Abstract
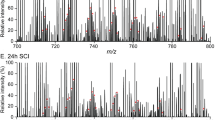
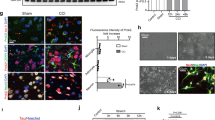
The brain contains a highly diversified complement of molecular species of a mitochondria-specific phospholipid, cardiolipin, which, because of its polyunsaturation, can readily undergo oxygenation. Using global lipidomics analysis in experimental traumatic brain injury (TBI), we found that TBI was accompanied by oxidative consumption of polyunsaturated cardiolipin and the accumulation of more than 150 new oxygenated molecular species of cardiolipin. RNAi-based manipulations of cardiolipin synthase and cardiolipin levels conferred resistance to mechanical stretch, an in vitro model of traumatic neuronal injury, in primary rat cortical neurons. By applying a brain-permeable mitochondria-targeted electron scavenger, we prevented cardiolipin oxidation in the brain, achieved a substantial reduction in neuronal death both in vitro and in vivo, and markedly reduced behavioral deficits and cortical lesion volume. We conclude that cardiolipin oxygenation generates neuronal death signals and that prevention of it by mitochondria-targeted small molecule inhibitors represents a new target for neuro-drug discovery.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
05 September 2012
In the version of this article initially published online, the name of author Alejandro K. Samhan-Arias was given as Alejandro S. Arias. The error has been corrected for the print, PDF and HTML versions of this article.
References
Faul, M., Xu, L., Wald, M.M. & Coronado, V.G. Traumatic Brain Injury in the United States: Emergency Department Visits, Hospitalizations and Deaths 2002–2006 (Centers for Disease Control and Prevention, National Center for Injury Prevention and Control, Atlanta, Georgia, 2010).
Thurman, D.J., Alverson, C., Dunn, K.A., Guerrero, J. & Sniezek, J.E. Traumatic brain injury in the United States: a public health perspective. J. Head Trauma Rehabil. 14, 602–615 (1999).
RAND. Invisible Wounds of War Study. <http://www.rand.org/multi/military/veterans.html> (26 June 2011).
Ghajar, J. Traumatic brain injury. Lancet 356, 923–929 (2000).
Maas, A.I., Roozenbeek, B. & Manley, G.T. Clinical trials in traumatic brain injury: past experience and current developments. Neurotherapeutics 7, 115–126 (2010).
Stoica, B.A. & Faden, A.I. Cell death mechanisms and modulation in traumatic brain injury. Neurotherapeutics 7, 3–12 (2010).
Broadhurst, C.L. et al. Brain-specific lipids from marine, lacustrine, or terrestrial food resources: potential impact on early African Homo sapiens. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 131, 653–673 (2002).
Kiebish, M.A. et al. Lipidomic analysis and electron transport chain activities in C57BL/6J mouse brain mitochondria. J. Neurochem. 106, 299–312 (2008).
Kagan, V.E. et al. Cytochrome c acts as a cardiolipin oxygenase required for release of proapoptotic factors. Nat. Chem. Biol. 1, 223–232 (2005).
Chan, P.H. & Fishman, R.A. Transient formation of superoxide radicals in polyunsaturated fatty acid–induced brain swelling. J. Neurochem. 35, 1004–1007 (1980).
Verweij, B.H. et al. Impaired cerebral mitochondrial function after traumatic brain injury in humans. J. Neurosurg. 93, 815–820 (2000).
Nomura, K., Imai, H., Koumura, T., Kobayashi, T. & Nakagawa, Y. Mitochondrial phospholipid hydroperoxide glutathione peroxidase inhibits the release of cytochrome c from mitochondria by suppressing the peroxidation of cardiolipin in hypoglycaemia-induced apoptosis. Biochem. J. 351, 183–193 (2000).
Ran, Q. et al. Transgenic mice overexpressing glutathione peroxidase 4 are protected against oxidative stress–induced apoptosis. J. Biol. Chem. 279, 55137–55146 (2004).
Wipf, P. et al. Mitochondrial targeting of selective electron scavengers: synthesis and biological analysis of hemigramicidin-TEMPO conjugates. J. Am. Chem. Soc. 127, 12460–12461 (2005).
