Abstract
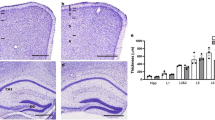
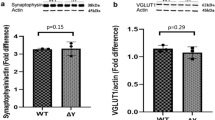
Major histocompatibility complex class I (MHCI) molecules modulate activity-dependent refinement and plasticity. We found that MHCI also negatively regulates the density and function of cortical synapses during their initial establishment both in vitro and in vivo. MHCI molecules are expressed on cortical neurons before and during synaptogenesis. In vitro, decreasing surface MHCI (sMHCI) on neurons increased glutamatergic and GABAergic synapse density, whereas overexpression decreased it. In vivo, synapse density was higher throughout development in β2m−/− mice. MHCI also negatively regulated the strength of excitatory, but not inhibitory, synapses and controlled the balance of excitation and inhibition onto cortical neurons. sMHCI levels were modulated by activity and were necessary for activity to negatively regulate glutamatergic synapse density. Finally, acute changes in sMHCI and activity altered synapse density exclusively during early postnatal development. These results identify a previously unknown function for immune proteins in the negative regulation of the initial establishment and function of cortical connections.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Waites, C.L., Craig, A.M. & Garner, C.C. Mechanisms of vertebrate synaptogenesis. Annu. Rev. Neurosci. 28, 251–274 (2005).
McAllister, A.K. Dynamic aspects of CNS synapse formation. Annu. Rev. Neurosci. 30, 425–450 (2007).
Flavell, S.W. et al. Activity-dependent regulation of MEF2 transcription factors suppresses excitatory synapse number. Science 311, 1008–1012 (2006).
Margolis, S.S. et al. EphB-mediated degradation of the RhoA GEF Ephexin5 relieves a developmental brake on excitatory synapse formation. Cell 143, 442–455 (2010).
O'Connor, T.P. et al. Semaphorin 5B mediates synapse elimination in hippocampal neurons. Neural Dev. 4, 18 (2009).
Patel, M.R. & Shen, K. RSY-1 is a local inhibitor of presynaptic assembly in C. elegans. Science 323, 1500–1503 (2009).
Boulanger, L.M. & Shatz, C.J. Immune signaling in neural development, synaptic plasticity and disease. Nat. Rev. Neurosci. 5, 521–531 (2004).
Corriveau, R.A., Huh, G.S. & Shatz, C.J. Regulation of class I MHC gene expression in the developing and mature CNS by neural activity. Neuron 21, 505–520 (1998).
Huh, G.S. et al. Functional requirement for class I MHC in CNS development and plasticity. Science 290, 2155–2159 (2000).
Needleman, L.A., Liu, X.B., El-Sabeawy, F., Jones, E.G. & McAllister, A.K. MHC class I molecules are present both pre- and postsynaptically in the visual cortex during postnatal development and in adulthood. Proc. Natl. Acad. Sci. USA 107, 16999–17004 (2010).
Spiliotis, E.T., Pentcheva, T. & Edidin, M. Probing for membrane domains in the endoplasmic reticulum: retention and degradation of unassembled MHC class I molecules. Mol. Biol. Cell 13, 1566–1581 (2002).
Williams, D.B., Barber, B.H., Flavell, R.A. & Allen, H. Role of beta 2-microglobulin in the intracellular transport and surface expression of murine class I histocompatibility molecules. J. Immunol. 142, 2796–2806 (1989).
Datwani, A. et al. Classical MHCI molecules regulate retinogeniculate refinement and limit ocular dominance plasticity. Neuron 64, 463–470 (2009).
Goddard, C.A., Butts, D.A. & Shatz, C.J. Regulation of CNS synapses by neuronal MHC class I. Proc. Natl. Acad. Sci. USA 104, 6828–6833 (2007).
Glynn, M.W. & McAllister, A.K. Immunocytochemistry and quantification of protein colocalization in cultured neurons. Nat. Protoc. 1, 1287–1296 (2006).
Sabo, S.L. & McAllister, A.K. Mobility and cycling of synaptic protein-containing vesicles in axonal growth cone filopodia. Nat. Neurosci. 6, 1264–1269 (2003).
Schnabl, E. et al. Activated human T lymphocytes express MHC class I heavy chains not associated with beta 2-microglobulin. J. Exp. Med. 171, 1431–1442 (1990).
Madrigal, J.A. et al. Molecular definition of a polymorphic antigen (LA45) of free HLA-A and -B heavy chains found on the surfaces of activated B and T cells. J. Exp. Med. 174, 1085–1095 (1991).
