Abstract
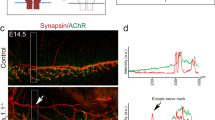
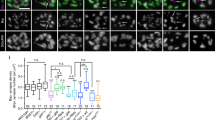
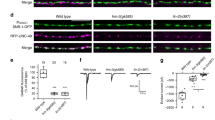
Positive and negative regulation of neurotransmitter receptor aggregation on the postsynaptic membrane is a critical event during synapse formation. Acetylcholine (ACh) and agrin are two opposing signals that regulate ACh receptor (AChR) clustering during neuromuscular junction (NMJ) development. ACh induces dispersion of AChR clusters that are not stabilized by agrin via a cyclin-dependent kinase 5 Cdk5)-mediated mechanism, but regulation of Cdk5 activation is poorly understood. We found that the intermediate filament protein nestin physically interacts with Cdk5 and is required for ACh-induced association of p35, the co-activator of Cdk5, with the muscle membrane. Blockade of nestin-dependent signaling inhibited ACh-induced Cdk5 activation and the dispersion of AChR clusters in cultured myotubes. Similar to the effects of Cdk5 gene inactivation, knockdown of nestin in agrin-deficient mouse embryos ubstantially restored AChR clusters. These results suggest that nestin is required for ACh-induced, Cdk5-dependent dispersion of AChR clusters during NMJ development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
13 February 2011
In the version of this article initially published online, the bottom two panels of Figure 7 were incorrectly labeled. Instead of reading Agrn, they should read Argn–/–. In addition, a grant number was missing from the Acknowledgments. The second sentence of the Acknowledgments should read “This study is supported by US National Institutes of Health grants HD034534, NS047345, NS060833 and NS044420 (K.-F.L.), Muscular Dystrophy Association fellowship MDA4230 (J.Y.), and Academy of Finland, the Research Institute of the Åbo Akademi University Foundation (J.E.E.)." These errors have been corrected for the print, PDF and HTML versions of this article.
References
Wu, H., Xiong, W.C. & Mei, L. To build a synapse: signaling pathways in neuromuscular junction assembly. Development 137, 1017–1033 (2010).
Sanes, J.R. & Lichtman, J.W. Development of the vertebrate neuromuscular junction. Annu. Rev. Neurosci. 22, 389–442 (1999).
Yang, X. et al. Patterning of muscle acetylcholine receptor gene expression in the absence of motor innervation. Neuron 30, 399–410 (2001).
Yang, X., Li, W., Prescott, E.D., Burden, S.J. & Wang, J.C. DNA topoisomerase IIβ and neural development. Science 287, 131–134 (2000).
Lin, W. et al. Neurotransmitter acetylcholine negatively regulates neuromuscular synapse formation by a Cdk5-dependent mechanism. Neuron 46, 569–579 (2005).
Flanagan-Steet, H., Fox, M.A., Meyer, D. & Sanes, J.R. Neuromuscular synapses can form in vivo by incorporation of initially aneural postsynaptic specializations. Development 132, 4471–4481 (2005).
Fu, A.K. et al. Aberrant motor axon projection, acetylcholine receptor clustering, and neurotransmission in cyclin-dependent kinase 5 null mice. Proc. Natl. Acad. Sci. USA 102, 15224–15229 (2005).
Patrick, G.N., Zhou, P., Kwon, Y.T., Howley, P.M. & Tsai, L.H. p35, the neuronal-specific activator of cyclin-dependent kinase 5 (Cdk5) is degraded by the ubiquitin-proteasome pathway. J. Biol. Chem. 273, 24057–24064 (1998).
Gilyarov, A.V. Nestin in central nervous system cells. Neurosci. Behav. Physiol. 38, 165–169 (2008).
Hombach-Klonisch, S. et al. Adult stem cells and their trans-differentiation potential—perspectives and therapeutic applications. J. Mol. Med. 86, 1301–1314 (2008).
Carlsson, L., Li, Z., Paulin, D. & Thornell, L.E. Nestin is expressed during development and in myotendinous and neuromuscular junctions in wild-type and desmin knock-out mice. Exp. Cell Res. 251, 213–223 (1999).
Nazarian, J., Bouri, K. & Hoffman, E.P. Intracellular expression profiling by laser capture microdissection: three novel components of the neuromuscular junction. Physiol. Genomics 21, 70–80 (2005).
Kang, H. et al. Regulation of the intermediate filament protein nestin at rodent neuromuscular junctions by innervation and activity. J. Neurosci. 27, 5948–5957 (2007).
