Abstract
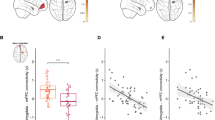
Early life stress (ELS) and function of the hypothalamic-pituitary-adrenal axis predict later psychopathology. Animal studies and cross-sectional human studies suggest that this process might operate through amygdala–ventromedial prefrontal cortex (vmPFC) circuitry implicated in the regulation of emotion. Here we prospectively investigated the roles of ELS and childhood basal cortisol amounts in the development of adolescent resting-state functional connectivity (rs-FC), assessed by functional connectivity magnetic resonance imaging (fcMRI), in the amygdala-PFC circuit. In females only, greater ELS predicted increased childhood cortisol levels, which predicted decreased amygdala-vmPFC rs-FC 14 years later. For females, adolescent amygdala-vmPFC functional connectivity was inversely correlated with concurrent anxiety symptoms but positively associated with depressive symptoms, suggesting differing pathways from childhood cortisol levels function through adolescent amygdala-vmPFC functional connectivity to anxiety and depression. These data highlight that, for females, the effects of ELS and early HPA-axis function may be detected much later in the intrinsic processing of emotion-related brain circuits.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
McEwen, B.S. Stress, adaptation, and disease: allostasis and allostatic load. Ann. NY Acad. Sci. 840, 33–44 (1998).
Gunnar, M. & Quevedo, K. The neurobiology of stress and development. Annu. Rev. Psychol. 58, 145–173 (2007).
Essex, M.J. et al. Influence of early life stress on later hypothalamic-pituitary-adrenal axis functioning and its covariation with mental health symptoms: a study of the allostatic process from childhood into adolescence. Dev. Psychopathol. 23, 1039–1058 (2011).
Essex, M.J., Klein, M.H., Cho, E. & Kalin, N.H. Maternal stress beginning in infancy may sensitize children to later stress exposure: effects on cortisol and behavior. Biol. Psychiatry 52, 776–784 (2002).
Lupien, S.J. et al. Larger amygdala but no change in hippocampal volume in 10-year-old children exposed to maternal depressive symptomatology since birth. Proc. Natl. Acad. Sci. USA 108, 14324–14329 (2011).
Ochsner, K.N. & Gross, J.J. The cognitive control of emotion. Trends Cogn. Sci. 9, 242–249 (2005).
Price, J.L. & Drevets, W.C. Neurocircuitry of mood disorders. Neuropsychopharmacology 35, 192–216 (2010).
Sánchez, M.M., Young, L.J., Plotsky, P.M. & Insel, T.R. Distribution of corticosteroid receptors in the rhesus brain: relative absence of glucocorticoid receptors in the hippocampal formation. J. Neurosci. 20, 4657–4668 (2000).
Lupien, S.J. et al. Effects of stress throughout the lifespan on the brain, behavior, and cognition. Nature Rev. Neuro 10, 434–445 (2009).
Anisman, H., Zaharia, M.D., Meaney, M.J. & Merali, Z. Do early-life events permanently alter behavioral and hormonal responses to stressors? Int. J. Dev. Neurosci. 16, 149–164 (1998).
Weinstock, M. Gender differences in the effects of prenatal stress on brain development and behaviour. Neurochem. Res. 32, 1730–1740 (2007).
Bourke, C.H., Harrell, C.S. & Neigh, G. N. Stress-induced sex differences: adaptations mediated by the glucocorticoid receptor. Horm. Behav. 62, 210–218 (2012).
Kim, M.J. et al. The structural and functional connectivity of the amygdala: from normal emotion to pathological anxiety. Behav. Brain Res. 223, 403–410 (2011).
Kim, M.J., Gee, D.G., Loucks, R.A., Davis, F.C. & Whalen, P.J. Anxiety dissociates dorsal and ventral medial prefrontal cortex functional connectivity with the amygdala at rest. Cereb. Cortex 21, 1667–1673 (2011).
Veer, I.M. et al. Endogenous cortisol is associated with functional connectivity between the amygdala and medial prefrontal cortex. Psychoneuroendocrinology 37, 1039–1047 (2012).
