Abstract
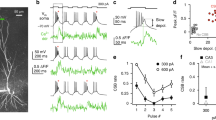
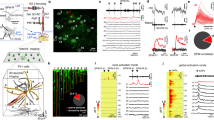
CA3 pyramidal neurons are important for memory formation and pattern completion in the hippocampal network. It is generally thought that proximal synapses from the mossy fibers activate these neurons most efficiently, whereas distal inputs from the perforant path have a weaker modulatory influence. We used confocally targeted patch-clamp recording from dendrites and axons to map the activation of rat CA3 pyramidal neurons at the subcellular level. Our results reveal two distinct dendritic domains. In the proximal domain, action potentials initiated in the axon backpropagate actively with large amplitude and fast time course. In the distal domain, Na+ channel–mediated dendritic spikes are efficiently initiated by waveforms mimicking synaptic events. CA3 pyramidal neuron dendrites showed a high Na+-to-K+ conductance density ratio, providing ideal conditions for active backpropagation and dendritic spike initiation. Dendritic spikes may enhance the computational power of CA3 pyramidal neurons in the hippocampal network.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
McNaughton, B.L. & Morris, R.G.M. Hippocampal synaptic enhancement and information storage within a distributed memory system. Trends Neurosci. 10, 408–415 (1987).
Lisman, J.E. Relating hippocampal circuitry to function: recall of memory sequences by reciprocal dentate–CA3 interactions. Neuron 22, 233–242 (1999).
Nakazawa, K. et al. Requirement for hippocampal CA3 NMDA receptors in associative memory recall. Science 297, 211–218 (2002).
Nakazawa, K., McHugh, T.J., Wilson, M.A. & Tonegawa, S. NMDA receptors, place cells and hippocampal spatial memory. Nat. Rev. Neurosci. 5, 361–372 (2004).
Marr, D. Simple memory: a theory for archicortex. Phil. Trans. R. Soc. Lond. B 262, 23–81 (1971).
Pelkey, K.A. & McBain, C.J. How to dismantle a detonator synapse. Neuron 45, 327–329 (2005).
Henze, D.A., Wittner, L. & Buzsáki, G. Single granule cells reliably discharge targets in the hippocampal CA3 network in vivo. Nat. Neurosci. 5, 790–795 (2002).
Bi, G.-Q. & Poo, M.-M. Synaptic modifications in cultured hippocampal neurons: Dependence on spike timing, synaptic strength, and postsynaptic cell type. J. Neurosci. 18, 10464–10472 (1998).
Magee, J.C. & Johnston, D. A synaptically controlled, associative signal for Hebbian plasticity in hippocampal neurons. Science 275, 209–213 (1997).
Debanne, D., Gähwiler, B.H. & Thompson, S.M. Long-term synaptic plasticity between pairs of individual CA3 pyramidal cells in rat hippocampal slice cultures. J. Physiol. (Lond.) 507, 237–247 (1998).
Hafting, T., Fyhn, M., Molden, S., Moser, M.B. & Moser, E.I. Microstructure of a spatial map in the entorhinal cortex. Nature 436, 801–806 (2005).
Stuart, G.J. & Sakmann, B. Active propagation of somatic action potentials into neocortical pyramidal cell dendrites. Nature 367, 69–72 (1994).
Spruston, N., Schiller, Y., Stuart, G. & Sakmann, B. Activity-dependent action potential invasion and calcium influx into hippocampal CA1 dendrites. Science 268, 297–300 (1995).
Hoffman, D.A., Magee, J.C., Colbert, C.M. & Johnston, D. K+ channel regulation of signal propagation in dendrites of hippocampal pyramidal neurons. Nature 387, 869–875 (1997).
Korngreen, A. & Sakmann, B. Voltage-gated K+ channels in layer 5 neocortical pyramidal neurones from young rats: subtypes and gradients. J. Physiol. (Lond.) 525, 621–639 (2000).
Stuart, G., Spruston, N. & Häusser, M. (eds.). Dendrites 2nd edn. (Oxford University Press, Oxford, 2007).
Bischofberger, J., Engel, D., Li, L., Geiger, J.R.P. & Jonas, P. Patch-clamp recording from mossy fiber terminals in hippocampal slices. Nat. Protoc. 1, 2075–2081 (2006).
