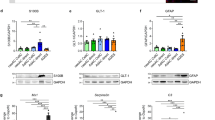
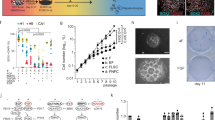
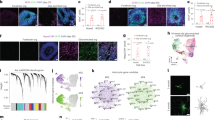
Abstract
Neurogenesis in the adult hippocampus involves activation of quiescent neural stem cells (NSCs) to yield transiently amplifying NSCs, progenitors, and, ultimately, neurons that affect learning and memory. This process is tightly controlled by microenvironmental cues, although a few endogenous factors are known to regulate neuronal differentiation. Astrocytes have been implicated, but their role in juxtacrine (that is, cell-cell contact dependent) signaling in NSC niches has not been investigated. We found that ephrin-B2 presented from rodent hippocampal astrocytes regulated neurogenesis in vivo. Furthermore, clonal analysis in NSC fate-mapping studies revealed a previously unknown role for ephrin-B2 in instructing neuronal differentiation. In addition, ephrin-B2 signaling, transduced by EphB4 receptors on NSCs, activated β-catenin in vitro and in vivo independently of Wnt signaling and upregulated proneural transcription factors. Ephrin-B2+ astrocytes therefore promote neuronal differentiation of adult NSCs through juxtacrine signaling, findings that advance our understanding of adult neurogenesis and may have future regenerative medicine implications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Eriksson, P.S. et al. Neurogenesis in the adult human hippocampus. Nat. Med. 4, 1313–1317 (1998).
Lois, C. & Alvarez-Buylla, A. Long-distance neuronal migration in the adult mammalian brain. Science 264, 1145–1148 (1994).
Spalding, K.L., Bhardwaj, R.D., Buchholz, B.A., Druid, H. & Frisén, J. Retrospective birth dating of cells in humans. Cell 122, 133–143 (2005).
Manganas, L.N. et al. Magnetic resonance spectroscopy identifies neural progenitor cells in the live human brain. Science 318, 980–985 (2007).
Jessberger, S. et al. Seizure-associated, aberrant neurogenesis in adult rats characterized with retrovirus-mediated cell labeling. J. Neurosci. 27, 9400–9407 (2007).
Ekdahl, C.T., Claasen, J.-H., Bonde, S., Kokaia, Z. & Lindvall, O. Inflammation is detrimental for neurogenesis in adult brain. Proc. Natl. Acad. Sci. USA 100, 13632–13637 (2003).
Tattersfield, A.S. et al. Neurogenesis in the striatum of the quinolinic acid lesion model of Huntington's disease. Neuroscience 127, 319–332 (2004).
Suh, H. et al. In vivo fate analysis reveals the multipotent and self-renewal capacities of Sox2+ neural stem cells in the adult hippocampus. Cell Stem Cell 1, 515–528 (2007).
Suh, H., Deng, W. & Gage, F.H. Signaling in adult neurogenesis. Annu. Rev. Cell Dev. Biol. 25, 253–275 (2009).
Bonaguidi, M.A. et al. In vivo clonal analysis reveals self-renewing and multipotent adult neural stem cell characteristics. Cell 145, 1142–1155 (2011).
Mira, H. et al. Signaling through BMPR-IA regulates quiescence and long-term activity of neural stem cells in the adult hippocampus. Cell Stem Cell 7, 78–89 (2010).
Ehm, O. et al. RBPJ-dependent signaling is essential for long-term maintenance of neural stem cells in the adult hippocampus. J. Neurosci. 30, 13794–13807 (2010).
Lugert, S. et al. Quiescent and active hippocampal neural stem cells with distinct morphologies respond selectively to physiological and pathological stimuli and aging. Cell Stem Cell 6, 445–456 (2010).
Lai, K., Kaspar, B.K., Gage, F.H. & Schaffer, D.V. Sonic hedgehog regulates adult neural progenitor proliferation in vitro and in vivo. Nat. Neurosci. 6, 21–27 (2003).
Jin, K. et al. Neurogenesis and aging: FGF-2 and HB-EGF restore neurogenesis in hippocampus and subventricular zone of aged mice. Aging Cell 2, 175–183 (2003).
Cao, L. et al. VEGF links hippocampal activity with neurogenesis, learning and memory. Nat. Genet. 36, 827–835 (2004).
Qu, Q. et al. Orphan nuclear receptor TLX activates Wnt/beta-catenin signaling to stimulate neural stem cell proliferation and self-renewal. Nat. Cell Biol. 12, 31–40 (2010).
Gage, F.H. et al. Survival and differentiation of adult neuronal progenitor cells transplanted to the adult brain. Proc. Natl. Acad. Sci. USA 92, 11879–11883 (1995).
Barkho, B.Z. et al. Identification of astrocyte-expressed factors that modulate neural stem/progenitor cell differentiation. Stem Cells Dev. 15, 407–421 (2006).
Tozuka, Y., Fukuda, S., Namba, T., Seki, T. & Hisatsune, T. GABAergic excitation promotes neuronal differentiation in adult hippocampal progenitor cells. Neuron 47, 803–815 (2005).