Bayir, H. et al. Selective early cardiolipin peroxidation after traumatic brain injury: an oxidative lipidomics analysis. Ann. Neurol. 62, 154–169 (2007).
Bayir, H. et al. Assessment of antioxidant reserves and oxidative stress in cerebrospinal fluid after severe traumatic brain injury in infants and children. Pediatr. Res. 51, 571–578 (2002).
Hamm, R.J. et al. Cognitive deficits following traumatic brain injury produced by controlled cortical impact. J. Neurotrauma 9, 11–20 (1992).
Kline, A.E., Massucci, J.L., Ma, X., Zafonte, R.D. & Dixon, C.E. Bromocriptine reduces lipid peroxidation and enhances spatial learning and hippocampal neuron survival in a rodent model of focal brain trauma. J. Neurotrauma 21, 1712–1722 (2004).
Scafidi, S., Racz, J., Hazelton, J., McKenna, M.C. & Fiskum, G. Neuroprotection by acetyl-L-carnitine after traumatic injury to the immature rat brain. Dev. Neurosci. 32, 480–487 (2010).
Schmued, L.C., Stowers, C.C., Scallet, A.C. & Xu, L. Fluoro-Jade C results in ultra high resolution and contrast labeling of degenerating neurons. Brain Res. 1035, 24–31 (2005).
Stoyanovsky, D.A. & Cederbaum, A.I. ESR and HPLC-EC analysis of ethanol oxidation to 1-hydroxyethyl radical: rapid reduction and quantification of POBN and PBN nitroxides. Free Radic. Biol. Med. 25, 536–545 (1998).
Zhang, R., Goldstein, S. & Samuni, A. Kinetics of superoxide-induced exchange among nitroxide antioxidants and their oxidized and reduced forms. Free Radic. Biol. Med. 26, 1245–1252 (1999).
Siesjö, B.K. Cell damage in the brain: a speculative synthesis. J. Cereb. Blood Flow Metab. 1, 155–185 (1981).
Wei, E.P., Kontos, H.A., Dietrich, W.D., Povlishock, J.T. & Ellis, E.F. Inhibition by free radical scavengers and by cyclooxygenase inhibitors of pial arteriolar abnormalities from concussive brain injury in cats. Circ. Res. 48, 95–103 (1981).
Muizelaar, J.P. et al. Improving the outcome of severe head injury with the oxygen radical scavenger polyethylene glycol–conjugated superoxide dismutase: a phase II trial. J. Neurosurg. 78, 375–382 (1993).
Lo, E.H., Singhal, A.B., Torchilin, V.P. & Abbott, N.J. Drug delivery to damaged brain. Brain Res. Brain Res. Rev. 38, 140–148 (2001).
Sorice, M. et al. Cardiolipin and its metabolites move from mitochondria to other cellular membranes during death receptor–mediated apoptosis. Cell Death Differ. 11, 1133–1145 (2004).
Nakagawa, Y. Initiation of apoptotic signal by the peroxidation of cardiolipin of mitochondria. Ann. NY Acad. Sci. 1011, 177–184 (2004).
Ott, M., Robertson, J.D., Gogvadze, V., Zhivotovsky, B. & Orrenius, S. Cytochrome c release from mitochondria proceeds by a two-step process. Proc. Natl. Acad. Sci. USA 99, 1259–1263 (2002).
Zhao, Z. et al. Protection of pancreatic beta-cells by group VIA phospholipase A(2)–mediated repair of mitochondrial membrane peroxidation. Endocrinology 151, 3038–3048 (2010).
Petrosillo, G., Moro, N., Paradies, V., Ruggiero, F.M. & Paradies, G. Increased susceptibility to Ca2+-induced permeability transition and to cytochrome c release in rat heart mitochondria with aging: effect of melatonin. J. Pineal Res. 48, 340–346 (2010).
Petrosillo, G., Casanova, G., Matera, M., Ruggiero, F.M. & Paradies, G. Interaction of peroxidized cardiolipin with rat-heart mitochondrial membranes: induction of permeability transition and cytochrome c release. FEBS Lett. 580, 6311–6316 (2006).