Vaittinen, S. et al. Specific and innervation-regulated expression of the intermediate filament protein nestin at neuromuscular and myotendinous junctions in skeletal muscle. Am. J. Pathol. 154, 591–600 (1999).
Herrmann, H. & Aebi, U. Intermediate filaments and their associates: multi-talented structural elements specifying cytoarchitecture and cytodynamics. Curr. Opin. Cell Biol. 12, 79–90 (2000).
Bloch, R.J. Acetylcholine receptor clustering in rat myotubes: requirement for Ca2+ and effects of drugs which depolymerize microtubules. J. Neurosci. 3, 2670–2680 (1983).
Weston, C., Yee, B., Hod, E. & Prives, J. Agrin-induced acetylcholine receptor clustering is mediated by the small guanosine triphosphatases Rac and Cdc42. J. Cell Biol. 150, 205–212 (2000).
Luo, Z.G. et al. Implication of geranylgeranyltransferase I in synapse formation. Neuron 40, 703–717 (2003).
Luo, Z.G. et al. Regulation of AChR clustering by Dishevelled interacting with MuSK and PAK1. Neuron 35, 489–505 (2002).
Linnoila, J., Wang, Y., Yao, Y. & Wang, Z.Z. A mammalian homolog of Drosophila tumorous imaginal discs, Tid1, mediates agrin signaling at the neuromuscular junction. Neuron 60, 625–641 (2008).
Lee, C.W. et al. Regulation of acetylcholine receptor clustering by ADF/cofilin-directed vesicular trafficking. Nat. Neurosci. 12, 848–856 (2009).
Shi, L. et al. Ephexin1 is required for structural maturation and neurotransmission at the neuromuscular junction. Neuron 65, 204–216 (2010).
Michalczyk, K. & Ziman, M. Nestin structure and predicted function in cellular cytoskeletal organisation. Histol. Histopathol. 20, 665–671 (2005).
Hyder, C.L., Pallari, H.M., Kochin, V. & Eriksson, J.E. Providing cellular signposts—post-translational modifications of intermediate filaments. FEBS Lett. 582, 2140–2148 (2008).
Izawa, I. & Inagaki, M. Regulatory mechanisms and functions of intermediate filaments: a study using site- and phosphorylation state–specific antibodies. Cancer Sci. 97, 167–174 (2006).
Omary, M.B., Ku, N.O., Tao, G.Z., Toivola, D.M. & Liao, J. “Heads and tails” of intermediate filament phosphorylation: multiple sites and functional insights. Trends Biochem. Sci. 31, 383–394 (2006).
Sihag, R.K., Inagaki, M., Yamaguchi, T., Shea, T.B. & Pant, H.C. Role of phosphorylation on the structural dynamics and function of types III and IV intermediate filaments. Exp. Cell Res. 313, 2098–2109 (2007).
Sahlgren, C.M. et al. Cdk5 regulates the organization of Nestin and its association with p35. Mol. Cell. Biol. 23, 5090–5106 (2003).
Helfand, B.T., Chou, Y.H., Shumaker, D.K. & Goldman, R.D. Intermediate filament proteins participate in signal transduction. Trends Cell Biol. 15, 568–570 (2005).
Pallari, H.M. & Eriksson, J.E. Intermediate filaments as signaling platforms. Sci. STKE 2006, pe53 (2006).
Sahlgren, C.M. et al. A nestin scaffold links Cdk5/p35 signaling to oxidant-induced cell death. EMBO J. 25, 4808–4819 (2006).
Kim, S. & Coulombe, P.A. Intermediate filament scaffolds fulfill mechanical, organizational, and signaling functions in the cytoplasm. Genes Dev. 21, 1581–1597 (2007).
Patrick, G.N. et al. Conversion of p35 to p25 deregulates Cdk5 activity and promotes neurodegeneration. Nature 402, 615–622 (1999).
Kesavapany, S. et al. p35/cyclin-dependent kinase 5 phosphorylation of ras guanine nucleotide releasing factor 2 (RasGRF2) mediates Rac-dependent extracellular signal-regulated kinase 1/2 activity, altering RasGRF2 and microtubule-associated protein 1b distribution in neurons. J. Neurosci. 24, 4421–4431 (2004).
Asada, A. et al. Myristoylation of p39 and p35 is a determinant of cytoplasmic or nuclear localization of active cyclin-dependent kinase 5 complexes. J. Neurochem. 106, 1325–1336 (2008).