Nevian, T., Larkum, M.E., Polsky, A. & Schiller, J. Properties of basal dendrites of layer 5 pyramidal neurons: a direct patch-clamp recording study. Nat. Neurosci. 10, 206–214 (2007).
Larkum, M.E., Nevian, T., Sandler, M., Polsky, A. & Schiller, J. Synaptic integration in tuft dendrites of layer 5 pyramidal neurons: a new unifying principle. Science 325, 756–760 (2009).
Hu, H., Martina, M. & Jonas, P. Dendritic mechanisms underlying rapid synaptic activation of fast-spiking hippocampal interneurons. Science 327, 52–58 (2010).
Shu, Y., Hasenstaub, A., Duque, A., Yu, Y. & McCormick, D.A. Modulation of intracortical synaptic potentials by presynaptic somatic membrane potential. Nature 441, 761–765 (2006).
Spruston, N. & Johnston, D. Perforated patch-clamp analysis of the passive membrane properties of three classes of hippocampal neurons. J. Neurophysiol. 67, 508–529 (1992).
Colbert, C.M. & Johnston, D. Axonal action-potential initiation and Na+ channel densities in the soma and axon initial segment of subicular pyramidal neurons. J. Neurosci. 16, 6676–6686 (1996).
Meeks, J.P. & Mennerick, S. Action potential initiation and propagation in CA3 pyramidal axons. J. Neurophysiol. 97, 3460–3472 (2007).
Traub, R.D. & Miles, R. Neuronal Networks of the Hippocampus (Cambridge University Press, Cambridge, 1991).
Jaffe, D.B. & Carnevale, N.T. Passive normalization of synaptic integration influenced by dendritic architecture. J. Neurophysiol. 82, 3268–3285 (1999).
Golding, N.L. & Spruston, N. Dendritic sodium spikes are variable triggers of axonal action potentials in hippocampal CA1 pyramidal neurons. Neuron 21, 1189–1200 (1998).
Kamondi, A., Acsády, L. & Buzsáki, G. Dendritic spikes are enhanced by cooperative network activity in the intact hippocampus. J. Neurosci. 18, 3919–3928 (1998).
Golding, N.L., Staff, N.P. & Spruston, N. Dendritic spikes as a mechanism for cooperative long-term potentiation. Nature 418, 326–331 (2002).
Gasparini, S., Migliore, M. & Magee, J.C. On the initiation and propagation of dendritic spikes in CA1 pyramidal neurons. J. Neurosci. 24, 11046–11056 (2004).
Losonczy, A., Makara, J.K. & Magee, J.C. Compartmentalized dendritic plasticity and input feature storage in neurons. Nature 452, 436–441 (2008).
Rall, W. Core conductor theory and cable properties of neurons. in Handbook of Physiology, The Nervous System, Cellular Biology of Neurones (ed. Kandel, E.R.) 39–98 (American Physiological Society, Bethesda, Maryland, 1977).
Spruston, N. Pyramidal neurons: dendritic structure and synaptic integration. Nat. Rev. Neurosci. 9, 206–221 (2008).
Krueppel, R., Remy, S. & Beck, H. Dendritic integration in hippocampal dentate granule cells. Neuron 71, 512–528 (2011).
Frick, A., Magee, J., Koester, H.J., Migliore, M. & Johnston, D. Normalization of Ca2+ signals by small oblique dendrites of CA1 pyramidal neurons. J. Neurosci. 23, 3243–3250 (2003).
Holmes, W.R. The role of dendritic diameters in maximizing the effectiveness of synaptic inputs. Brain Res. 478, 127–137 (1989).
Major, G., Larkman, A.U., Jonas, P., Sakmann, B. & Jack, J.J.B. Detailed passive cable models of whole-cell recorded CA3 pyramidal neurons in rat hippocampal slices. J. Neurosci. 14, 4613–4638 (1994).
Jarsky, T., Roxin, A., Kath, W.L. & Spruston, N. Conditional dendritic spike propagation following distal synaptic activation of hippocampal CA1 pyramidal neurons. Nat. Neurosci. 8, 1667–1676 (2005).
Softky, W. Sub-millisecond coincidence detection in active dendritic trees. Neuroscience 58, 13–41 (1994).
