Abstract
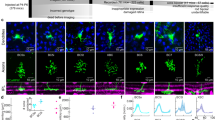
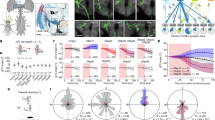
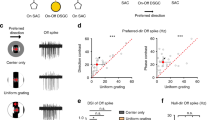
The detection of approaching objects, such as looming predators, is necessary for survival. Which neurons and circuits mediate this function? We combined genetic labeling of cell types, two-photon microscopy, electrophysiology and theoretical modeling to address this question. We identify an approach-sensitive ganglion cell type in the mouse retina, resolve elements of its afferent neural circuit, and describe how these confer approach sensitivity on the ganglion cell. The circuit's essential building block is a rapid inhibitory pathway: it selectively suppresses responses to non-approaching objects. This rapid inhibitory pathway, which includes AII amacrine cells connected to bipolar cells through electrical synapses, was previously described in the context of night-time vision. In the daytime conditions of our experiments, the same pathway conveys signals in the reverse direction. The dual use of a neural pathway in different physiological conditions illustrates the efficiency with which several functions can be accommodated in a single circuit.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Schiff, W., Caviness, J.A. & Gibson, J.J. Persistent fear responses in rhesus monkeys to the optical stimulus of “looming”. Science 136, 982–983 (1962).
King, S.M. & Cowey, A. Defensive responses to looming visual stimuli in monkeys with unilateral striate cortex ablation. Neuropsychologia 30, 1017–1024 (1992).
Waldeck, R.F. & Gruberg, E.R. Studies on the optic chiasm of the leopard frog. I. Selective loss of visually elicited avoidance behavior after optic chiasm hemisection. Brain Behav. Evol. 46, 84–94 (1995).
King, J.G. Jr., Lettvin, J.Y. & Gruberg, E.D. Selective, unilateral, reversible loss of behavioral responses to looming stimuli after injection of tetrodotoxin of cadmium chloride into the frog optic nerve. Brain Res. 841, 20–26 (1999).
Ishikane, H., Gangi, M., Honda, S. & Tachibana, M. Synchronized retinal oscillations encode essential information for escape behavior in frogs. Nat. Neurosci. 8, 1087–1095 (2005).
Ball, W. & Tronick, E. Infant responses to impending collision: optical and real. Science 171, 818–820 (1971).
King, S.M., Dykeman, C., Redgrave, P. & Dean, P. Use of a distracting task to obtain defensive head movements to looming visual stimuli by human adults in a laboratory setting. Perception 21, 245–259 (1992).
Hatsopoulos, N., Gabbiani, F. & Laurent, G. Elementary computation of object approach by wide-field visual neuron. Science 270, 1000–1003 (1995).
Gabbiani, F., Krapp, H.G. & Laurent, G. Computation of object approach by a wide-field, motion-sensitive neuron. J. Neurosci. 19, 1122–1141 (1999).
Gabbiani, F., Cohen, I. & Laurent, G. Time-dependent activation of feed-forward inhibition in a looming-sensitive neuron. J. Neurophysiol. 94, 2150–2161 (2005).
Sun, H. & Frost, B.J. Computation of different optical variables of looming objects in pigeon nucleus rotundus neurons. Nat. Neurosci. 1, 296–303 (1998).
Huberman, A.D. et al. Architecture and activity-mediated refinement of axonal projections from a mosaic of genetically identified retinal ganglion cells. Neuron 59, 425–438 (2008).
Kim, I.J., Zhang, Y., Yamagata, M., Meister, M. & Sanes, J.R. Molecular identification of a retinal cell type that responds to upward motion. Nature 452, 478–482 (2008).
Haverkamp, S. & Wässle, H. Immunocytochemical analysis of the mouse retina. J. Comp. Neurol. 424, 1–23 (2000).
Manookin, M.B., Beaudoin, D.L., Ernst, Z.R., Flagel, L.J. & Demb, J.B. Disinhibition combines with excitation to extend the operating range of the OFF visual pathway in daylight. J. Neurosci. 28, 4136–4150 (2008).
