. Cell Biol. 156, 199–209 (2002).
Hagel, C. et al. Polyneuropathy in neurofibromatosis 2: clinical findings, molecular genetics and neuropathological alterations in sural nerve biopsy specimens. Acta Neuropathol. 104, 179–187 (2002).
Hanemann, C.O., Diebold, R. & Kaufmann, D. Role of NF2 haploinsufficiency in NF2-associated polyneuropathy. Brain Pathol. 17, 371–376 (2007).
Jackson, S.J., Pryce, G., Diemel, L.T., Cuzner, M.L. & Baker, D. Cannabinoid-receptor 1 null mice are susceptible to neurofilament damage and caspase 3 activation. Neuroscience 134, 261–268 (2005).
Morrison, J.H. et al. A monoclonal antibody to non-phosphorylated neurofilament protein marks the vulnerable cortical neurons in Alzheimer's disease. Brain Res. 416, 331–336 (1987).
Iseki, C. et al. Rinsho Shinkeigaku [A case of neurofibromatosis type 2 (NF2) presenting with late–onset axonal polyneuropathy] 49, 419–423 (2009).
Scherer, S.S. & Gutmann, D.H. Expression of the neurofibromatosis 2 tumor suppressor gene product, merlin, in Schwann cells. J. Neurosci. Res. 46, 595–605 (1996).
Morrison, H. et al. The NF2 tumor suppressor gene product, merlin, mediates contact inhibition of growth through interactions with CD44. Genes Dev. 15, 968–980 (2001).
Baader, S.L. & Schilling, K. Glutamate receptors mediate dynamic regulation of nitric oxide synthase expression in cerebellar granule cells. J. Neurosci. 16, 1440–1449 (1996).
Malin, S.A., Davis, B.M. & Molliver, D.C. Production of dissociated sensory neuron cultures and considerations for their use in studying neuronal function and plasticity. Nat. Protoc. 2, 152–160 (2007).
Watanabe, S.Y. et al. Calcium phosphate–mediated transfection of primary cultured brain neurons using GFP expression as a marker: application for single neuron electrophysiology. Neurosci. Res. 33, 71–78 (1999).
Jankowski, J., Miething, A., Schilling, K. & Baader, S.L. Physiological purkinje cell death is spatiotemporally organized in the developing mouse cerebellum. Cerebellum 8, 277–290 (2009).
Michailov, G.V. et al. Axonal neuregulin-1 regulates myelin sheath thickness. Science 304, 700–703 (2004).
Mundegar, R.R., Franke, E., Schafer, R., Zweyer, M. & Wernig, A. Reduction of high background staining by heating unfixed mouse skeletal muscle tissue sections allows for detection of thermostable antigens with murine monoclonal antibodies. J. Histochem. Cytochem. 56, 969–975 (2008).
Mulisch, M.W.U. Romeis–Mikroskopische Technik (Spektrum Akademischer Verlag, 2010).
Xia, R.H., Yosef, N. & Ubogu, E.E. Dorsal caudal tail and sciatic motor nerve conduction studies in adult mice: technical aspects and normative data. Muscle Nerve 41, 850–856 (2010).
Acknowledgements
The authors would like to thank U. Petz, C. Poser and S. Ramrath for their expert technical assistance, H. Rosemann, F. Kaufmann and D. Galendo for their skilled breeding and husbandry of animals, and R.E. Ferner for discussions and support. Mpz-cre mice were kindly provided M.L. Feltri (Hunter James Kelly Research Institute). This work was supported by Sonderforschungsbereich 604, Deutsche Forschungsgemeinschaft MO 1421/2–1 and Krebshilfe 107089. A.S. is recipient of a Young Investigator Award from the Children's Tumor Foundation.
Author information
Authors and Affiliations
Contributions
A.S. and H.M. conceived and designed the study. H.M. supervised the experimental program and prepared the manuscript. A.S. performed and analyzed the majority of the experiments and prepared the manuscript. S.L.B. performed the nerve section analysis of both knockout mice and patient biopsies, as well as their analysis. M.N.-K. and M.G. generated the Nf2-iso1−/− and Nf2-iso2−/− mice. R.B. designed the electrophysiological experiments and participated in data acquisition. C.G. and D.H.G. synthesized isoform-specific merlin antibodies. A.Z. and M.J.J. participated in the behavioral analysis of merlin knockout mice and the preparation of primary cell cultures. S.S. conducted nucleotide exchange and binding assays. X.-P.D. and D.B.P. provided tissue samples of Mpz-cre; Nf2loxP/loxP mice. C.H. and V.-F.M. provided NF2 patient biopsy sections for immunohistochemistry. C.O.H. provided NF2 patient biopsy sections for ultrastructural analysis. J.W. and J.M.S. performed the ultrastructural analysis of human NF2 patient biopsies.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 and Supplementary Table 1 (PDF 4832 kb)
Rights and permissions
About this article
Cite this article
Schulz, A., Baader, S., Niwa-Kawakita, M. et al. Merlin isoform 2 in neurofibromatosis type 2–associated polyneuropathy. Nat Neurosci 16, 426–433 (2013). https://doi.org/10.1038/nn.3348
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3348
This article is cited by
-
Long-term Follow-up and Histological Correlation of Peripheral Nervous System Alterations in Neurofibromatosis Type 2
Clinical Neuroradiology (2022)
-
PLPP/CIN-mediated NF2-serine 10 dephosphorylation regulates F-actin stability and Mdm2 degradation in an activity-dependent manner
Cell Death & Disease (2021)
-
Peripheral Nerve Development and the Pathogenesis of Peripheral Neuropathy: the Sorting Point
Neurotherapeutics (2021)
-
Association of intraneural perineurioma with neurofibromatosis type 2
Acta Neurochirurgica (2020)
-
Merlin modulates process outgrowth and synaptogenesis in the cerebellum
Brain Structure and Function (2019)