Abstract
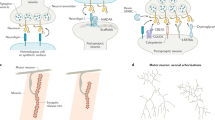
Ephrin ligands and their cognate Eph receptors guide axons during neural development and regulate synapse formation and neuronal plasticity in the adult. Because ephrins are tethered to the plasma membrane and possess reverse signaling properties, the Eph-ephrin system can function in a bidirectional, contact-mediated fashion between two opposing cells. Eph receptors expressed on dendrites are activated by ephrins (on axons or on astrocytes) and regulate spine and synapse formation. They also participate in activity-induced long-term changes in synaptic strength such as long-term potentiation (LTP). When expressed on axon terminals, ephrins promote presynaptic differentiation and enhance neurotransmitter release, thereby supporting presynaptic forms of LTP. In some cases, Eph receptors can simply act as ligands for ephrins without any requirement for Eph receptor signaling, suggesting that the system does not always function bidirectionally.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Yamada, S. & Nelson, W.J. Synapses: sites of cell recognition, adhesion and functional specification. Annu. Rev. Biochem. 76, 267–294 (2007).
Pasquale, E.B. Eph-ephrin bidirectional signaling in physiology and disease. Cell 133, 38–52 (2008).
Egea, J. & Klein, R. Bidirectional Eph-ephrin signaling during axon guidance. Trends Cell Biol. 17, 230–238 (2007).
Flanagan, J.G. Neural map specification by gradients. Curr. Opin. Neurobiol. 16, 59–66 (2006).
Bliss, T.V. & Collingridge, G.L. A synaptic model of memory: long-term potentiation in the hippocampus. Nature 361, 31–39 (1993).
Bourne, J.N. & Harris, K.M. Balancing structure and function at hippocampal dendritic spines. Annu. Rev. Neurosci. 31, 47–67 (2008).
Henkemeyer, M., Itkis, O.S., Ngo, M., Hickmott, P.W. & Ethell, I.M. Multiple EphB receptor tyrosine kinases shape dendritic spines in the hippocampus. J. Cell Biol. 163, 1313–1326 (2003).
Kayser, M.S., McClelland, A.C., Hughes, E.G. & Dalva, M.B. Intracellular and trans-synaptic regulation of glutamatergic synaptogenesis by EphB receptors. J. Neurosci. 26, 12152–12164 (2006).
Kayser, M.S., Nolt, M.J. & Dalva, M.B. EphB receptors couple dendritic filopodia motility to synapse formation. Neuron 59, 56–69 (2008).
Irie, F. & Yamaguchi, Y. EphB receptors regulate dendritic spine development via intersectin, Cdc42 and N-WASP. Nat. Neurosci. 5, 1117–1118 (2002).
Penzes, P. et al. Rapid induction of dendritic spine morphogenesis by trans-synaptic ephrinB-EphB receptor activation of the Rho-GEF kalirin. Neuron 37, 263–274 (2003).
Moeller, M.L., Shi, Y., Reichardt, L.F. & Ethell, I.M. EphB receptors regulate dendritic spine morphogenesis through the recruitment/phosphorylation of focal adhesion kinase and RhoA activation. J. Biol. Chem. 281, 1587–1598 (2006).
Dalva, M.B. et al. EphB receptors interact with NMDA receptors and regulate excitatory synapse formation. Cell 103, 945–956 (2000).
Takasu, M.A., Dalva, M.B., Zigmond, R.E. & Greenberg, M.E. Modulation of NMDA receptor–dependent calcium influx and gene expression through EphB receptors. Science 295, 491–495 (2002).
Tolias, K.F. et al. The Rac1-GEF Tiam1 couples the NMDA receptor to the activity-dependent development of dendritic arbors and spines. Neuron 45, 525–538 (2005).
Tolias, K.F. et al. The Rac1 guanine nucleotide exchange factor Tiam1 mediates EphB receptor–dependent dendritic spine development. Proc. Natl. Acad. Sci. USA 104, 7265–7270 (2007).
Murai, K.K., Nguyen, L.N., Irie, F., Yamaguchi, Y. & Pasquale, E.B. Control of hippocampal dendritic spine morphology through ephrin-A3/EphA4 signaling. Nat. Neurosci. 6, 153–160 (2003).
Tremblay, M.E. et al. Localization of EphA4 in axon terminals and dendritic spines of adult rat hippocampus. J. Comp. Neurol. 501, 691–702 (2007).
Fu, W.Y. et al. Cdk5 regulates EphA4-mediated dendritic spine retraction through an ephexin1-dependent mechanism. Nat. Neurosci. 10, 67–76 (2007).
Sahin, M. et al. Eph-dependent tyrosine phosphorylation of ephexin1 modulates growth cone collapse. Neuron 46, 191–204 (2005).
Bourgin, C., Murai, K.K., Richter, M. & Pasquale, E.B. The EphA4 receptor regulates dendritic spine remodeling by affecting beta1-integrin signaling pathways. J. Cell Biol. 178, 1295–1307 (2007).
Richter, M., Murai, K.K., Bourgin, C., Pak, D.T. & Pasquale, E.B. The EphA4 receptor regulates neuronal morphology through SPAR-mediated inactivation of Rap GTPases. J. Neurosci. 27, 14205–14215 (2007).
Zhou, L. et al. EphA4 signaling regulates phospholipase Cgamma1 activation, cofilin membrane association and dendritic spine morphology. J. Neurosci. 27, 5127–5138 (2007).
Lim, B.K., Matsuda, N. & Poo, M.M. Ephrin-B reverse signaling promotes structural and functional synaptic maturation in vivo. Nat. Neurosci. 11, 160–169 (2008).
