Abstract
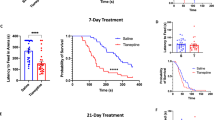
Neurotransmitter transporters can affect neuronal excitability indirectly via modulation of neurotransmitter concentrations or directly via transporter currents. A physiological or pathophysiological role for transporter currents has not been described. We found that GABA transporter 1 (GAT-1) cation currents directly increased GABAergic neuronal excitability and synaptic GABA release in the periaqueductal gray (PAG) during opioid withdrawal in rodents. In contrast, GAT-1 did not indirectly alter GABA receptor responses via modulation of extracellular GABA concentrations. Notably, we found that GAT-1–induced increases in GABAergic activity contributed to many PAG-mediated signs of opioid withdrawal. Together, these data support the hypothesis that GAT-1 activity directly produces opioid withdrawal signs through direct hyperexcitation of GABAergic PAG neurons and nerve terminals, which presumably enhances GABAergic inhibition of PAG output neurons. These data provide, to the best of our knowledge, the first evidence that dysregulation of a neurotransmitter transporter current is important for the maladaptive plasticity that underlies opiate withdrawal.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Veruki, M.L., Morkve, S.H. & Hartveit, E. Activation of a presynaptic glutamate transporter regulates synaptic transmission through electrical signaling. Nat. Neurosci. 9, 1388–1396 (2006).
Bagley, E.E., Gerke, M.B., Vaughan, C.W., Hack, S.P. & Christie, M.J. GABA transporter currents activated by protein kinase A excite midbrain neurons during opioid withdrawal. Neuron 45, 433–445 (2005).
Ingram, S.L., Prasad, B.M. & Amara, S.G. Dopamine transporter–mediated conductances increase excitability of midbrain dopamine neurons. Nat. Neurosci. 5, 971–978 (2002).
Mann, J.J. The medical management of depression. N. Engl. J. Med. 353, 1819–1834 (2005).
Cushman, P. & Dole, V.P. Detoxification of rehabilitated methadone-maintained patients. J. Am. Med. Assoc. 226, 747–752 (1973).
Bozarth, M.A. & Wise, R.A. Anatomically distinct opiate receptor fields mediate reward and physical dependence. Science 224, 516–517 (1984).
Laschka, E., Teschemacher, H., Mehraein, P. & Herz, A. Sites of action of morphine involved in the development of physical dependence in rats. II. Morphine withdrawal precipitated by application of morphine antagonists into restricted parts of the ventricular system and by microinjection into various brain areas. Psychopharmacologia 46, 141–147 (1976).
Williams, J.T., Christie, M.J. & Manzoni, O. Cellular and synaptic adaptations mediating opioid dependence. Physiol. Rev. 81, 299–343 (2001).
Chieng, B. & Christie, M.J. Local opioid withdrawal in rat single periaqueductal gray neurons in vitro. J. Neurosci. 16, 7128–7136 (1996).
Ingram, S.L., Vaughan, C.W., Bagley, E.E., Connor, M. & Christie, M.J. Enhanced opioid efficacy in opioid dependence is caused by an altered signal transduction pathway. J. Neurosci. 18, 10269–10276 (1998).
Chieng, B.C., Hallberg, C., Nyberg, F.J. & Christie, M.J. Enhanced c-Fos in periaqueductal grey GABAergic neurons during opioid withdrawal. Neuroreport 16, 1279–1283 (2005).
Hacker, J., Pedersen, N.P., Chieng, B.C., Keay, K.A. & Christie, M.J. Enhanced Fos expression in glutamic acid decarboxylase immunoreactive neurons of the mouse periaqueductal grey during opioid withdrawal. Neuroscience 137, 1389–1396 (2006).
Tamamaki, N. et al. Green fluorescent protein expression and colocalization with calretinin, parvalbumin and somatostatin in the GAD67-GFP knock-in mouse. J. Comp. Neurol. 467, 60–79 (2003).
Cherubini, E. & Conti, F. Generating diversity at GABAergic synapses. Trends Neurosci. 24, 155–162 (2001).
Barbaresi, P., Gazzanelli, G. & Malatesta, M. γ-Aminobutyric acid transporters in the cat periaqueductal gray: a light and electron microscopic immunocytochemical study. J. Comp. Neurol. 429, 337–354 (2001).
Hack, S.P., Vaughan, C.W. & Christie, M.J. Modulation of GABA release during morphine withdrawal in midbrain neurons in vitro. Neuropharmacology 45, 575–584 (2003).
Jensen, K., Chiu, C.S., Sokolova, I., Lester, H.A. & Mody, I. GABA transporter-1 (GAT1)-deficient mice: differential tonic activation of GABAA versus GABAB receptors in the hippocampus. J. Neurophysiol. 90, 2690–2701 (2003).
Vaughan, C.W., Ingram, S.L., Connor, M.A. & Christie, M.J. How opioids inhibit GABA-mediated neurotransmission. Nature 390, 611–614 (1997).
Kullmann, D.M. et al. Presynaptic, extrasynaptic and axonal GABAA receptors in the CNS: where and why? Prog. Biophys. Mol. Biol. 87, 33–46 (2005).
Chiu, C.S. et al. GABA transporter deficiency causes tremor, ataxia, nervousness and increased GABA-induced tonic conductance in cerebellum. J. Neurosci. 25, 3234–3245 (2005).
Koob, G.F., Maldonado, R. & Stinus, L. Neural substrates of opiate withdrawal. Trends Neurosci. 15, 186–191 (1992).
DiStefano, P.S. & Brown, O.M. Biochemical correlates of morphine withdrawal. 2. Effects of clonidine. J. Pharmacol. Exp. Ther. 233, 339–344 (1985).