Roska, B. & Werblin, F. Vertical interactions across ten parallel, stacked representations in the mammalian retina. Nature 410, 583–587 (2001).
Fried, S.I., Münch, T.A. & Werblin, F.S. Mechanisms and circuitry underlying directional selectivity in the retina. Nature 420, 411–414 (2002).
Roska, B., Molnar, A. & Werblin, F.S. Parallel processing in retinal ganglion cells: how integration of space-time patterns of excitation and inhibition form the spiking output. J. Neurophysiol. 95, 3810–3822 (2006).
Slaughter, M.M. & Miller, R.F. 2-Amino-4-phosphonobutyric acid: a new pharmacological tool for retina research. Science 211, 182–185 (1981).
Belgum, J.H., Dvorak, D.R., McReynolds, J.S. & Miyachi, E. Push-pull effect of surround illumination on excitatory and inhibitory inputs to mudpuppy retinal ganglion cells. J. Physiol. (Lond.) 388, 233–243 (1987).
McGuire, B.A., Stevens, J.K. & Sterling, P. Microcircuitry of beta ganglion cells in cat retina. J. Neurosci. 6, 907–918 (1986).
Mills, S.L., O'Brien, J.J., Li, W., O'Brien, J. & Massey, S.C. Rod pathways in the mammalian retina use connexin 36. J. Comp. Neurol. 436, 336–350 (2001).
Feigenspan, A., Teubner, B., Willecke, K. & Weiler, R. Expression of neuronal connexin36 in AII amacrine cells of the mammalian retina. J. Neurosci. 21, 230–239 (2001).
Massey, S.C. et al. Multiple neuronal connexins in the mammalian retina. Cell Commun. Adhes. 10, 425–430 (2003).
Bloomfield, S.A. & Dacheux, R.F. Rod vision: pathways and processing in the mammalian retina. Prog. Retin. Eye Res. 20, 351–384 (2001).
Pourcho, R.G. & Goebel, D.J. A combined Golgi and autoradiographic study of (3H)glycine-accumulating amacrine cells in the cat retina. J. Comp. Neurol. 233, 473–480 (1985).
Veruki, M.L. & Hartveit, E. Electrical synapses mediate signal transmission in the rod pathway of the mammalian retina. J. Neurosci. 22, 10558–10566 (2002).
Geraghty, R.J., Krummenacher, C., Cohen, G.H., Eisenberg, R.J. & Spear, P.G. Entry of alphaherpesviruses mediated by poliovirus receptor-related protein 1 and poliovirus receptor. Science 280, 1618–1620 (1998).
Cohen, E.D. & Miller, R.F. The network-selective actions of quinoxalines on the neurocircuitry operations of the rabbit retina. Brain Res. 831, 206–228 (1999).
Murphy, G.J. & Rieke, F. Signals and noise in an inhibitory interneuron diverge to control activity in nearby retinal ganglion cells. Nat. Neurosci. 11, 318–326 (2008).
Xin, D. & Bloomfield, S.A. Comparison of the responses of AII amacrine cells in the dark- and light-adapted rabbit retina. Vis. Neurosci. 16, 653–665 (1999).
Rice, D.S. & Curran, T. Disabled-1 is expressed in type AII amacrine cells in the mouse retina. J. Comp. Neurol. 424, 327–338 (2000).
Huang, L. et al. G protein subunit G gamma 13 is coexpressed with G alpha o, G beta 3, and G beta 4 in retinal ON bipolar cells. J. Comp. Neurol. 455, 1–10 (2003).
Greferath, U., Grunert, U. & Wassle, H. Rod bipolar cells in the mammalian retina show protein kinase C-like immunoreactivity. J. Comp. Neurol. 301, 433–442 (1990).
Oyster, C.W. The analysis of image motion by the rabbit retina. J. Physiol. (Lond.) 199, 613–635 (1968).
Olveczky, B.P., Baccus, S.A. & Meister, M. Segregation of object and background motion in the retina. Nature 423, 401–408 (2003).