Poirazi, P. & Mel, B.W. Impact of active dendrites and structural plasticity on the memory capacity of neural tissue. Neuron 29, 779–796 (2001).
Kampa, B.M., Clements, J., Jonas, P. & Stuart, G.J. Kinetics of Mg2+ unblock of NMDA receptors: implications for spike timing–dependent synaptic plasticity. J. Physiol. (Lond.) 556, 337–345 (2004).
McMahon, D.B.T. & Barrionuevo, G. Short- and long-term plasticity of the perforant path synapse in hippocampal area CA3. J. Neurophysiol. 88, 528–533 (2002).
Kobayashi, K. & Poo, M.-M. Spike train timing–dependent associative modification of hippocampal CA3 recurrent synapses by mossy fibers. Neuron 41, 445–454 (2004).
Urban, N.N., Henze, D.A. & Barrionuevo, G. Amplification of perforant-path EPSPs in CA3 pyramidal cells by LVA calcium and sodium channels. J. Neurophysiol. 80, 1558–1561 (1998).
Urban, N.N. & Barrionuevo, G. Active summation of excitatory postsynaptic potentials in hippocampal CA3 pyramidal neurons. Proc. Natl. Acad. Sci. USA 95, 11450–11455 (1998).
Davie, J.T. et al. Dendritic patch-clamp recording. Nat. Protoc. 1, 1235–1247 (2006).
Spruston, N., Jonas, P. & Sakmann, B. Dendritic glutamate receptor channels in rat hippocampal CA3 and CA1 pyramidal neurons. J. Physiol. (Lond.) 482, 325–352 (1995).
Jonas, P., Major, G. & Sakmann, B. Quantal components of unitary EPSCs at the mossy fibre synapse on CA3 pyramidal cells of rat hippocampus. J. Physiol. (Lond.) 472, 615–663 (1993).
Lien, C.C. & Jonas, P. Kv3 potassium conductance is necessary and kinetically optimized for high frequency action potential generation in hippocampal interneurons. J. Neurosci. 23, 2058–2068 (2003).
Chicurel, M.E. & Harris, K.M. Three-dimensional analysis of the structure and composition of CA3 branched dendritic spines and their synaptic relationships with mossy fiber boutons in the rat hippocampus. J. Comp. Neurol. 325, 169–182 (1992).
Song, W.-J. et al. Somatodendritic depolarization-activated potassium currents in rat neostriatal cholinergic interneurons are predominantly of the A type and attributable to coexpression of Kv4.2 and Kv4.1 subunits. J. Neurosci. 18, 3124–3137 (1998).
Sakmann, B. & Neher, E. Geometric parameters of pipettes and membrane patches. in Single-Channel Recording (eds. Sakmann, B. & Neher, E.) (Plenum Press, New York, 1995).
Acknowledgements
We thank G. Buzsáki and J. Lisman for critically reading previous manuscript versions. We also thank F. Marr and I. Koeva for technical assistance and E. Kramberger for perfect editorial support. This work was supported by the Deutsche Forschungsgemeinschaft (TR 3/B10) and the European Union (European Research Council Advanced grant to P.J.).
Author information
Authors and Affiliations
Contributions
S.K. performed the experiments and analyzed the data. H.H. and S.J.G. contributed to initial experiments. P.J. analyzed data and wrote the paper. All of the authors revised the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Tables 1–4 (PDF 1622 kb)
Rights and permissions
About this article
Cite this article
Kim, S., Guzman, S., Hu, H. et al. Active dendrites support efficient initiation of dendritic spikes in hippocampal CA3 pyramidal neurons. Nat Neurosci 15, 600–606 (2012). https://doi.org/10.1038/nn.3060
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3060
This article is cited by
-
Distinguishing examples while building concepts in hippocampal and artificial networks
Nature Communications (2024)
-
Dendritic Morphology Affects the Velocity and Amplitude of Back-propagating Action Potentials
Neuroscience Bulletin (2022)
-
Transsynaptic modulation of presynaptic short-term plasticity in hippocampal mossy fiber synapses
Nature Communications (2021)
-
Illuminating dendritic function with computational models
Nature Reviews Neuroscience (2020)
-
Diverse synaptic and dendritic mechanisms of complex spike burst generation in hippocampal CA3 pyramidal cells
Nature Communications (2019)