Abstract
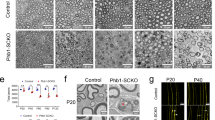
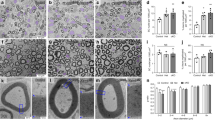
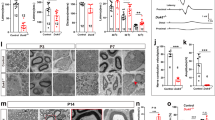
The integrity of peripheral nerves relies on communication between axons and Schwann cells. The axonal signals that ensure myelin maintenance are distinct from those that direct myelination and are largely unknown. Here we show that ablation of the prion protein PrPC triggers a chronic demyelinating polyneuropathy (CDP) in four independently targeted mouse strains. Ablation of the neighboring Prnd locus, or inbreeding to four distinct mouse strains, did not modulate the CDP. CDP was triggered by depletion of PrPC specifically in neurons, but not in Schwann cells, and was suppressed by PrPC expression restricted to neurons but not to Schwann cells. CDP was prevented by PrPC variants that undergo proteolytic amino-proximal cleavage, but not by variants that are nonpermissive for cleavage, including secreted PrPC lacking its glycolipid membrane anchor. These results indicate that neuronal expression and regulated proteolysis of PrPC are essential for myelin maintenance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Aguzzi, A., Baumann, F. & Bremer, J. The prion's elusive reason for being. Annu. Rev. Neurosci. 31, 439–477 (2008).
Brandner, S. et al. Normal host prion protein necessary for scrapie-induced neurotoxicity. Nature 379, 339–343 (1996).
Laurén, J., Gimbel, D.A., Nygaard, H.B., Gilbert, J.W. & Strittmatter, S.M. Cellular prion protein mediates impairment of synaptic plasticity by amyloid-beta oligomers. Nature 457, 1128–1132 (2009).
Büeler, H. et al. Normal development and behavior of mice lacking the neuronal cell-surface PrP protein. Nature 356, 577–582 (1992).
Lledo, P.M., Tremblay, P., Dearmond, S.J., Prusiner, S.B. & Nicoll, R.A. Mice deficient for prion protein exhibit normal neuronal excitability and synaptic transmission in the hippocampus. Proc. Natl. Acad. Sci. USA 93, 2403–2407 (1996).
Collinge, J. et al. Prion protein is necessary for normal synaptic function. Nature 370, 295–297 (1994).
Mallucci, G.R. et al. Postnatal knockout of prion protein alters hippocampal CA1 properties, but does not result in neurodegeneration. EMBO J. 21, 202–210 (2002).
Nazor, K.E., Seward, T. & Telling, G.C. Motor behavioral and neuropathological deficits in mice deficient for normal prion protein expression. Biochim. Biophys. Acta 1772, 645–653 (2007).
Steele, A.D., Lindquist, S. & Aguzzi, A. The prion protein knockout mouse: a phenotype under challenge. Prion 1, 83–93 (2007).
Nishida, N. et al. A mouse prion protein transgene rescues mice deficient for the prion protein gene from Purkinje cell degeneration and demyelination. Lab. Invest. 79, 689–697 (1999).
Heikenwalder, M. et al. Lymphotoxin-dependent prion replication in inflammatory stromal cells of granulomas. Immunity 29, 998–1008 (2008).
Fischer, M. et al. Prion protein (PrP) with amino-proximal deletions restoring susceptibility of PrP knockout mice to scrapie. EMBO J. 15, 1255–1264 (1996).
Moore, R.C. et al. Ataxia in prion protein (PrP)-deficient mice is associated with upregulation of the novel PrP-like protein doppel. J. Mol. Biol. 292, 797–817 (1999).
Genoud, N. et al. Disruption of Doppel prevents neurodegeneration in mice with extensive Prnp deletions. Proc. Natl. Acad. Sci. USA 101, 4198–4203 (2004).
Behrens, A. et al. Absence of the prion protein homologue Doppel causes male sterility. EMBO J. 21, 3652–3658 (2002).
Ogata, T. et al. Opposing extracellular signal-regulated kinase and Akt pathways control Schwann cell myelination. J. Neurosci. 24, 6724–6732 (2004).
