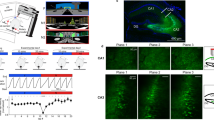
Abstract
Using Ca2+ imaging in freely behaving mice that repeatedly explored a familiar environment, we tracked thousands of CA1 pyramidal cells' place fields over weeks. Place coding was dynamic, as each day the ensemble representation of this environment involved a unique subset of cells. However, cells in the ∼15–25% overlap between any two of these subsets retained the same place fields, which sufficed to preserve an accurate spatial representation across weeks.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
11 February 2013
In the version of this article initially published online, the competing financial interests statement was missing. The error has been corrected in the HTML version of the article.
References
O'Keefe, J.N.L. The Hippocampus as a Cognitive Map (Clarendon, 1978).
Leutgeb, S. et al. Science 309, 619–623 (2005).
Muller, R.U., Kubie, J.L. & Ranck, J.B. Jr. J. Neurosci. 7, 1935–1950 (1987).
Thompson, L.T. & Best, P.J. Brain. Res. 509, 299–308 (1990).
Kentros, C. et al. Science 280, 2121–2126 (1998).
Lever, C., Wills, T., Cacucci, F., Burgess, N. & O'Keefe, J. Nature 416, 90–94 (2002).
Kentros, C.G., Agnihotri, N.T., Streater, S., Hawkins, R.D. & Kandel, E.R. Neuron 42, 283–295 (2004).
Cacucci, F., Wills, T.J., Lever, C., Giese, K.P. & O'Keefe, J. J. Neurosci. 27, 7854–7859 (2007).
Muzzio, I.A. et al. PLoS Biol. 7, e1000140 (2009).
Tian, L. et al. Nat. Methods 6, 875–881 (2009).
Barretto, R.P. et al. Nat. Med. 17, 223–228 (2011).
Ghosh, K.K. et al. Nat. Methods 8, 871–878 (2011).
Mukamel, E.A., Nimmerjahn, A. & Schnitzer, M.J. Neuron 63, 747–760 (2009).
Dombeck, D.A., Harvey, C.D., Tian, L., Looger, L.L. & Tank, D.W. Nat. Neurosci. 13, 1433–1440 (2010).
McHugh, T.J., Blum, K.I., Tsien, J.Z., Tonegawa, S. & Wilson, M.A. Cell 87, 1339–1349 (1996).
Markus, E.J., Barnes, C.A., McNaughton, B.L., Gladden, V.L. & Skaggs, W.E. Hippocampus 4, 410–421 (1994).
Nakazawa, K. et al. Neuron 38, 305–315 (2003).
Rotenberg, A., Mayford, M., Hawkins, R.D., Kandel, E.R. & Muller, R.U. Cell 87, 1351–1361 (1996).
Lisman, J.E. Trends Neurosci. 20, 38–43 (1997).
Gradinaru, V. et al. J. Neurosci. 27, 14231–14238 (2007).
Barretto, R.P., Messerschmidt, B. & Schnitzer, M.J. Nat. Methods 6, 511–512 (2009).
Barretto, R.P.J. & Schnitzer, M.J. in Imaging: a Laboratory Manual (ed. R. Yuste) Ch. 50 (Cold Spring Harbor Laboratory Press, 2011).
Thévenaz, P., Ruttimann, U.E. & Unser, M. IEEE Trans. Image Process. 7, 27–41 (1998).
Nimmerjahn, A., Mukamel, E.A. & Schnitzer, M.J. Neuron 62, 400–412 (2009).
Shannon, C.E. & Weaver, W. The Mathematical Theory of Communication (University of Illinois Press, 1949).
Brown, E.N., Frank, L.M., Tang, D., Quirk, M.C. & Wilson, M.A. J. Neurosci. 18, 7411–7425 (1998).
Quian Quiroga, R. & Panzeri, S. Nat. Rev. Neurosci. 10, 173–185 (2009).
Acknowledgements
We thank L. Looger (Janelia Farm Research Campus) for GCaMP3 plasmid, and A. Attardo, T. Davidson, J. Fitzgerald, J. Li, J.Z. Li, A. Lui, C. Ramachandran, O. Yizhar and T. Zhang for conversations and assistance. We appreciate fellowships from the US National Science Foundation (L.D.B., L.J.K.), the Simons (L.J.K.) and Machiah (Y.Z.) Foundations and research funding to M.J.S. from the Paul G. Allen Family Foundation and the US National Institutes of Health (grants DP1OD003560, R21AG038771 and R21MH099469).
Author information
Authors and Affiliations
Contributions
Y.Z., L.D.B. and M.J.S. designed experiments. Y.Z., L.D.B. and E.O.H. acquired data. L.D.B. and L.J.K. analyzed data. L.D.B., E.D.C. and K.K.G. built equipment. Y.Z., L.D.B., L.J.K. and M.J.S. wrote the paper. A.E.G. and M.J.S. supervised.
Corresponding authors
Ethics declarations
Competing interests
E.D.C., K.K.G. and L.D.B. are now full-time employees of Inscopix Inc., a company that has exclusive license to and is commercializing the integrated microscope technology. Y.Z., L.D.B., E.D.C., K.K.G., A.E.G. and M.J.S. have equity interests in Inscopix. Y.Z., A.E.G. and M.J.S. are consultants to Inscopix.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 2196 kb)
Supplementary Movie 1
Ca2+-imaging in hundreds of CA1 pyramidal cells in a freely behaving mouse. A video showing a mouse exploring a circular arena (left panel) and the simultaneously acquired brain-imaging data of CA1 pyramidal cell Ca2+ activity, displayed as relative changes in fluorescence (ΔF/F) (right panel). 705 pyramidal cells were identified in the total data set and correspond to the neurons of Fig. 1b. The Ca2+-imaging frame rate was 20 Hz, but these data are shown down-sampled to 5 Hz to aid visualization of the Ca2+ transients. The video playback rate is sped up four-fold from how the events actually occurred. (MOV 22073 kb)
Rights and permissions
About this article
Cite this article
Ziv, Y., Burns, L., Cocker, E. et al. Long-term dynamics of CA1 hippocampal place codes. Nat Neurosci 16, 264–266 (2013). https://doi.org/10.1038/nn.3329
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3329
This article is cited by
-
A consistent map in the medial entorhinal cortex supports spatial memory
Nature Communications (2024)
-
Dynamic and selective engrams emerge with memory consolidation
Nature Neuroscience (2024)
-
Subfield-specific interneuron circuits govern the hippocampal response to novelty in male mice
Nature Communications (2024)
-
Neural ensembles in the murine medial prefrontal cortex process distinct information during visual perceptual learning
BMC Biology (2023)
-
Optogenetic frequency scrambling of hippocampal theta oscillations dissociates working memory retrieval from hippocampal spatiotemporal codes
Nature Communications (2023)