Jacobs, S. et al. Retinoic acid is required early during adult neurogenesis in the dentate gyrus. Proc. Natl. Acad. Sci. USA 103, 3902–3907 (2006).
Song, H., Stevens, C.F. & Gage, F.H. Astroglia induce neurogenesis from adult neural stem cells. Nature 417, 39–44 (2002).
Lie, D.-C. Wnt signaling regulates adult hippocampal neurogenesis. Nature 437, 1370–1375 (2005).
Pasquale, E.B. Eph receptor signaling casts a wide net on cell behaviour. Nat. Rev. Mol. Cell Biol. 6, 462–475 (2005).
Knöll, B. & Drescher, U. Ephrin-As as receptors in topographic projections. Trends Neurosci. 25, 145–149 (2002).
Martínez, A. & Soriano, E. Functions of ephrin/Eph interactions in the development of the nervous system: emphasis on the hippocampal system. Brain Res. Brain Res. Rev. 49, 211–226 (2005).
Xu, N.-J., Sun, S., Gibson, J.R. & Henkemeyer, M. A dual shaping mechanism for postsynaptic ephrin-B3 as a receptor that sculpts dendrites and synapses. Nat. Neurosci. 14, 1421–1429 (2011).
Aoki, M., Yamashita, T. & Tohyama, M. EphA receptors direct the differentiation of mammalian neural precursor cells through a mitogen-activated protein kinase–dependent pathway. J. Biol. Chem. 279, 32643–32650 (2004).
del Valle, K., Theus, M.H., Bethea, J.R., Liebl, D.J. & Ricard, J. Neural progenitors proliferation is inhibited by EphB3 in the developing subventricular zone. Int. J. Dev. Neurosci. 29, 9–14 (2011).
Conover, J.C. et al. Disruption of Eph/ephrin signaling affects migration and proliferation in the adult subventricular zone. Nat. Neurosci. 3, 1091–1097 (2000).
Holmberg, J. et al. Ephrin-A2 reverse signaling negatively regulates neural progenitor proliferation and neurogenesis. Genes Dev. 19, 462–471 (2005).
Chumley, M.J., Catchpole, T., Silvany, R.E., Kernie, S.G. & Henkemeyer, M. EphB receptors regulate stem/progenitor cell proliferation, migration, and polarity during hippocampal neurogenesis. J. Neurosci. 27, 13481–13490 (2007).
Hara, Y., Nomura, T., Yoshizaki, K., Frisén, J. & Osumi, N. Impaired hippocampal neurogenesis and vascular formation in ephrin-A5–deficient mice. Stem Cells 28, 974–983 (2010).
Davis, S. et al. Ligands for EPH-related receptor tyrosine kinases that require membrane attachment or clustering for activity. Science 266, 816–819 (1994).
Battiste, J. et al. Ascl1 defines sequentially generated lineage-restricted neuronal and oligodendrocyte precursor cells in the spinal cord. Development 134, 285–293 (2007).
Soriano, P. Generalized lacZ expression with the ROSA26 Cre reporter strain. Nat. Genet. 21, 70–71 (1999).
Kuwabara, T. et al. Wnt-mediated activation of NeuroD1 and retro-elements during adult neurogenesis. Nat. Neurosci. 12, 1097–1105 (2009).
Barrios, A. et al. Eph/Ephrin signaling regulates the mesenchymal-to-epithelial transition of the paraxial mesoderm during somite morphogenesis. Curr. Biol. 13, 1571–1582 (2003).
Batlle, E. et al. Beta-catenin and TCF mediate cell positioning in the intestinal epithelium by controlling the expression of EphB/ephrinB. Cell 111, 251–263 (2002).
Kim, W.-Y. et al. GSK-3 is a master regulator of neural progenitor homeostasis. Nat. Neurosci. 12, 1390–1397 (2009).
Stambolic, V. & Woodgett, J.R. Mitogen inactivation of glycogen synthase kinase-3 beta in intact cells via serine 9 phosphorylation. Biochem. J. 303, 701–704 (1994).
Fuerer, C. & Nusse, R. Lentiviral vectors to probe and manipulate the Wnt signaling pathway. PLoS ONE 5, e9370 (2010).
Elmi, M. et al. TLX activates MASH1 for induction of neuronal lineage commitment of adult hippocampal neuroprogenitors. Mol. Cell. Neurosci. 45, 121–131 (2010).
Jiao, J.-W., Feldheim, D.A. & Chen, D.F. Ephrins as negative regulators of adult neurogenesis in diverse regions of the central nervous system. Proc. Natl. Acad. Sci. USA 105, 8778–8783 (2008).
Nomura, T., Göritz, C., Catchpole, T., Henkemeyer, M. & Frisén, J. EphB signaling controls lineage plasticity of adult neural stem cell niche cells. Cell Stem Cell 7, 730–743 (2010).
Ables, J.L. et al. Notch1 is required for maintenance of the reservoir of adult hippocampal stem cells. J. Neurosci. 30, 10484–10492 (2010).
Wu, H.-H. et al. Autoregulation of neurogenesis by GDF11. Neuron 37, 197–207 (2003).
