Abstract
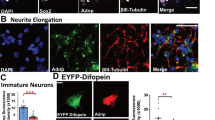
How neurons develop their morphology is an important question in neurobiology. Here we describe a new pathway that specifically affects the formation of basal dendrites and axonal projections in cortical pyramidal neurons. We report that thousand-and-one-amino acid 2 kinase (TAOK2), also known as TAO2, is essential for dendrite morphogenesis. TAOK2 downregulation impairs basal dendrite formation in vivo without affecting apical dendrites. Moreover, TAOK2 interacts with Neuropilin 1 (Nrp1), a receptor protein that binds the secreted guidance cue Semaphorin 3A (Sema3A). TAOK2 overexpression restores dendrite formation in cultured cortical neurons from Nrp1Sema− mice, which express Nrp1 receptors incapable of binding Sema3A. TAOK2 overexpression also ameliorates the basal dendrite impairment resulting from Nrp1 downregulation in vivo. Finally, Sema3A and TAOK2 modulate the formation of basal dendrites through the activation of the c-Jun N-terminal kinase (JNK). These results delineate a pathway whereby Sema3A and Nrp1 transduce signals through TAOK2 and JNK to regulate basal dendrite development in cortical neurons.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
09 July 2012
In the version of this article initially published, the scale bar length for Figure 7d was not given. The bar represents 10 μm. The error has been corrected in the HTML and PDF versions of the article.
References
Spruston, N. Pyramidal neurons: dendritic structure and synaptic integration. Nat. Rev. Neurosci. 9, 206–221 (2008).
Mukaetova-Ladinska, E.B., Arnold, H., Jaros, E., Perry, R. & Perry, E. Depletion of MAP2 expression and laminar cytoarchitectonic changes in dorsolateral prefrontal cortex in adult autistic individuals. Neuropathol. Appl. Neurobiol. 30, 615–623 (2004).
Raymond, G.V., Bauman, M.L. & Kemper, T.L. Hippocampus in autism: a Golgi analysis. Acta Neuropathol. 91, 117–119 (1996).
Larkman, A.U. Dendritic morphology of pyramidal neurones of the visual cortex of the rat: III. Spine distributions. J. Comp. Neurol. 306, 332–343 (1991).
Chen, Z. & Cobb, M.H. Regulation of stress-responsive mitogen-activated protein (MAP) kinase pathways by TAO2. J. Biol. Chem. 276, 16070–16075 (2001).
Chen, Z. et al. TAO (thousand-and-one amino acid) protein kinases mediate signaling from carbachol to p38 mitogen-activated protein kinase and ternary complex factors. J. Biol. Chem. 278, 22278–22283 (2003).
Weiss, L.A. et al. Association between microdeletion and microduplication at 16p11.2 and autism. N. Engl. J. Med. 358, 667–675 (2008).
McCarthy, S.E. et al. Microduplications of 16p11.2 are associated with schizophrenia. Nat. Genet. 41, 1223–1227 (2009).
Darnell, J.C. et al. FMRP stalls ribosomal translocation on mRNAs linked to synaptic function and autism. Cell 146, 247–261 (2011).
Chen, Z., Hutchison, M. & Cobb, M.H. Isolation of the protein kinase TAO2 and identification of its mitogen-activated protein kinase/extracellular signal-regulated kinase kinase binding domain. J. Biol. Chem. 274, 28803–28807 (1999).
Moore, T.M. et al. PSK, a novel STE20-like kinase derived from prostatic carcinoma that activates the c-Jun N-terminal kinase mitogen-activated protein kinase pathway and regulates actin cytoskeletal organization. J. Biol. Chem. 275, 4311–4322 (2000).
Yasuda, S. et al. Activity-induced protocadherin arcadlin regulates dendritic spine number by triggering N-cadherin endocytosis via TAO2beta and p38 MAP kinases. Neuron 56, 456–471 (2007).
Zihni, C. et al. Prostate-derived sterile 20-like kinase 1-alpha induces apoptosis. JNK- and caspase-dependent nuclear localization is a requirement for membrane blebbing. J. Biol. Chem. 282, 6484–6493 (2007).
Gu, C. et al. Neuropilin-1 conveys semaphorin and VEGF signaling during neural and cardiovascular development. Dev. Cell 5, 45–57 (2003).
Tran, T.S. et al. Secreted semaphorins control spine distribution and morphogenesis in the postnatal CNS. Nature 462, 1065–1069 (2009).
Chen, G. et al. Semaphorin-3A guides radial migration of cortical neurons during development. Nat. Neurosci. 11, 36–44 (2008).