Sassoè-Pognetto, M., Panzanelli, P., Sieghart, W. & Fritschy, J.M. Colocalization of multiple GABA(A) receptor subtypes with gephyrin at postsynaptic sites. J. Comp. Neurol. 420, 481–498 (2000).
DeFelipe, J., Marco, P., Busturia, I. & Merchan-Perez, A. Estimation of the number of synapses in the cerebral cortex: methodological considerations. Cereb. Cortex 9, 722–732 (1999).
Cochilla, A.J., Angleson, J.K. & Betz, W.J. Monitoring secretory membrane with FM1–43 fluorescence. Annu. Rev. Neurosci. 22, 1–10 (1999).
Demaria, S., Schwab, R. & Bushkin, Y. The origin and fate of beta2m-free MHC class I molecules induced on activated T cells. Cell. Immunol. 142, 103–113 (1992).
Chakrabarti, A., Matko, J., Rahman, N.A., Barisas, B.G. & Edidin, M. Self-association of class I major histocompatibility complex molecules in liposome and cell surface membranes. Biochemistry 31, 7182–7189 (1992).
Santos, S.G., Powis, S.J. & Arosa, F.A. Misfolding of major histocompatibility complex class I molecules in activated T cells allows cis-interactions with receptors and signaling molecules and is associated with tyrosine phosphorylation. J. Biol. Chem. 279, 53062–53070 (2004).
Bodnár, A. et al. Class I HLA oligomerization at the surface of B cells is controlled by exogenous beta(2)-microglobulin: implications in activation of cytotoxic T lymphocytes. Int. Immunol. 15, 331–339 (2003).
Hochman, J.H., Shimizu, Y., DeMars, R. & Edidin, M. Specific associations of fluorescent beta-2-microglobulin with cell surfaces. The affinity of different H-2 and HLA antigens for beta-2-microglobulin. J. Immunol. 140, 2322–2329 (1988).
Neumann, H., Schmidt, H., Cavalie, A., Jenne, D. & Wekerle, H. Major histocompatibility complex (MHC) class I gene expression in single neurons of the central nervous system: differential regulation by interferon (IFN)-gamma and tumor necrosis factor (TNF)-alpha. J. Exp. Med. 185, 305–316 (1997).
McAllister, A.K. Dynamic aspects of cortical synaptogenesis. Annu. Rev. Neurosci. (in the press) (2007).
Craig, A.M., Blackstone, C.D., Huganir, R.L. & Banker, G. Selective clustering of glutamate and gamma-aminobutyric acid receptors opposite terminals releasing the corresponding neurotransmitters. Proc. Natl. Acad. Sci. USA 91, 12373–12377 (1994).
Harms, K.J. & Craig, A.M. Synapse composition and organization following chronic activity blockade in cultured hippocampal neurons. J. Comp. Neurol. 490, 72–84 (2005).
Varoqueaux, F. et al. Total arrest of spontaneous and evoked synaptic transmission but normal synaptogenesis in the absence of Munc13-mediated vesicle priming. Proc. Natl. Acad. Sci. USA 99, 9037–9042 (2002).
Verhage, M. et al. Synaptic assembly of the brain in the absence of neurotransmitter secretion. Science 287, 864–869 (2000).
Bouwman, J. et al. Quantification of synapse formation and maintenance in vivo in the absence of synaptic release. Neuroscience 126, 115–126 (2004).
Rao, A. & Craig, A.M. Activity regulates the synaptic localization of the NMDA receptor in hippocampal neurons. Neuron 19, 801–812 (1997).
Lauri, S.E. et al. Activity blockade increases the number of functional synapses in the hippocampus of newborn rats. Mol. Cell. Neurosci. 22, 107–117 (2003).
Wierenga, C.J., Walsh, M.F. & Turrigiano, G.G. Temporal regulation of the expression locus of homeostatic plasticity. J. Neurophysiol. 96, 2127–2133 (2006).
Burrone, J., O'Byrne, M. & Murthy, V.N. Multiple forms of synaptic plasticity triggered by selective suppression of activity in individual neurons. Nature 420, 414–418 (2002).
Burrone, J. & Murthy, V.N. Synaptic gain control and homeostasis. Curr. Opin. Neurobiol. 13, 560–567 (2003).
Huupponen, J., Molchanova, S.M., Taira, T. & Lauri, S.E. Susceptibility for homeostatic plasticity is down-regulated in parallel with maturation of the rat hippocampal synaptic circuitry. J. Physiol. (Lond.) 581, 505–514 (2007).
Kirov, S.A., Goddard, C.A. & Harris, K.M. Age-dependence in the homeostatic upregulation of hippocampal dendritic spine number during blocked synaptic transmission. Neuropharmacology 47, 640–648 (2004).