Korytowski, W., Basova, L.V., Pilat, A., Kernstock, R.M. & Girotti, A.W. Permeabilization of the mitochondrial outer membrane by Bax/truncated Bid (tBid) proteins as sensitized by cardiolipin hydroperoxide translocation: mechanistic implications for the intrinsic pathway of oxidative apoptosis. J. Biol. Chem. 286, 26334–26343 (2011).
Betaneli, V., Petrov, E.P. & Schwille, P. The role of lipids in VDAC oligomerization. Biophys. J. 102, 523–531 (2012).
Rostovtseva, T.K., Kazemi, N., Weinrich, M. & Bezrukov, S.M. Voltage gating of VDAC is regulated by nonlamellar lipids of mitochondrial membranes. J. Biol. Chem. 281, 37496–37506 (2006).
Deng-Bryant, Y., Singh, I.N., Carrico, K.M. & Hall, E.D. Neuroprotective effects of tempol, a catalytic scavenger of peroxynitrite-derived free radicals, in a mouse traumatic brain injury model. J. Cereb. Blood Flow Metab. 28, 1114–1126 (2008).
Wilcox, C.S. & Pearlman, A. Chemistry and antihypertensive effects of tempol and other nitroxides. Pharmacol. Rev. 60, 418–469 (2008).
Mitchell, J.B. et al. Biologically active metal-independent superoxide dismutase mimics. Biochemistry 29, 2802–2807 (1990).
Bobko, A.A., Kirilyuk, I.A., Grigor'ev, I.A., Zweier, J.L. & Khramtsov, V.V. Reversible reduction of nitroxides to hydroxylamines: roles for ascorbate and glutathione. Free Radic. Biol. Med. 42, 404–412 (2007).
Soule, B.P. et al. The chemistry and biology of nitroxide compounds. Free Radic. Biol. Med. 42, 1632–1650 (2007).
Trnka, J., Blaikie, F.H., Smith, R.A. & Murphy, M.P. A mitochondria-targeted nitroxide is reduced to its hydroxylamine by ubiquinol in mitochondria. Free Radic. Biol. Med. 44, 1406–1419 (2008).
Fan, P., Yamauchi, T., Noble, L.J. & Ferriero, D.M. Age-dependent differences in glutathione peroxidase activity after traumatic brain injury. J. Neurotrauma 20, 437–445 (2003).
Scheff, S.W. & Sullivan, P.G. Cyclosporin A significantly ameliorates cortical damage following experimental traumatic brain injury in rodents. J. Neurotrauma 16, 783–792 (1999).
Singh, I.N., Sullivan, P.G. & Hall, E.D. Peroxynitrite-mediated oxidative damage to brain mitochondria: protective effects of peroxynitrite scavengers. J. Neurosci. Res. 85, 2216–2223 (2007).
Alessandri, B. et al. Cyclosporin A improves brain tissue oxygen consumption and learning/memory performance after lateral fluid percussion injury in rats. J. Neurotrauma 19, 829–841 (2002).
Lewén, A. et al. Oxidative stress-dependent release of mitochondrial cytochrome c after traumatic brain injury. J. Cereb. Blood Flow Metab. 21, 914–920 (2001).
DeKosky, S.T., Ikonomovic, M.D. & Gandy, S. Traumatic brain injury—football, warfare and long-term effects. N. Engl. J. Med. 363, 1293–1296 (2010).
McKee, A.C. et al. TDP-43 proteinopathy and motor neuron disease in chronic traumatic encephalopathy. J. Neuropathol. Exp. Neurol. 69, 918–929 (2010).
Ji, J. et al. Mitochondrial injury after mechanical stretch of cortical neurons in vitro: biomarkers of apoptosis and selective peroxidation of anionic phospholipids. J. Neurotrauma 29, 776–788 (2012).
Koizumi, S. et al. Imaging mass spectrometry revealed the production of lyso-phosphatidylcholine in the injured ischemic rat brain. Neuroscience 168, 219–225 (2010).
Folch, J., Lees, M. & Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 226, 497–509 (1957).