Fenstermaker, V., Chen, Y., Ghosh, A. & Yuste, R. Regulation of dendritic length and branching by semaphorin 3A. J. Neurobiol. 58, 403–412 (2004).
Bradke, F. & Dotti, C.G. The role of local actin instability in axon formation. Science 283, 1931–1934 (1999).
King, I. et al. Drosophila tao controls mushroom body development and ethanol-stimulated behavior through par-1. J. Neurosci. 31, 1139–1148 (2011).
Barnes, A.P., Solecki, D. & Polleux, F. New insights into the molecular mechanisms specifying neuronal polarity in vivo. Curr. Opin. Neurobiol. 18, 44–52 (2008).
Witte, H. & Bradke, F. The role of the cytoskeleton during neuronal polarization. Curr. Opin. Neurobiol. 18, 479–487 (2008).
Arimura, N. & Kaibuchi, K. Neuronal polarity: from extracellular signals to intracellular mechanisms. Nat. Rev. Neurosci. 8, 194–205 (2007).
de Anda, F.C., Meletis, K., Ge, X., Rei, D. & Tsai, L.H. Centrosome motility is essential for initial axon formation in the neocortex. J. Neurosci. 30, 10391–10406 (2010).
Zhou, T. et al. Crystal structure of the TAO2 kinase domain: activation and specificity of a Ste20p MAP3K. Structure 12, 1891–1900 (2004).
Romand, S., Wang, Y., Toledo-Rodriguez, M. & Markram, H. Morphological development of thick-tufted layer V pyramidal cells in the rat somatosensory cortex. Front. Neuroanat. 5, 5 (2011).
Polleux, F. & Snider, W. Initiating and growing an axon. Cold Spring Harb. Perspect. Biol. 2, a001925 (2010).
Huangfu, W.C., Omori, E., Akira, S., Matsumoto, K. & Ninomiya-Tsuji, J. Osmotic stress activates the TAK1-JNK pathway while blocking TAK1-mediated NF-κB activation: TAO2 regulates TAK1 pathways. J. Biol. Chem. 281, 28802–28810 (2006).
Ben-Zvi, A. et al. Semaphorin 3A and neurotrophins: a balance between apoptosis and survival signaling in embryonic DRG neurons. J. Neurochem. 96, 585–597 (2006).
Tararuk, T. et al. JNK1 phosphorylation of SCG10 determines microtubule dynamics and axodendritic length. J. Cell Biol. 173, 265–277 (2006).
Bjorkblom, B. et al. Constitutively active cytoplasmic c-Jun N-terminal kinase 1 is a dominant regulator of dendritic architecture: role of microtubule-associated protein 2 as an effector. J. Neurosci. 25, 6350–6361 (2005).
Oliva, A.A. Jr., Atkins, C.M., Copenagle, L. & Banker, G.A. Activated c-Jun N-terminal kinase is required for axon formation. J. Neurosci. 26, 9462–9470 (2006).
Barnat, M. et al. Distinct roles of c-Jun N-terminal kinase isoforms in neurite initiation and elongation during axonal regeneration. J. Neurosci. 30, 7804–7816 (2010).
Rosso, S.B., Sussman, D., Wynshaw-Boris, A. & Salinas, P.C. Wnt signaling through Dishevelled, Rac and JNK regulates dendritic development. Nat. Neurosci. 8, 34–42 (2005).
Chang, L., Jones, Y., Ellisman, M.H., Goldstein, L.S. & Karin, M. JNK1 is required for maintenance of neuronal microtubules and controls phosphorylation of microtubule-associated proteins. Dev. Cell 4, 521–533 (2003).
Jeanneteau, F., Deinhardt, K., Miyoshi, G., Bennett, A.M. & Chao, M.V. The MAP kinase phosphatase MKP-1 regulates BDNF-induced axon branching. Nat. Neurosci. 13, 1373–1379 (2010).
Lei, K. et al. The Bax subfamily of Bcl2-related proteins is essential for apoptotic signal transduction by c-Jun NH2-terminal kinase. Mol. Cell Biol. 22, 4929–4942 (2002).
Yamasaki, T. et al. Stress-activated protein kinase MKK7 regulates axon elongation in the developing cerebral cortex. J. Neurosci. 31, 16872–16883 (2011).
Jan, Y.N. & Jan, L.Y. Branching out: mechanisms of dendritic arborization. Nat. Rev. Neurosci. 11, 316–328 (2010).
Baas, P.W., Black, M.M. & Banker, G.A. Changes in microtubule polarity orientation during the development of hippocampal neurons in culture. J. Cell Biol. 109, 3085–3094 (1989).