Itouji, A., Sakai, N., Tanaka, C. & Saito, N. Neuronal and glial localization of two GABA transporters (GAT1 and GAT3) in the rat cerebellum. Brain Res. Mol. Brain Res. 37, 309–316 (1996).
Overstreet, L.S. & Westbrook, G. Synapse density regulates independence at unitary inhibitory synapses. J. Neurosci. 23, 2618–2626 (2003).
Belelli, D. et al. Extrasynaptic GABAA receptors: form, pharmacology and function. J. Neurosci. 29, 12757–12763 (2009).
Yang, K., Furue, H., Kumamoto, E., Dong, Y.X. & Yoshimura, M. Pre- and postsynaptic inhibition mediated by GABAB receptors in rat ventrolateral periaqueductal gray neurons. Biochem. Biophys. Res. Commun. 302, 233–237 (2003).
Luer, M.S. & Rhoney, D.H. Tiagabine: a novel antiepileptic drug. Ann. Pharmacother. 32, 1173–1180 (1998).
González, G. et al. Clinical efficacy of gabapentin versus tiagabine for reducing cocaine use among cocaine-dependent methadone-treated patients. Drug Alcohol Depend. 87, 1–9 (2007).
Minozzi, S. et al. Anticonvulsants for cocaine dependence. Cochrane Database Syst. Rev. 16, CD006754 (2008).
Sofuoglu, M., Mouratidis, M., Yoo, S., Culligan, K. & Kosten, T. Effects of tiagabine in combination with intravenous nicotine in overnight abstinent smokers. Psychopharmacology (Berl.) 181, 504–510 (2005).
Myrick, H. et al. A retrospective chart review comparing tiagabine and benzodiazepines for the treatment of alcohol withdrawal. J. Psychoactive Drugs 37, 409–414 (2005).
Nestler, E.J. Historical review: molecular and cellular mechanisms of opiate and cocaine addiction. Trends Pharmacol. Sci. 25, 210–218 (2004).
Hanoune, J. & Defer, N. Regulation and role of adenylyl cyclase isoforms. Annu. Rev. Pharmacol. Toxicol. 41, 145–174 (2001).
Tzavara, E.T., Monory, K., Hanoune, J. & Nomikos, G.G. Nicotine withdrawal syndrome: behavioral distress and selective up-regulation of the cyclic AMP pathway in the amygdala. Eur. J. Neurosci. 16, 149–153 (2002).
Chieng, B. & Williams, J.T. Increased opioid inhibition of GABA release in nucleus accumbens during morphine withdrawal. J. Neurosci. 18, 7033–7039 (1998).
Bonci, A. & Williams, J.T. Increased probability of GABA release during withdrawal from morphine. J. Neurosci. 17, 796–803 (1997).
Paxinos, G. & Franklin, K.B.J. The Mouse Brain in Stereotaxic Co-ordinates, 2nd edn. (Academic Press, Sydney, 2001).
Paxinos, G. & Watson, C. The Rat Brain in Stereotaxic Co-ordinates, 4thedn. (Academic Press, Sydney, 1998).
Chefer, V.I., Denoroy, L., Zapata, A. & Shippenberg, T.S. Mu opioid receptor modulation of somatodendritic dopamine overflow: GABAergic and glutamatergic mechanisms. Eur. J. Neurosci. 30, 272–278 (2009).
McNally, G.P., Pigg, M. & Weidemann, G. Opioid receptors in the midbrain periaqueductal gray regulate extinction of pavlovian fear conditioning. J. Neurosci. 24, 6912–6919 (2004).
Acknowledgements
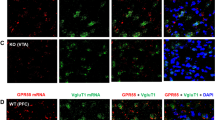
We thank Y. Azriel for preparing the image shown in Figure 1a, and T. Kaneko (Kyoto University) for donating Gad67-GFP mice. This work was supported by the National Health and Medical Research Council of Australia and the National Institute on Drug Abuse Intramural Research Program of the US National Institutes of Health.
Author information
Authors and Affiliations
Contributions
E.E.B. designed and performed all of the electrophysiological experiments, with the exception of some of the IPSC experiments, contributed to research design and prepared the manuscript, with contributions from all of the other authors. C.M. performed and analyzed some of the IPSC experiments. J.H. and J.P. performed immunohistochemical and behavioral studies under the supervision and with the direct assistance of B.C.H.C., M.J.C. and G.P.M. G.P.M. also performed and supervised the microinjection studies. V.I.C. and T.S.S. designed and performed all of the microdialysis experiments. M.J.C. contributed to experimental design, supervision, coordinated experimental approaches and preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 and Supplementary Table 1 (PDF 567 kb)
Rights and permissions
About this article
Cite this article
Bagley, E., Hacker, J., Chefer, V. et al. Drug-induced GABA transporter currents enhance GABA release to induce opioid withdrawal behaviors. Nat Neurosci 14, 1548–1554 (2011). https://doi.org/10.1038/nn.2940
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2940
This article is cited by
-
Opioid withdrawal: role in addiction and neural mechanisms
Psychopharmacology (2023)
-
FORWARDS-1: an adaptive, single-blind, placebo-controlled ascending dose study of acute baclofen on safety parameters in opioid dependence during methadone-maintenance treatment—a pharmacokinetic-pharmacodynamic study
Trials (2022)
-
Cell type-specific modulation of sensory and affective components of itch in the periaqueductal gray
Nature Communications (2019)
-
GABA Uptake Inhibition Reduces In Vivo Extraction Fraction in the Ventral Tegmental Area of Long Evans Rats Measured by Quantitative Microdialysis Under Transient Conditions
Neurochemical Research (2018)
-
Blocking microglial pannexin-1 channels alleviates morphine withdrawal in rodents
Nature Medicine (2017)