Franconeri, S.L. & Simons, D.J. Moving and looming stimuli capture attention. Percept. Psychophys. 65, 999–1010 (2003).
Bradley, D.C. & Goyal, M.S. Velocity computation in the primate visual system. Nat. Rev. Neurosci. 9, 686–695 (2008).
Roska, B., Nemeth, E. & Werblin, F.S. Response to change is facilitated by a three-neuron disinhibitory pathway in the tiger salamander retina. J. Neurosci. 18, 3451–3459 (1998).
Volgyi, B., Xin, D. & Bloomfield, S.A. Feedback inhibition in the inner plexiform layer underlies the surround-mediated responses of AII amacrine cells in the mammalian retina. J. Physiol. (Lond.) 539, 603–614 (2002).
Wässle, H. Parallel processing in the mammalian retina. Nat. Rev. Neurosci. 5, 747–757 (2004).
Pang, J.J. et al. Relative contributions of rod and cone bipolar cell inputs to AII amacrine cell light responses in the mouse retina. J. Physiol. (Lond.) 580, 397–410 (2007).
Hippenmeyer, S. et al. A developmental switch in the response of DRG neurons to ETS transcription factor signaling. PLoS Biol. 3, e159 (2005).
Buffelli, M. et al. Genetic evidence that relative synaptic efficacy biases the outcome of synaptic competition. Nature 424, 430–434 (2003).
Metzger, D. & Feil, R. Engineering the mouse genome by site-specific recombination. Curr. Opin. Biotechnol. 10, 470–476 (1999).
Deans, M.R., Gibson, J.R., Sellitto, C., Connors, B.W. & Paul, D.L. Synchronous activity of inhibitory networks in neocortex requires electrical synapses containing connexin36. Neuron 31, 477–485 (2001).
Acknowledgements
We are grateful to S. Arber (Friedrich Miescher Institute), D. Paul (Harvard Medical School) and J. Sanes (Harvard University) for providing mouse lines and Robert Margolskee (Mount Sinai School of Medicine) for providing the Gγ13 antibody. We are grateful for the technical assistance of S. Djaffer, B. Gross Scherf and Y. Shimada. We thank members of the Roska lab, P. Lagali, P. Caroni, R. Friedrich and A. Lüthi for comments on the manuscript. The study was supported by Friedrich Miescher Institute funds, a US Office of Naval Research Naval International Cooperative Opportunities in Science and Technology program grant, a Marie Curie Excellence Grant, a Human Frontier Science Program Young Investigator grant, a National Centers of Competence in Research in Genetics grant and a European Union HEALTH-F2-223156 grant to B.R., a Marie Curie Postdoctoral Fellowship to T.A.M., the Centre National de la Recherche Scientifique through the Unité Mixte de Recherche 8550 to R.A.d.S.
Author information
Authors and Affiliations
Contributions
T.A.M. performed electrophysiological experiments, designed experiments and model, and wrote manuscript; R.A.S. designed experiments and model and wrote manuscript; S.S. performed immunohistochemistry; T.J.V. performed electrophysiological experiments, G.B.A. performed and designed electrophysiological experiments; and B.R. designed experiments and model and wrote manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 4881 kb)
Rights and permissions
About this article
Cite this article
Münch, T., da Silveira, R., Siegert, S. et al. Approach sensitivity in the retina processed by a multifunctional neural circuit. Nat Neurosci 12, 1308–1316 (2009). https://doi.org/10.1038/nn.2389
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2389
This article is cited by
-
A Non-canonical Excitatory PV RGC–PV SC Visual Pathway for Mediating the Looming-evoked Innate Defensive Response
Neuroscience Bulletin (2023)
-
Neural Circuit Mechanisms Involved in Animals’ Detection of and Response to Visual Threats
Neuroscience Bulletin (2023)
-
Suppression without inhibition: how retinal computation contributes to saccadic suppression
Communications Biology (2022)
-
Classical center-surround receptive fields facilitate novel object detection in retinal bipolar cells
Nature Communications (2022)
-
Anthropogenic Marsh Impoundments Alter Collective Tendency in Schooling Fish
Estuaries and Coasts (2022)