Minkler, P.E. & Hoppel, C.L. Separation and characterization of cardiolipin molecular species by reverse-phase ion pair high-performance liquid chromatography-mass spectrometry. J. Lipid Res. 51, 856–865 (2010).
Kim, J., Minkler, P.E., Salomon, R.G., Anderson, V.E. & Hoppel, C.L. Cardiolipin: characterization of distinct oxidized molecular species. J. Lipid Res. 52, 125–135 (2011).
Adelson, P.D., Dixon, C.E., Robichaud, P. & Kochanek, P.M. Motor and cognitive functional deficits following diffuse traumatic brain injury in the immature rat. J. Neurotrauma 14, 99–108 (1997).
Shellington, D.K. et al. Polynitroxylated pegylated hemoglobin: a novel neuroprotective hemoglobin for acute volume-limited fluid resuscitation after combined traumatic brain injury and hemorrhagic hypotension in mice. Crit. Care Med. 39, 494–505 (2011).
Mouton, PR. Unbiased Stereology: a Concise Guide (The John Hopkins University Press, 2011).
Acknowledgements
The authors would like to thank J. Lewis for the technical assistance of unbiased stereology for cortical lesion volume, J. Davoren for the preparation of XJB-5-131 and Y.M. Frapart (Laboratoire de Chimie Biochimie Pharmacologique et Toxicologique, Université Paris Descartes) for providing an L-band EPR spectrometer for in vivo imaging. This study was supported in part by grants from the US National Institutes of Health (NS061817 (H.B.), NS060005 (A.E.K.), HL070755 (V.E.K.), ES020693 (Y.Y.T. and V.E.K.), NS076511 and U19AI068021 (H.B. and V.E.K.)), the US National Institute for Occupational Safety and Health (OH008282 to V.E.K.) and the US Army (W81XWH-09-0187 to P.M.K.). A.K.S.-A. is a recipient of a research fellowship from La Junta de Extremadura y el Fondo Social Europeo (2010063090).
Author information
Authors and Affiliations
Contributions
J.J. designed and performed experiments, analyzed data and wrote the manuscript. A.E.K. and J.P.C. contributed to the neurocognitive outcome assessment and writing of the manuscript. A.A., Y.Y.T. and A.K.S.-A. contributed to the assessment of cardiolipin oxidation by 2D-LC-MS. L.J.S. contributed to the mass spectrometry imaging. V.A.T. contributed to the EPR measurements. B.F. contributed to the in vivo EPR imaging. M.D.M. contributed to the unbiased stereology for cortical lesion volume. A.M.P. and D.O.O. contributed to evaluation of human TBI tissue and writing of the manuscript. H.A. performed in vivo TBI experiments. R.S.B.C. and P.M.K. contributed to data analysis and manuscript writing. P.W. contributed to the preparation of XJB-5-131 and manuscript writing. V.E.K. and H.B. initiated and directed the entire study, designed experiments and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 and Supplementary Table 1 (PDF 1288 kb)
Rights and permissions
About this article
Cite this article
Ji, J., Kline, A., Amoscato, A. et al. Lipidomics identifies cardiolipin oxidation as a mitochondrial target for redox therapy of brain injury. Nat Neurosci 15, 1407–1413 (2012). https://doi.org/10.1038/nn.3195
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3195
This article is cited by
-
Fetuin-A alleviates neuroinflammation against traumatic brain injury-induced microglial necroptosis by regulating Nrf-2/HO-1 pathway
Journal of Neuroinflammation (2022)
-
Restoring mitochondrial cardiolipin homeostasis reduces cell death and promotes recovery after spinal cord injury
Cell Death & Disease (2022)
-
Lipidic profiles of patients starting peritoneal dialysis suggest an increased cardiovascular risk beyond classical dyslipidemia biomarkers
Scientific Reports (2022)
-
Prokineticin-2 prevents neuronal cell deaths in a model of traumatic brain injury
Nature Communications (2021)
-
Neuroinflammation trajectories precede cognitive impairment after experimental meningitis—evidence from an in vivo PET study
Journal of Neuroinflammation (2020)