Patterson, P.H. Maternal infection: window on neuroimmune interactions in fetal brain development and mental illness. Curr. Opin. Neurobiol. 12, 115–118 (2002).
Naranda, T., Goldstein, A. & Olsson, L. A peptide derived from an extracellular domain selectively inhibits receptor internalization: target sequences on insulin and insulin-like growth factor 1 receptors. Proc. Natl. Acad. Sci. USA 94, 11692–11697 (1997).
Olsson, L., Goldstein, A. & Stagsted, J. Regulation of receptor internalization by the major histocompatibility complex class I molecule. Proc. Natl. Acad. Sci. USA 91, 9086–9090 (1994).
Stagsted, J. Journey beyond immunology. Regulation of receptor internalization by major histocompatibility complex class I (MHC-I) and effect of peptides derived from MHC-I. APMIS Supple. 85, 1–40 (1998).
Fishman, D., Elhyany, S. & Segal, S. Non-immune functions of MHC class I glycoproteins in normal and malignant cells. Folia Biol. (Praha) 50, 35–42 (2004).
Abbas, A., Lichtman, A.H. & Pober, J. Cellular and Molecular Immunology (W.E. Saunders Company, Philadelphia, 2000).
Nawa, H., Takahashi, M. & Patterson, P.H. Cytokine and growth factor involvement in schizophrenia: support for the developmental model. Mol. Psychiatry 5, 594–603 (2000).
Shi, L., Fatemi, S.H., Sidwell, R.W. & Patterson, P.H. Maternal influenza infection causes marked behavioral and pharmacological changes in the offspring. J. Neurosci. 23, 297–302 (2003).
Glynn, M.W. & McAllister, A.K. Immunocytochemistry and quantification of protein colocalization in cultured neurons. Nat. Protoc. 1, 1287–1296 (2006).
Hartman, K.N., Pal, S.K., Burrone, J. & Murthy, V.N. Activity-dependent regulation of inhibitory synaptic transmission in hippocampal neurons. Nat. Neurosci. 9, 642–649 (2006).
Acknowledgements
We are grateful to M. Edidin (Johns Hopkins University) for the H2-Kb-CFP construct, J. Fritschy (University of Zurich) for antibodies to GABA receptor and J. Trimmer (University of California Davis) for the antibody to PSD-95. We also thank J. Trimmer and E. Diaz (University of California Davis) for advice and for supporting the project. This project was funded by a National Institute for Environmental Health Sciences Training grant (M.W.G.), the John Merck Fund (A.K.M.), the March of Dimes (A.K.M.), Cure Autism Now (A.K.M.), Autism Speaks (A.K.M.) and National Institute of Neurological Disorders and Stroke grant R01NS060125 (A.K.M.). We are especially grateful to the Higgins and Gassin Family Foundations for their invaluable support of basic science research and our work.
Author information
Authors and Affiliations
Contributions
M.W.G. initiated the project, conducted most of the experiments using ICC to measure glutamatergic synapse density and wrote a draft of the manuscript. B.M.E. worked with X.-B.L. to generate and quantify the electron microscopy images. P.A.G. established whole-cell patch-clamp recording and performed all of the electrophysiology experiments. L.A.N. performed essential control experiments for antibody specificity. F.E.-S. performed all of the ICC experiments on GABAergic synapses and most of the TTX and MHC experiments. All of the authors edited the manuscript. A.K.M. supported all aspects of this project, designed and helped to analyze all experiments and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 (PDF 3612 kb)
Rights and permissions
About this article
Cite this article
Glynn, M., Elmer, B., Garay, P. et al. MHCI negatively regulates synapse density during the establishment of cortical connections. Nat Neurosci 14, 442–451 (2011). https://doi.org/10.1038/nn.2764
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2764
This article is cited by
-
Plasma β2-microglobulin and cerebrospinal fluid biomarkers of Alzheimer’s disease pathology in cognitively intact older adults: the CABLE study
Alzheimer's Research & Therapy (2023)
-
Hypoimmunogenic human pluripotent stem cells are valid cell sources for cell therapeutics with normal self-renewal and multilineage differentiation capacity
Stem Cell Research & Therapy (2023)
-
Neuronal MHC-I complex is destabilized by amyloid-β and its implications in Alzheimer’s disease
Cell & Bioscience (2023)
-
Brain-specific Pd1 deficiency leads to cortical neurogenesis defects and depressive-like behaviors in mice
Cell Death & Differentiation (2023)
-
Outside-in signaling through the major histocompatibility complex class-I cytoplasmic tail modulates glutamate receptor expression in neurons
Scientific Reports (2023)