Aguirre, A., Rubio, M.E. & Gallo, V. Notch and EGFR pathway interaction regulates neural stem cell number and self-renewal. Nature 467, 323–327 (2010).
Picco, V., Hudson, C. & Yasuo, H. Ephrin-Eph signalling drives the asymmetric division of notochord/neural precursors in Ciona embryos. Development 134, 1491–1497 (2007).
Qin, X.F., An, D.S., Chen, I.S.Y. & Baltimore, D. Inhibiting HIV-1 infection in human T cells by lentiviral-mediated delivery of small interfering RNA against CCR5. Proc. Natl. Acad. Sci. USA 100, 183–188 (2003).
Lois, C., Hong, E.J., Pease, S., Brown, E.J. & Baltimore, D. Germline transmission and tissue-specific expression of transgenes delivered by lentiviral vectors. Science 295, 868–872 (2002).
Yu, J.H. & Schaffer, D.V. Selection of novel vesicular stomatitis virus glycoprotein variants from a peptide insertion library for enhanced purification of retroviral and lentiviral vectors. J. Virol. 80, 3285–3292 (2006).
Lim, K.-I., Klimczak, R., Yu, J.H. & Schaffer, D.V. Specific insertions of zinc finger domains into Gag-Pol yield engineered retroviral vectors with selective integration properties. Proc. Natl. Acad. Sci. USA 107, 12475–12480 (2010).
Greenberg, K.P., Geller, S.F., Schaffer, D.V. & Flannery, J.G. Targeted transgene expression in muller glia of normal and diseased retinas using lentiviral vectors. Invest. Ophthalmol. Vis. Sci. 48, 1844–1852 (2007).
Lawlor, P.A., Bland, R.J., Mouravlev, A., Young, D. & During, M.J. Efficient gene delivery and selective transduction of glial cells in the mammalian brain by AAV serotypes isolated from nonhuman primates. Mol. Ther. 17, 1692–1702 (2009).
Peltier, J., O'Neill, A. & Schaffer, D.V. PI3K/Akt and CREB regulate adult neural hippocampal progenitor proliferation and differentiation. Dev. Neurobiol. 67, 1348–1361 (2007).
Peltier, J., Agrawal, S., Robertson, M.J. & Schaffer, D.V. In vitro culture and analysis of adult hippocampal neural progenitors. Methods Mol. Biol. 621, 65–87 (2010).
Acknowledgements
We thank R. Fletcher for his guidance in mouse breeding and genotyping. This research was supported by US National Institutes of Health grant EB007295, Lawrence Berkeley National Lab Laboratory Directed Research and Development grant 3668DS and California Institute of Regenerative Medicine training grant T1-00007.
Author information
Authors and Affiliations
Contributions
C.P. designed and executed the initial in vitro ephrin ligand studies. R.S.A. performed in vitro immunocytochemistry and A.C. performed in vivo immunohistochemistry. R.S.A. designed all of the ephrin-B2 in vivo gain-of-function and loss-of-function experiments, and A.C. and J.B. equally assisted in conducting the experiments. RNAi vectors were designed by R.S.A. and K.-I.L., cloned by K.-I.L. and P.S., and R.S.A. validated vectors experimentally and conducted in vitro RNAi and co-culture experiments. A.C. validated the expression of shRNA vectors in hippocampal astrocytes. R.S.A. conducted mouse breeding and designed and conducted fate mapping experiments with substantial input and assistance from A.C. A.C. conducted clonal analysis experiments, designed and conducted all of the experiments related to in vivo β-catenin activity studies, and analyzed tissue sample from both fate mapping and β-catenin activity studies. A.C. and C.P., and to a lesser extent R.S.A., executed the ephrin-B2, GSK3β S9A, β-catenin, Mash1 and NeuroD1 mechanistic studies. R.S.A. and A.C. performed statistical analysis. A.C. illustrated Supplementary Figure 8. R.S.A. and D.S. wrote the manuscript, with substantial input from A.C. D.V.S. supervised all aspects of this work and M.B. provided important scientific input and feedback.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 and Supplementary Table 1 (PDF 4409 kb)
Rights and permissions
About this article
Cite this article
Ashton, R., Conway, A., Pangarkar, C. et al. Astrocytes regulate adult hippocampal neurogenesis through ephrin-B signaling. Nat Neurosci 15, 1399–1406 (2012). https://doi.org/10.1038/nn.3212
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3212
This article is cited by
-
Neonatal cortical astrocytes possess intrinsic potential in neuronal conversion in defined media
Acta Pharmacologica Sinica (2021)
-
Roles and Mechanisms of Axon-Guidance Molecules in Alzheimer’s Disease
Molecular Neurobiology (2021)
-
Ephrin-B2 paces neuronal production in the developing neocortex
BMC Developmental Biology (2020)
-
The role of adult hippocampal neurogenesis in brain health and disease
Molecular Psychiatry (2019)
-
Possible Involvement of PI3-K/Akt-Dependent GSK-3β Signaling in Proliferation of Neural Progenitor Cells After Hypoxic Exposure
Molecular Neurobiology (2019)