Davidson, R.J. & McEwen, B.S. Social influences on neuroplasticity: stress and interventions to promote well-being. Nat. Neurosci. 15, 689–695 (2012).
Kim, M.J. & Whalen, P.J. The structural integrity of an amygdala–prefrontal pathway predicts trait anxiety. J. Neurosci. 29, 11614–11618 (2009).
Urry, H.L. et al. Amygdala and ventromedial prefrontal cortex are inversely coupled during regulation of negative affect and predict the diurnal pattern of cortisol secretion among older adults. J. Neurosci. 26, 4415–4425 (2006).
Pruessner, J.C. et al. Stress regulation in the central nervous system: evidence from structural and functional neuroimaging studies in human populations-2008 Curt Richter Award Winner. Psychoneuroendocrinology 35, 179–191 (2010).
Thomason, M.E., Hamilton, J.P. & Gotlib, I.H. Stress-induced activation of the HPA axis predicts connectivity between subgenual cingulate and salience network during rest in adolescents. J. Child Psychol. Psychiatry 52, 1026–1034 (2011).
Damosiseaux, J.S. & Greicus, M.D. Greater than the sum of its parts: a review of studies combining structural connectivity and resting-state functional connectivity. Brain Struct. Funct. 213, 525–533 (2009).
van den Heuvel, M.P., Mandl, R.C., Kahn, R.S. & Hulshoff Pol, H.E. Functionally linked resting-state networks reflect the underlying structural connectivity architecture of the human brain. Hum. Brain Mapp. 30, 3127–3141 (2009).
Christianson, J.P., Thompson, B.M., Watkins, L.R. & Maier, S.F. Medial prefrontal cortical activation modulates the impact of controllable and uncontrollable stressor exposure on a social exploration test of anxiety in the rat. Stress 12, 445–450 (2009).
Hakamata, Y. et al. Gender difference in relationship between anxiety-related personality traits and cerebral brain glucose metabolism. Psychiat. Res. 173, 206–211 (2009).
Northoff, G., Wiebking, C., Feinberg, T. & Panksepp, J. The 'resting-state hypothesis' of major depressive disorder—a translational subcortical-cortical framework for a system disorder. Neurosci. Biobehav. Rev. 35, 1929–1945 (2011).
Sheline, Y.I. et al. The default mode network and self-referential processes in depression. Proc. Natl. Acad. Sci. USA 106, 1942–1947 (2009).
Lui, S. et al. Resting-state functional connectivity in treatment-resistant depression. Am. J. Psychiatry 168, 642–648 (2011).
Myers-Schulz, B. & Koenigs, M. Functional anatomy of ventromedial prefrontal cortex: implications for mood and anxiety disorders. Mol. Psychiatry 17, 132–141 (2012).
Beesdo, K. et al. Common and distinct amygdala-function perturbations in depressed vs anxious adolescents. Arch. Gen. Psychiatry 66, 275–285 (2009).
Amunts, K. et al. Cytoarchitectonic mapping of the human amygdala, hippocampal region and entorhinal cortex: intersubject variability and probability maps. Anat. Embryol. (Berl.) 210, 343–352 (2005).
Talairach, J. & Tournoux, P. Co-planar Stereotaxic Atlas of the Human Brain: 3-Dimensional Proportional System: An Approach to Cerebral Imaging. (Thieme, 1988).
Schreiber, J.E. et al. Environmental influences on family similarity in afternoon cortisol levels: twin and parent-offspring designs. Psychoneuroendocrinology 31, 1131–1137 (2006).
Essex, M.J. et al. The confluence of mental, physical, social, and academic difficulties in middle childhood. II: Developing the MacArthur Health and Behavior questionnaire. Am. Acad. Child Adolesc. Psychiatry 41, 588–603 (2002).
Compas, B.E. Coping with stress during childhood and adolescence. Psychol. Bull. 101, 393–403 (1987).
Sarason, I. et al. Assessing the impact of life changes: development of the Life Experiences Survey. J. Consult. Clin. Psychol. 46, 932–946 (1978).
Muthén, L.K. & Muthén, B.O. Mplus User's Guide 5th edn. (2008).