Radovanovic, I. et al. Truncated prion protein and Doppel are myelinotoxic in the absence of oligodendrocytic PrPC. J. Neurosci. 25, 4879–4888 (2005).
Prinz, M. et al. Intrinsic resistance of oligodendrocytes to prion infection. J. Neurosci. 24, 5974–5981 (2004).
Polymenidou, M. et al. The POM monoclonals: a comprehensive set of antibodies to non-overlapping prion protein epitopes. PLoS One 3, e3872 (2008).
Watt, N.T. & Hooper, N.M. Reactive oxygen species (ROS)-mediated beta-cleavage of the prion protein in the mechanism of the cellular response to oxidative stress. Biochem. Soc. Trans. 33, 1123–1125 (2005).
Mangé, A. et al. Alpha- and beta- cleavages of the amino-terminus of the cellular prion protein. Biol. Cell 96, 125–132 (2004).
Chesebro, B. et al. Anchorless prion protein results in infectious amyloid disease without clinical scrapie. Science 308, 1435–1439 (2005).
Shmerling, D. et al. Expression of amino-terminally truncated PrP in the mouse leading to ataxia and specific cerebellar lesions. Cell 93, 203–214 (1998).
Mouillet-Richard, S. et al. Signal transduction through prion protein. Science 289, 1925–1928 (2000).
Schneider, B. et al. NADPH oxidase and extracellular regulated kinases 1/2 are targets of prion protein signaling in neuronal and nonneuronal cells. Proc. Natl. Acad. Sci. USA 100, 13326–13331 (2003).
Toni, M. et al. Cellular prion protein and caveolin-1 interaction in a neuronal cell line precedes fyn/erk 1/2 signal transduction. J. Biomed. Biotechnol. 2006, 69469 (2006).
Chen, S., Mange, A., Dong, L., Lehmann, S. & Schachner, M. Prion protein as trans-interacting partner for neurons is involved in neurite outgrowth and neuronal survival. Mol. Cell. Neurosci. 22, 227–233 (2003).
Santuccione, A., Sytnyk, V., Leshchyns′ka, I. & Schachner, M. Prion protein recruits its neuronal receptor NCAM to lipid rafts to activate p59fyn and to enhance neurite outgrowth. J. Cell Biol. 169, 341–354 (2005).
Meotti, F.C. et al. Involvement of cellular prion protein in the nociceptive response in mice. Brain Res. 1151, 84–90 (2007).
Nico, P.B. et al. Altered behavioral response to acute stress in mice lacking cellular prion protein. Behav. Brain Res. 162, 173–181 (2005).
Liu, T. et al. Intercellular transfer of the cellular prion protein. J. Biol. Chem. 277, 47671–47678 (2002).
Nave, K.A. & Trapp, B.D. Axon-glial signaling and the glial support of axon function. Annu. Rev. Neurosci. 31, 535–561 (2008).
Britsch, S. The neuregulin-I/ErbB signaling system in development and disease. Adv. Anat. Embryol. Cell Biol. 190, 1–65 (2007).
Parkin, E.T. et al. Cellular prion protein regulates beta-secretase cleavage of the Alzheimer′s amyloid precursor protein. Proc. Natl. Acad. Sci. USA 104, 11062–11067 (2007).
Hu, X. et al. Bace1 modulates myelination in the central and peripheral nervous system. Nat. Neurosci. 9, 1520–1525 (2006).
Willem, M. et al. Control of peripheral nerve myelination by the beta-secretase BACE1. Science 314, 664–666 (2006).
Rutishauser, D. et al. The comprehensive native interactome of a fully functional tagged prion protein. PLoS One 4, e4446 (2009).
Baumann, F. et al. Lethal recessive myelin toxicity of prion protein lacking its central domain. EMBO J. 26, 538–547 (2007).
McMahon, H.E. et al. Cleavage of the amino terminus of the prion protein by reactive oxygen species. J. Biol. Chem. 276, 2286–2291 (2001).