Kwan, A.C., Dombeck, D.A. & Webb, W.W. Polarized microtubule arrays in apical dendrites and axons. Proc. Natl. Acad. Sci. USA 105, 11370–11375 (2008).
Horton, A.C. et al. Polarized secretory trafficking directs cargo for asymmetric dendrite growth and morphogenesis. Neuron 48, 757–771 (2005).
Shelly, M. et al. Semaphorin3A regulates neuronal polarization by suppressing axon formation and promoting dendrite growth. Neuron 71, 433–446 (2011).
Nishiyama, M. et al. Semaphorin 3A induces CaV2.3 channel-dependent conversion of axons to dendrites. Nat. Cell Biol. 13, 676–685 (2011).
Polleux, F., Morrow, T. & Ghosh, A. Semaphorin 3A is a chemoattractant for cortical apical dendrites. Nature 404, 567–573 (2000).
Geschwind, D.H. & Levitt, P. Autism spectrum disorders: developmental disconnection syndromes. Curr. Opin. Neurobiol. 17, 103–111 (2007).
Just, M.A., Cherkassky, V.L., Keller, T.A., Kana, R.K. & Minshew, N.J. Functional and anatomical cortical underconnectivity in autism: evidence from an FMRI study of an executive function task and corpus callosum morphometry. Cereb. Cortex 17, 951–961 (2007).
Walsh, C.A., Morrow, E.M. & Rubenstein, J.L. Autism and brain development. Cell 135, 396–400 (2008).
Weiss, L.A., Arking, D.E., Daly, M.J. & Chakravarti, A. A genome-wide linkage and association scan reveals novel loci for autism. Nature 461, 802–808 (2009).
Pinto, D. et al. Functional impact of global rare copy number variation in autism spectrum disorders. Nature 466, 368–372 (2010).
Kelleher, R.J. III & Bear, M.F. The autistic neuron: troubled translation? Cell 135, 401–406 (2008).
Mao, Y. et al. Disrupted in schizophrenia 1 regulates neuronal progenitor proliferation via modulation of GSK3beta/beta-catenin signaling. Cell 136, 1017–1031 (2009).
Acknowledgements
We thank A. Mungenast, E.J. Kwon, M.H. Cobb (University of Texas Southwestern Medical Center) and Y. Gotoh (University of Tokyo) for critical reading of the manuscript, providing vectors, antibodies, and suggestions. The Simons Foundation Autism Research Initiative supported this work. F.C.d.A. is supported by a postdoctoral fellowship from the Simons Foundation (Simons Center for the Social Brain, Massachusetts Institute of Technology). US National Institutes of Health grant R01 MH59199 to A.L.K. and D.D.G. supported this work. L.-H.T., A.L.K. and D.D.G. are investigators of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
This study was designed, directed and coordinated by F.C.d.A. and L.-H.T. L.-H.T., as the principal investigator, provided conceptual and technical guidance for all aspects of the project. F.C.d.A. planned and performed the in utero electroporations and analyzed the data with A.L.R. and O.D. F.C.d.A. performed and analyzed the immunohistochemistry experiments. K.M. generated and characterized the shRNA constructs. K.M., A.L.R. and D.R. contributed to the neuronal cultures. T.T. performed and analyzed the data from the neuronal cultures of Nrp1Sema− mice. A.L.R., O.D., J.G. and R.M. contributed to the biochemistry experiments. T.S. generated the lentiviral shRNA construct and produced the virus particles. D.D.G. and A.L.K. provided the Nrp1Sema− mouse brains and suggested and commented on the design of the experiments. The manuscript was written by F.C.d.A. and L.-H.T. and commented on by all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 4521 kb)
Rights and permissions
About this article
Cite this article
Calderon de Anda, F., Rosario, A., Durak, O. et al. Autism spectrum disorder susceptibility gene TAOK2 affects basal dendrite formation in the neocortex. Nat Neurosci 15, 1022–1031 (2012). https://doi.org/10.1038/nn.3141
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3141
This article is cited by
-
Dissecting 16p11.2 hemi-deletion to study sex-specific striatal phenotypes of neurodevelopmental disorders
Molecular Psychiatry (2024)
-
Identifying foetal forebrain interneurons as a target for monogenic autism risk factors and the polygenic 16p11.2 microdeletion
BMC Neuroscience (2023)
-
Human pluripotent stem cell (hPSC) and organoid models of autism: opportunities and limitations
Translational Psychiatry (2023)
-
Genetic aetiologies for childhood speech disorder: novel pathways co-expressed during brain development
Molecular Psychiatry (2023)
-
WDFY3 mutation alters laminar position and morphology of cortical neurons
Molecular Autism (2022)