Murgatroyd, C. & Spengler, D. Epigenetic programming of the HPA axis: early life decides. Stress 14, 581–589 (2011).
Michels, N. et al. Negative life events, emotions and psychological difficulties as determinants of salivary cortisol in Belgian primary school children. Psychoneuroendocrinology 37, 1506–1515 (2012).
Cox, R.W. AFNI: software for analysis and visualization of functional magnetic resonance neuroimages. Comput. Biomed. Res. 29, 162–173 (1996).
Smith, S.M. et al. Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage 23, S208–S219 (2004).
Saad, Z.S. et al. A new method for improving functional-to-structural MRI alignment using local Pearson correlation. Neuroimage 44, 839–848 (2009).
Zhang, Y., Brady, M. & Smith, S. Segmentation of brain MR images through a hidden Markov random field model and the expectation-maximization algorithm. IEEE Trans. Med. Imaging 20, 45–57 (2001).
Biswal, B., Zerrin Yetkin, F., Haughton, V.M. & Hyde, J.S. Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn. Reson. Med. 34, 537–541 (1995).
Barrett, P. Structural equation modelling: adjudging model fit. Pers. Individ. Dif. 42, 815–824 (2007).
Hu, L. & Bentler, P.M. Cutoff criteria for fit indexes in covariance structure analysis: conventional criteria versus new alternatives. Struct. Equ. Modeling 6, 1–55 (1999).
Bentler, P.M. & Chou, C.P. Practical issues in structural modeling. Sociol. Methods Res. 16, 78–117 (1987).
Preacher, K.J. & Hayes, A.F. Asymptotic and resampling strategies for assessing and comparing indirect effects in multiple mediator models. Behav. Res. Methods 40, 879–891 (2008).
Acknowledgements
This work was supported by US National Institutes of Health grants P50 MH084051, R01-MH044340 and P50-MH052354; the John D. and Catherine T. MacArthur Foundation Research Network on Psychopathology and Development; and the HealthEmotions Research Institute, Department of Psychiatry, University of Wisconsin School of Medicine and Public Health. Support for P.L.R. was provided by the Canadian Institutes for Health Research post-doctoral fellowship. We thank M. Anderle, R. Fisher, L. Angelos, C. Hermes, A. Koppenhaver and C. Boldt for assistance with data collection and recruitment, and D. Jackson, J. Ollinger, G. Kirk, N. Vack, J. Koger and I. Dolski for general, technical and administrative assistance.
Author information
Authors and Affiliations
Contributions
C.A.B., D.E.S., P.L.R., J.M.A., M.J.E., R.J.D. and R.M.B. wrote and revised the manuscript. C.A.B., D.E.S., P.L.R., E.K.M., N.H.K., M.J.E. and R.M.B. performed data processing, statistical and/or image analyses. M.E.F. and A.S.H. collected data, created figures and assisted with editing the manuscript. C.A.B., D.E.S., P.L.R., J.A.O., M.J.E., R.J.D. and R.M.B. contributed to the interpretation of the data. M.J.E., R.J.D. and R.M.B. supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Table 1 (PDF 45 kb)
Rights and permissions
About this article
Cite this article
Burghy, C., Stodola, D., Ruttle, P. et al. Developmental pathways to amygdala-prefrontal function and internalizing symptoms in adolescence. Nat Neurosci 15, 1736–1741 (2012). https://doi.org/10.1038/nn.3257
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3257
This article is cited by
-
Testing links between unfavorable living conditions, fast life-history strategy adoption, and overeating: a four-wave longitudinal study
European Child & Adolescent Psychiatry (2023)
-
Emotion regulation, negative self-evaluation, and social anxiety symptoms: The mediating role of depressive symptoms
Current Psychology (2023)
-
Glucocorticoid treatment for non-cerebral diseases in children and adolescents is associated with differences in uncinate fasciculus microstructure
Pediatric Research (2022)
-
Structural connectome-based prediction of trait anxiety
Brain Imaging and Behavior (2022)
-
The Development of Externalizing and Internalizing Behaviors Among Youth With or Without a Family History of Substance Use Disorder: The Indirect Effects of Early-Life Stress and Impulsivity
Child Psychiatry & Human Development (2021)