Sunyach, C., Cisse, M.A., da Costa, C.A., Vincent, B. & Checler, F. The C-terminal products of cellular prion protein processing, C1 and C2, exert distinct influence on p53-dependent staurosporine-induced caspase-3 activation. J. Biol. Chem. 282, 1956–1963 (2007).
Walmsley, A.R., Watt, N.T., Taylor, D.R., Perera, W.S. & Hooper, N.M. Alpha-cleavage of the prion protein occurs in a late compartment of the secretory pathway and is independent of lipid rafts. Mol. Cell. Neurosci. 40, 242–248 (2009).
Isaacs, J.D., Jackson, G.S. & Altmann, D.M. The role of the cellular prion protein in the immune system. Clin. Exp. Immunol. 146, 1–8 (2006).
Manson, J.C. et al. 129/Ola mice carrying a null mutation in PrP that abolishes mRNA production are developmentally normal. Mol. Neurobiol. 8, 121–127 (1994).
Mombaerts, P. et al. RAG-1-deficient mice have no mature B and T lymphocytes. Cell 68, 869–877 (1992).
Lindeboom, F. et al. A tissue-specific knockout reveals that Gata1 is not essential for Sertoli cell function in the mouse. Nucleic Acids Res. 31, 5405–5412 (2003).
Zielasek, J., Martini, R. & Toyka, K.V. Functional abnormalities in P0-deficient mice resemble human hereditary neuropathies linked to P0 gene mutations. Muscle Nerve 19, 946–952 (1996).
Bermingham, J.R. Jr. et al. The claw paw mutation reveals a role for Lgi4 in peripheral nerve development. Nat. Neurosci. 9, 76–84 (2006).
Acknowledgements
We thank M. Delic, R. Moos, D. Goriounov, C. Tostado, H. Mader, K. Nairz, M. Bieri, N. Wey and D. Meijer for methodological advice and technical help. G. Mallucci provided tgNFH-Cre and tgPrnploxP mice, D. Meijer provided tgDhh-Cre mice, J.C. Manson provided PrnpEdbg/Edbg mice, J. Collinge provided Prnpo/o FVB mice, S. Lindquist and W. Jackson provided PrnpGFP/GFP mice and P. Saftig and A. Rittger provided nerves from BACE1−/− mice. We thank W.B. Macklin for the PLP plasmid; T. Rülicke for pronuclear injections and H. Welzl, I. Drescher and S. Wirth for help with behavioral tests. J.A. Girault and M.T. Dours-Zimmermann donated anti-paranodine/Caspr and anti-versican antibodies, respectively. We thank B. Seifert for statistical consulting. A.A. received an ERC Advanced Investigator Grant and grants from the European Union (PRIORITY and LUPAS), the Novartis Foundation and the Swiss National Foundation. J.B. received a Career Development award from the University of Zürich. C.W. and K.V.T. were supported by the Departmental Research Fund.
Author information
Authors and Affiliations
Contributions
J.B. and A.A. designed the study and wrote the manuscript. J.B., F.B., C.T., C.W., H.F., P.S., A.D.S., K.V.T. and J.W. did the experiments. J.B., F.B., C.T., C.W., H.F., A.D.S., K.V.T., K.-A.N., J.W. and A.A. analyzed the data.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–15 and Supplementary Table 1 (PDF 3544 kb)
Rights and permissions
About this article
Cite this article
Bremer, J., Baumann, F., Tiberi, C. et al. Axonal prion protein is required for peripheral myelin maintenance. Nat Neurosci 13, 310–318 (2010). https://doi.org/10.1038/nn.2483
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2483
This article is cited by
-
Cellular toxicity of scrapie prions in prion diseases; a biochemical and molecular overview
Molecular Biology Reports (2023)
-
Epidemiological and clinical characteristics of patients with late-onset Creutzfeldt-Jakob disease
Neurological Sciences (2022)
-
Protein disulfide isomerase ERp57 protects early muscle denervation in experimental ALS
Acta Neuropathologica Communications (2021)
-
Prion type 2 selection in sporadic Creutzfeldt–Jakob disease affecting peripheral ganglia
Acta Neuropathologica Communications (2021)
-
Myelin Biology
Neurotherapeutics (2021)