Ku, N.O., Fu, H. & Omary, M.B. Raf-1 activation disrupts its binding to keratins during cell stress. J. Cell Biol. 166, 479–485 (2004).
Ku, N.O., Michie, S., Resurreccion, E.Z., Broome, R.L. & Omary, M.B. Keratin binding to 14-3-3 proteins modulates keratin filaments and hepatocyte mitotic progression. Proc. Natl. Acad. Sci. USA 99, 4373–4378 (2002).
Perlson, E. et al. Vimentin-dependent spatial translocation of an activated MAP kinase in injured nerve. Neuron 45, 715–726 (2005).
Wu, S. et al. Ultraviolet light inhibits translation through activation of the unfolded protein response kinase PERK in the lumen of the endoplasmic reticulum. J. Biol. Chem. 277, 18077–18083 (2002).
Chen, F. et al. Rapsyn interaction with calpain stabilizes AChR clusters at the neuromuscular junction. Neuron 55, 247–260 (2007).
Bloch, R.J. & Steinbach, J.H. Reversible loss of acetylcholine receptor clusters at the developing rat neuromuscular junction. Dev. Biol. 81, 386–391 (1981).
Lin, S. et al. Muscle-wide secretion of a miniaturized form of neural agrin rescues focal neuromuscular innervation in agrin mutant mice. Proc. Natl. Acad. Sci. USA 105, 11406–11411 (2008).
Hamann, M. et al. Synthesis and release of an acetylcholine-like compound by human myoblasts and myotubes. J. Physiol. (Lond.) 489, 791–803 (1995).
Madhavan, R., Zhao, X.T., Ruegg, M.A. & Peng, H.B. Tyrosine phosphatase regulation of MuSK-dependent acetylcholine receptor clustering. Mol. Cell. Neurosci. 28, 403–416 (2005).
Dong, X.P. et al. Shp2 is dispensable in the formation and maintenance of the neuromuscular junction. Neurosignals 15, 53–63 (2006).
An, M.C. et al. Acetylcholine negatively regulates development of the neuromuscular junction through distinct cellular mechanisms. Proc. Natl. Acad. Sci. USA 107, 10702–10707 (2010).
Tiscornia, G., Singer, O. & Verma, I.M. Design and cloning of lentiviral vectors expressing small interfering RNAs. Nat. Protoc. 1, 234–240 (2006).
Singer, O., Tiscornia, G., Ikawa, M. & Verma, I.M. Rapid generation of knockdown transgenic mice by silencing lentiviral vectors. Nat. Protoc. 1, 286–292 (2006).
Sahlgren, C.M. et al. Mitotic reorganization of the intermediate filament protein nestin involves phosphorylation by cdc2 kinase. J. Biol. Chem. 276, 16456–16463 (2001).
Acknowledgements
We thank L.-H. Tsai for the Cdk5 and p35 constructs and A. Nagy for communicating his unpublished results. This study is supported by US National Institutes of Health grants HD034534, NS047345, NS060833 and NS044420 (K.-F.L.), Muscular Dystrophy Association fellowship MDA4230 (J.Y.), and Academy of Finland, the Research Institute of the Åbo Akademi University (J.E.E.).
Author information
Authors and Affiliations
Contributions
J.Y. designed and performed majority of experiments, conducted data analysis and wrote the manuscript. B.D. conducted the transgenic mouse experiment. F.d.W. conducted the transgenic mouse experiment and assisted in data interpretation. T.W.G. assisted in data interpretation and wrote the manuscript. J.E.E. provided constructs and assisted in data interpretation. K.-F.L. supervised the project, designed experiments and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Table 1 (PDF 256 kb)
Rights and permissions
About this article
Cite this article
Yang, J., Dominguez, B., de Winter, F. et al. Nestin negatively regulates postsynaptic differentiation of the neuromuscular synapse. Nat Neurosci 14, 324–330 (2011). https://doi.org/10.1038/nn.2747
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2747
This article is cited by
-
A systems-approach reveals human nestin is an endothelial-enriched, angiogenesis-independent intermediate filament protein
Scientific Reports (2018)
-
Nestin depletion induces melanoma matrix metalloproteinases and invasion
Laboratory Investigation (2014)
-
Post-translational modifications of intermediate filament proteins: mechanisms and functions
Nature Reviews Molecular Cell Biology (2014)
-
Non-apoptotic role of caspase-3 in synapse refinement
Neuroscience Bulletin (2014)