Bouzioukh, F. et al. Tyrosine phosphorylation sites in ephrinB2 are required for hippocampal long-term potentiation, but not long-term depression. J. Neurosci. 27, 11279–11288 (2007).
Grunwald, I.C. et al. Hippocampal plasticity requires postsynaptic ephrinBs. Nat. Neurosci. 7, 33–40 (2004).
Segura, I., Essmann, C.L., Weinges, S. & Acker-Palmer, A. Grb4 and GIT1 transduce ephrinB reverse signals modulating spine morphogenesis and synapse formation. Nat. Neurosci. 10, 301–310 (2007).
Aoto, J. et al. Postsynaptic ephrinB3 promotes shaft glutamatergic synapse formation. J. Neurosci. 27, 7508–7519 (2007).
Cowan, C.A. & Henkemeyer, M. The SH2/SH3 adaptor Grb4 transduces B-ephrin reverse signals. Nature 413, 174–179 (2001).
Grunwald, I.C. et al. Kinase-independent requirement of EphB2 receptors in hippocampal synaptic plasticity. Neuron 32, 1027–1040 (2001).
Henderson, J.T. et al. The receptor tyrosine kinase EphB2 regulates NMDA-dependent synaptic function. Neuron 32, 1041–1056 (2001).
Contractor, A. et al. Trans-synaptic Eph receptor–ephrin signaling in hippocampal mossy fiber LTP. Science 296, 1864–1869 (2002).
Ghosh, A. Neurobiology. Learning more about NMDA receptor regulation. Science 295, 449–451 (2002).
Aoto, J. & Chen, L. Bidirectional ephrin/Eph signaling in synaptic functions. Brain Res. 1184, 72–80 (2007).
Liebl, D.J., Morris, C.J., Henkemeyer, M. & Parada, L.F. mRNA expression of ephrins and Eph receptor tyrosine kinases in the neonatal and adult mouse central nervous system. J. Neurosci. Res. 71, 7–22 (2003).
Palmer, A. et al. EphrinB phosphorylation and reverse signaling: regulation by Src kinases and PTP-BL phosphatase. Mol. Cell 9, 725–737 (2002).
Makinen, T. et al. PDZ interaction site in ephrinB2 is required for the remodeling of lymphatic vasculature. Genes Dev. 19, 397–410 (2005).
Lu, Y.M., Roder, J.C., Davidow, J. & Salter, M.W. Src activation in the induction of long-term potentiation in CA1 hippocampal neurons. Science 279, 1363–1367 (1998).
Rodenas-Ruano, A., Perez-Pinzon, M.A., Green, E.J., Henkemeyer, M. & Liebl, D.J. Distinct roles for ephrinB3 in the formation and function of hippocampal synapses. Dev. Biol. 292, 34–45 (2006).
Armstrong, J.N. et al. B-ephrin reverse signaling is required for NMDA-independent long-term potentiation of mossy fibers in the hippocampus. J. Neurosci. 26, 3474–3481 (2006).
Deininger, K. et al. The Rab5 guanylate exchange factor Rin1 regulates endocytosis of the EphA4 receptor in mature excitatory neurons. Proc. Natl. Acad. Sci. USA 105, 12539–12544 (2008).
Cowan, C.W. et al. Vav family GEFs link activated Ephs to endocytosis and axon guidance. Neuron 46, 205–217 (2005).
Essmann, C.L. et al. Serine phosphorylation of ephrinB2 regulates trafficking of synaptic AMPA receptors. Nat. Neurosci. 11, 1035–1043 (2008).
Lauterbach, J. & Klein, R. Release of full-length EphB2 receptors from hippocampal neurons to cocultured glial cells. J. Neurosci. 26, 11575–11581 (2006).
Nishida, H. & Okabe, S. Direct astrocytic contacts regulate local maturation of dendritic spines. J. Neurosci. 27, 331–340 (2007).
Nestor, M.W., Mok, L.P., Tulapurkar, M.E. & Thompson, S.M. Plasticity of neuron-glial interactions mediated by astrocytic EphARs. J. Neurosci. 27, 12817–12828 (2007).
Lim, Y.S. et al. p75(NTR) mediates ephrin-A reverse signaling required for axon repulsion and mapping. Neuron 59, 746–758 (2008).
Zagrebelsky, M. et al. The p75 neurotrophin receptor negatively modulates dendrite complexity and spine density in hippocampal neurons. J. Neurosci. 25, 9989–9999 (2005).
Acknowledgements
I'd like to thank K. Deininger, S. Paixão and A. Filosa for critical reading of the manuscript, and R. Schorner and C. Erlacher for help with the figures.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Klein, R. Bidirectional modulation of synaptic functions by Eph/ephrin signaling. Nat Neurosci 12, 15–20 (2009). https://doi.org/10.1038/nn.2231
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2231
This article is cited by
-
Fear Conditioning Leads to Enduring Alterations in RNA Transcripts in Hippocampal Neuropil that are Dependent on EphB2 Forward Signaling
Molecular Neurobiology (2023)
-
Unraveling the Potential of EphA4: A Breakthrough Target and Beacon of Hope for Neurological Diseases
Cellular and Molecular Neurobiology (2023)
-
EphrinA4 mimetic peptide impairs fear conditioning memory reconsolidation in lateral amygdala
Scientific Reports (2022)
-
EphA4 Obstructs Spinal Cord Neuron Regeneration by Promoting Excessive Activation of Astrocytes
Cellular and Molecular Neurobiology (2022)
-
Neuronal hibernation following hippocampal demyelination
Acta Neuropathologica Communications (2021)